Advanced Catalytic Hydrolysis for Commercial Scale Pentafluorophenol Production
Advanced Catalytic Hydrolysis for Commercial Scale Pentafluorophenol Production
The rapid evolution of the flat-panel display industry has necessitated the development of highly specialized chemical intermediates capable of meeting stringent electro-optical performance criteria. Patent CN102887817A introduces a transformative methodology for the synthesis of 2,3,4,5,6-pentafluorophenol, a critical building block for next-generation TFT liquid crystal materials. This innovation addresses the growing demand for polyfluoro-class monomers that enhance macromolecular dipole moments while maintaining low viscosity and high dielectric anisotropy. By leveraging a novel copper-catalyzed hydrolysis pathway, the technology enables the production of high-purity intermediates essential for improving response times and display clarity in advanced electronic devices. The strategic implementation of this synthetic route offers significant advantages in terms of process simplicity and environmental compatibility compared to legacy manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing polyfluorinated phenols often suffer from severe inefficiencies that hinder large-scale commercial viability and economic feasibility. Conventional methodologies frequently rely on harsh reaction conditions that can lead to the formation of complex impurity profiles, necessitating costly and time-consuming purification steps to meet electronic grade specifications. Furthermore, many established processes exhibit poor atom economy, generating substantial quantities of hazardous waste streams that complicate regulatory compliance and increase the overall environmental footprint of the manufacturing operation. The reliance on expensive or difficult-to-handle reagents in older techniques also introduces supply chain vulnerabilities and operational risks that can disrupt continuous production schedules. These inherent limitations create a significant barrier to entry for manufacturers seeking to optimize cost structures while maintaining the rigorous quality standards required by the global liquid crystal market.
The Novel Approach
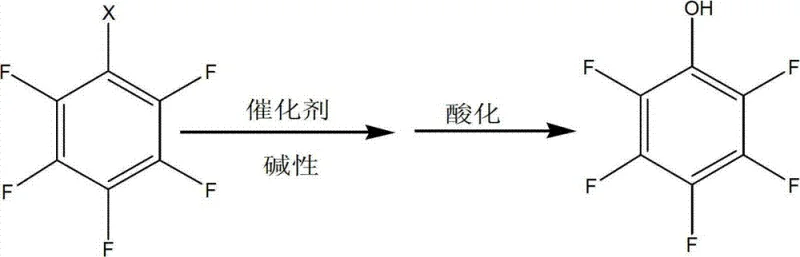
The patented methodology revolutionizes the production landscape by utilizing a direct hydrolysis strategy that converts readily available 2,3,4,5,6-pentafluoro-1-halogenated benzenes into the desired phenol derivative with exceptional efficiency. This approach eliminates the need for multi-step functional group transformations, thereby streamlining the workflow and reducing the cumulative loss of material typically associated with longer synthetic sequences. The use of a robust copper-based catalytic system allows the reaction to proceed under relatively mild thermal conditions, significantly lowering energy consumption and enhancing operational safety within the production facility. By optimizing the stoichiometric ratios of alkali and catalyst, the process achieves high conversion rates while minimizing the generation of by-products, ensuring a cleaner reaction profile. This streamlined architecture not only improves the overall yield but also simplifies the downstream processing requirements, making it an ideal candidate for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Copper-Catalyzed Hydrolysis
The core of this technological advancement lies in the sophisticated interaction between the copper catalyst and the electron-deficient aromatic ring of the pentafluorohalobenzene substrate. The catalytic cycle likely involves the coordination of the copper species to the halogen substituent, facilitating the nucleophilic attack by hydroxide ions generated from the alkaline medium. This activation lowers the energy barrier for the substitution reaction, allowing the displacement of the halogen atom with a hydroxyl group to occur efficiently even with less reactive halogen variants like fluorine or chlorine. The selection of red copper oxide as the preferred catalyst suggests a specific oxidation state mechanism that maximizes turnover frequency while maintaining catalyst stability throughout the reaction duration. Understanding these mechanistic nuances is crucial for R&D directors aiming to replicate or further optimize the process for specific purity targets required in high-end optical applications.
Impurity control is inherently managed through the precise regulation of reaction parameters such as temperature and alkali concentration, which dictate the selectivity of the hydrolysis step. The patent data indicates that maintaining the reaction temperature within the range of 130°C to 160°C prevents the degradation of the sensitive polyfluorinated structure while ensuring complete conversion of the starting material. Additionally, the subsequent acidification step is carefully controlled to precipitate the product without inducing side reactions that could compromise the integrity of the phenolic hydroxyl group. This dual-stage control mechanism ensures that the final product exhibits a purity profile suitable for direct use in liquid crystal formulation without extensive recrystallization. Such robust control over the chemical environment is essential for producing high-purity OLED material and liquid crystal intermediates that demand consistent batch-to-batch reproducibility.
How to Synthesize 2,3,4,5,6-Pentafluorophenol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to industrial production with minimal technical friction. The process begins with the preparation of an aqueous alkaline solution containing specific ratios of sodium hydroxide and sodium carbonate, which serves as both the reactant source and the pH buffer for the system. Following the addition of the copper catalyst and the halogenated precursor, the mixture is subjected to hydrothermal conditions in a sealed autoclave to drive the hydrolysis to completion.
- Prepare the reaction mixture by combining 2,3,4,5,6-pentafluoro-1-halogenated benzene with a copper-based catalyst and an alkaline solution.
- Conduct the hydrolysis reaction in an autoclave at elevated temperatures between 100°C and 180°C to form the sodium salt intermediate.
- Filter the reaction mixture and perform acidification using mineral acids like hydrochloric acid to precipitate the final 2,3,4,5,6-pentafluorophenol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route presents a compelling value proposition centered around raw material accessibility and process robustness. The primary starting materials, specifically pentafluorohalobenzenes, are commodity chemicals with well-established global supply chains, reducing the risk of sourcing bottlenecks that often plague specialty chemical production. The elimination of exotic or proprietary reagents means that purchasing teams can leverage existing vendor relationships to secure competitive pricing, directly contributing to significant cost savings in the overall manufacturing budget. Furthermore, the simplified two-step nature of the process reduces the requirement for specialized equipment and complex infrastructure, allowing for faster deployment of production capacity in existing facilities. This operational agility ensures that supply chain heads can respond more rapidly to fluctuating market demands for liquid crystal components without incurring prohibitive capital expenditures.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom economy and the use of inexpensive, earth-abundant copper catalysts instead of precious metals. By avoiding the use of costly transition metals like palladium or platinum, the variable costs associated with catalyst recovery and metal residue removal are drastically reduced. The high yield reported in the patent examples implies that less raw material is wasted per unit of output, further enhancing the gross margin potential for manufacturers. Additionally, the ability to recover and recycle solvents such as methyl tert-butyl ether adds another layer of financial efficiency to the operation. These factors combine to create a lean manufacturing model that is highly resilient to raw material price volatility.
- Enhanced Supply Chain Reliability: The reliance on standard industrial chemicals for both the substrate and the reagents ensures a stable and predictable supply chain that is less susceptible to geopolitical disruptions. Since the process does not depend on single-source proprietary catalysts, manufacturers have the flexibility to qualify multiple suppliers for key inputs, thereby mitigating the risk of production stoppages. The mild reaction conditions also reduce the wear and tear on reactor vessels and associated piping, leading to lower maintenance downtime and higher asset utilization rates over time. This reliability is critical for maintaining the continuous flow of materials required by just-in-time manufacturing systems in the electronics sector.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates fewer hazardous by-products compared to traditional nitration or sulfonation routes, simplifying waste treatment and disposal protocols. The aqueous nature of the hydrolysis step reduces the volume of organic solvents required, aligning with green chemistry principles and helping facilities meet increasingly strict environmental regulations. The scalability of the autoclave-based reaction is well-understood in the chemical industry, allowing for straightforward scale-up from pilot plants to multi-ton commercial production without significant re-engineering. This ease of scale-up ensures that the commercial scale-up of complex polymer additives or liquid crystal precursors can be achieved with confidence and speed.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pentafluorophenol synthesis technology. These insights are derived directly from the experimental data and claims presented in the underlying patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: What catalyst system provides the highest yield for pentafluorophenol synthesis?
A: According to patent data, red copper oxide (Cu2O) serves as the preferred catalyst, often yielding superior results compared to other copper salts when used in conjunction with a mixed alkali system.
Q: Can this method be adapted for different halogenated starting materials?
A: Yes, the process is versatile and accommodates various halogen substituents including fluorine, chlorine, bromine, and iodine, with bromine-substituted precursors often showing optimal reactivity profiles.
Q: How does this synthesis route impact impurity profiles in liquid crystal intermediates?
A: The method achieves high atom economy and utilizes mild reaction conditions relative to traditional harsh substitutions, resulting in product purity levels exceeding 99% which is critical for display applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,4,5,6-Pentafluorophenol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate innovative patent methodologies into reliable commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that clients receive a consistent supply of high-quality intermediates regardless of volume requirements. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2,3,4,5,6-pentafluorophenol meets the exacting standards necessary for high-performance liquid crystal and pharmaceutical applications. Our commitment to quality assurance extends beyond simple testing, encompassing a holistic approach to process optimization that maximizes yield and minimizes impurities.
We invite global partners to collaborate with us to leverage this advanced synthesis technology for their specific product lines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current supply chain needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your operational efficiency and product performance.
