Advanced Synthesis of 2-Guanidino-Thiazolium Compounds for Commercial Famotidine Production
Advanced Synthesis of 2-Guanidino-Thiazolium Compounds for Commercial Famotidine Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical gastric acid inhibitors like Famotidine, and patent CN1033521A presents a significant technological leap in this domain. This intellectual property discloses novel 2-guanidino-thiazolium compounds and their preparation methods, specifically designed to serve as high-quality intermediates in the manufacture of Famotidine. Unlike conventional pathways that struggle with toxicity and impurity profiles, this invention leverages bicyclic amidine salts, such as those derived from DBU or DBN, to achieve quantitative yields in key transformation steps. The technical breakthrough lies in the stabilization of the thiolate species through salt formation, which effectively bypasses the oxidative degradation and disulfide formation that plague traditional alkaline hydrolysis methods. For R&D directors and process chemists, this represents a viable pathway to enhance purity specifications while simultaneously addressing critical environmental and safety concerns associated with legacy manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-guanidino-4-thiazolyl compounds has been fraught with significant operational hazards and chemical inefficiencies that compromise both yield and product quality. Traditional methodologies often necessitate the use of highly toxic and flammable hydrogen sulfide gas, requiring rigorous containment systems and complex waste gas treatment protocols to ensure worker safety and environmental compliance. Furthermore, alternative routes involving 1,3-dichloroacetone introduce carcinogenic risks and generate reaction mixtures contaminated with sulfides and disulfides that are notoriously difficult to separate. The reliance on sodium hydroxide for in situ thiolate generation frequently leads to the formation of brown, viscous oily substances that resist dissolution in common organic solvents like methylene chloride, thereby rendering effective extraction and purification nearly impossible. These inherent flaws result in low overall yields, inconsistent purity profiles, and substantial increases in production costs due to the extensive downstream processing required to remove persistent impurities.
The Novel Approach
The methodology outlined in CN1033521A fundamentally reengineers the synthetic landscape by introducing stable bicyclic amidine salts as key intermediates, effectively neutralizing the vulnerabilities of previous techniques. By reacting S-(2-guanidino-thiazole-4-yl-methyl) isothiouronium salts with bases like 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), the process generates a precipitable salt that can be isolated with high purity, typically ranging from 95% to 98%. This solid intermediate eliminates the handling of free thiols in alkaline media, thereby preventing oxidation to disulfides and ensuring a clean starting material for subsequent beta-cyanoethylation. The reaction with acrylonitrile proceeds in aqueous solution with almost quantitative conversion, allowing for the simple filtration and recycling of the bicyclic amidine base. This shift from unstable, oily intermediates to crystalline, isolable salts streamlines the workflow, reduces solvent consumption, and provides a reproducible foundation for large-scale manufacturing operations.
Mechanistic Insights into Bicyclic Amidine Salt Catalysis and Silylation
The core mechanistic advantage of this patent lies in the precise control of nucleophilicity and electrophilicity through salt formation and silylation chemistry. Initially, the bicyclic amidine acts as a non-nucleophilic base to deprotonate the isothiouronium species, generating a stabilized thiolate anion paired with the bulky amidinium cation. This ion pair exhibits enhanced solubility characteristics and reactivity towards acrylonitrile, facilitating a rapid Michael addition to form the propionitrile derivative without the side reactions typical of free thiolates. Subsequently, the transformation of the nitrile group into the requisite amidine functionality is achieved through a novel imido-ester pathway using dioxane hydrochloride complexes. This reagent system allows for the in situ generation of imino-chlorides which react selectively with methanol to form propionylimidates, avoiding the harsh conditions and excessive acid usage seen in prior art.
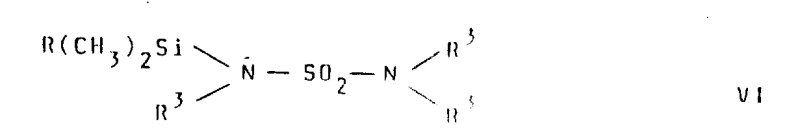
Following the formation of the imido-ester, the synthesis employs advanced silylation strategies to construct the final sulfamoyl amidine linkage. The process utilizes N-trialkylsilyl sulfonamides, such as the structure depicted below, which act as protected nucleophiles capable of reacting with the imido-ester under mild, anhydrous conditions. This approach prevents the hydrolysis of sensitive functional groups and ensures that the sulfonamide moiety is introduced with high regioselectivity. The use of trimethylsilyl derivatives creates a transient intermediate that is stable enough to be handled yet reactive enough to undergo the final condensation efficiently.
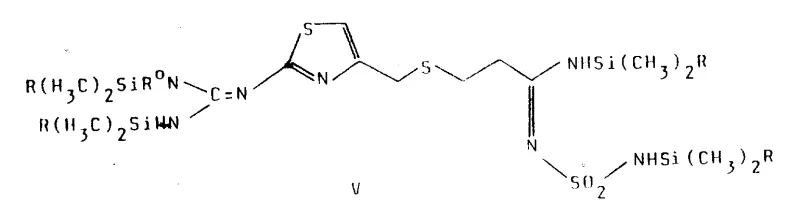
The final stage involves the conversion of the silylated intermediate into the target Famotidine structure through a controlled desilylation process. As illustrated in the complex molecular architecture below, the fully assembled precursor contains multiple silyl protecting groups that mask the reactive nitrogen centers during the coupling phase. Treatment with stoichiometric amounts of alcohol, such as methanol or isopropanol, at room temperature cleaves these silicon-nitrogen bonds, releasing the free amine and sulfonamide functionalities to yield the final active pharmaceutical ingredient. This step is characterized by high selectivity and avoids the formation of polymeric byproducts, resulting in a clean reaction profile that simplifies the final crystallization and purification of the polymorphic forms of Famotidine.
How to Synthesize Famotidine Intermediates Efficiently
The synthesis protocol described in the patent offers a standardized approach for producing high-purity intermediates suitable for regulatory submission and commercial supply. The process begins with the preparation of the DBU or DBN salt of the guanidino-thiazole precursor, followed by cyanoethylation and subsequent functional group manipulations using silyl chemistry. Detailed operational parameters, including specific molar ratios, temperature controls, and solvent systems, are critical for replicating the high yields reported in the examples. For process engineers looking to implement this technology, adherence to the specific precipitation and washing steps is essential to maintain the integrity of the intermediate salts and prevent contamination.
- Formation of Bicyclic Amidine Salt: React S-(2-guanidino-thiazole-4-yl-methyl) isothiouronium dihydrochloride with DBU or DBN in acetonitrile/water to precipitate the stable salt intermediate.
- Beta-Cyanoethylation: Treat the isolated salt with acrylonitrile in aqueous solution to quantitatively form 3-(2-guanidino-thiazole-4-yl-methylthio)propionitrile.
- Silylation and Cyclization: Convert the nitrile to an imido-ester using dioxane hydrochloride, then react with N-trimethylsilyl sulfonamide derivatives followed by desilylation to obtain Famotidine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthetic route offers substantial benefits regarding cost structure and supply chain resilience. By eliminating the need for hazardous hydrogen sulfide gas and carcinogenic chloroacetone, manufacturers can significantly reduce expenditures related to specialized safety equipment, waste disposal, and regulatory compliance monitoring. The ability to isolate stable solid intermediates rather than handling unstable oils minimizes material loss during transfer and storage, leading to improved overall mass balance and reduced raw material consumption. Furthermore, the quantitative nature of the key reaction steps ensures predictable output volumes, allowing supply chain planners to forecast production capacity with greater accuracy and reliability.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the elimination of expensive purification steps required to remove disulfide impurities and heavy metal contaminants found in traditional routes. The recycling of the bicyclic amidine base, such as DBU, from the aqueous mother liquor further lowers the effective cost of goods sold by reducing the consumption of high-value reagents. Additionally, the simplified work-up procedures, which rely on filtration rather than complex extractions, decrease solvent usage and energy consumption associated with distillation and drying operations.
- Enhanced Supply Chain Reliability: Utilizing stable, isolable intermediates mitigates the risk of batch failures caused by the degradation of sensitive thiol species during storage or transport. The robustness of the salt formation step ensures a consistent supply of high-quality starting material for downstream reactions, reducing the variability that often leads to production delays. This stability allows for the strategic stocking of key intermediates, providing a buffer against raw material shortages and enabling a more responsive manufacturing schedule to meet market demand fluctuations.
- Scalability and Environmental Compliance: The avoidance of toxic gases and the reduction of organic solvent waste align perfectly with increasingly stringent environmental regulations, facilitating smoother permitting processes for facility expansion. The aqueous nature of the cyanoethylation step reduces the fire load in the production plant and simplifies wastewater treatment requirements compared to processes relying heavily on chlorinated solvents. These factors collectively enhance the long-term viability of the manufacturing site and reduce the risk of operational shutdowns due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology for Famotidine production. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical advantages of the new method. Understanding these details is crucial for stakeholders evaluating the feasibility of technology transfer or sourcing intermediates produced via this route.
Q: How does this patent improve safety compared to traditional Famotidine synthesis?
A: Traditional methods often rely on hazardous hydrogen sulfide gas and carcinogenic 1,3-dichloroacetone. This patent introduces a route using stable bicyclic amidine salts (DBU/DBN), eliminating the need for handling toxic gases and reducing the formation of difficult-to-remove disulfide impurities.
Q: What is the role of the silylating agent in the final steps?
A: The process utilizes N-trialkylsilyl sulfonamides and silylated imido-esters to facilitate the formation of the sulfamoyl amidine structure under mild, non-hydroxyl solvent conditions. This prevents premature hydrolysis and ensures high selectivity before the final desilylation step yields the active pharmaceutical ingredient.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the patent describes quantitative yields for the salt formation and cyanoethylation steps, with robust precipitation methods that simplify filtration and washing. The avoidance of complex extraction procedures for brown viscous oils significantly enhances scalability and operational efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Famotidine Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of pharmaceutical intermediate manufacturing, leveraging advanced synthetic methodologies like those described in CN1033521A to deliver superior value to global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-guanidino-thiazolium compounds meets the highest international standards for safety and efficacy.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your supply chain goals. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits of switching to our high-purity intermediates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific project requirements, ensuring a partnership built on transparency, quality, and mutual success.
