Advanced Synthesis of 5-Alkoxy Triazolopyrimidine Thiones for High-Efficiency Herbicide Production
The global demand for high-efficiency sulfonylurea herbicides continues to drive innovation in the synthesis of their critical heterocyclic precursors. Patent CN1052484C introduces a transformative methodology for preparing 5-alkoxy[1,2,4]triazolo[1,5-c]pyrimidine-2(3H)-thione compounds and their subsequent conversion into valuable 2-chlorosulfonyl derivatives. This technology addresses long-standing challenges in agrochemical intermediate manufacturing by replacing cumbersome multi-step sequences with a more direct, oxidative pathway. For R&D directors and procurement specialists seeking reliable agrochemical intermediate suppliers, understanding this mechanistic shift is crucial for optimizing supply chains and reducing the cost of goods sold in herbicide manufacturing. The patent details a robust route that enhances purity profiles while simplifying the operational complexity typically associated with triazolopyrimidine chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2-chlorosulfonyl-5-alkoxy[1,2,4]triazolo[1,5-c]pyrimidine intermediates relied heavily on multistep processes involving benzylthio or lower alkylthio protecting groups. These conventional routes often suffered from variable yields due to the inherent instability of the intermediates and the difficulty in controlling side reactions during the oxychlorination steps. The presence of sulfur-containing byproducts and the need for extensive purification to remove unreacted thio-ethers resulted in significant waste generation and increased processing costs. Furthermore, the isolation of the final chlorosulfonyl compound was frequently complicated by impurities that degraded the quality of the subsequent sulfonamide herbicides, leading to inconsistent agronomic performance and necessitating costly recrystallization protocols.
The Novel Approach
The innovative strategy outlined in the patent circumvents these issues by utilizing 5-alkoxy[1,2,4]triazolo[1,5-c]pyrimidine-2(3H)-thione compounds as direct starting materials. Instead of relying on alkylthio protection, the process employs a controlled oxidation step to generate stable 2,2'-dithio-bis intermediates, which are then subjected to chloroxidation. This approach not only streamlines the synthetic sequence but also significantly improves the purity of the final product by eliminating the formation of complex sulfur-based impurities. The ability to isolate the dithio-bis intermediate as a crystalline solid offers distinct logistical advantages, allowing for better inventory management and quality control before the final chlorination step.
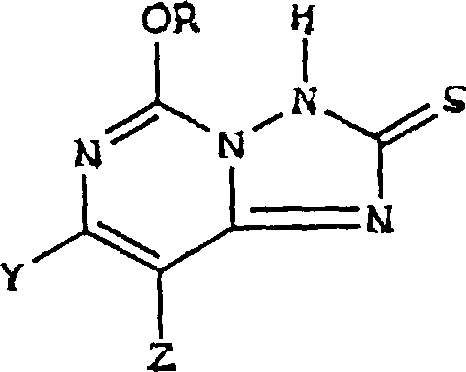
This oxidative transformation is pivotal, as it converts the potentially unstable thione moiety into a robust disulfide linkage that withstands subsequent harsh reaction conditions. The use of hydrogen peroxide as the oxidant is particularly advantageous from an environmental and safety perspective, as it generates water as the primary byproduct, aligning with green chemistry principles increasingly demanded by regulatory bodies and corporate sustainability goals.
Mechanistic Insights into Oxidative Chlorination Pathways
The chemical elegance of this process lies in the precise control of oxidation states and the management of tautomeric equilibria. The starting 5-alkoxy[1,2,4]triazolo[1,5-c]pyrimidine-2(3H)-thione compounds exist in equilibrium with their mercaptan tautomers, a dynamic that can complicate reactivity if not properly managed. The patent elucidates that under the specified reaction conditions, the thione form is preferentially oxidized to the symmetrical disulfide. This selectivity is critical for preventing over-oxidation to sulfonic acids or other degradation products that could compromise the integrity of the heterocyclic core.
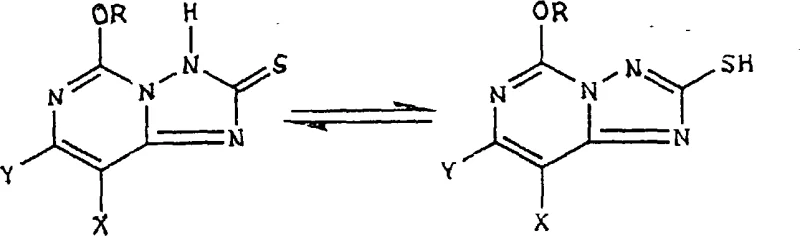
Following the formation of the disulfide intermediate, the subsequent chloroxidation step involves the cleavage of the sulfur-sulfur bond and the introduction of the chlorosulfonyl functionality. This reaction requires careful temperature control and stoichiometric addition of chlorine gas to ensure complete conversion without damaging the sensitive alkoxy and halogen substituents on the pyrimidine ring. The mechanism likely proceeds through the formation of sulfenyl chloride species which are further oxidized to the sulfonyl chloride. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters such as solvent composition and addition rates to maximize yield and minimize the formation of dichloro or des-alkoxy impurities.
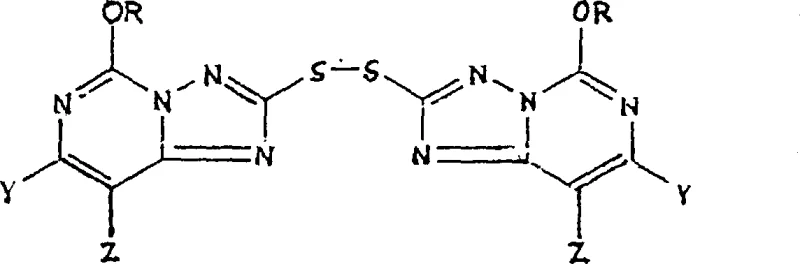
Furthermore, the choice of solvent systems plays a vital role in stabilizing the transition states and facilitating the separation of products. The use of biphasic systems comprising water and organic solvents like methylene chloride allows for efficient extraction of the hydrophobic chlorosulfonyl product while retaining inorganic salts and acidic byproducts in the aqueous phase. This phase behavior is instrumental in achieving the high purity levels required for pharmaceutical and agrochemical applications, ensuring that the final intermediate meets stringent specifications for downstream coupling reactions.
How to Synthesize 5-Alkoxy Triazolopyrimidine Thiones Efficiently
The synthesis of these high-value intermediates requires strict adherence to the optimized protocols regarding reagent stoichiometry and thermal management. The process begins with the preparation of the thione precursor, followed by its immediate conversion to the disulfide to prevent decomposition. Detailed operational guidelines regarding solvent selection, pH control during acidification, and filtration techniques are essential for reproducibility. For a comprehensive breakdown of the specific experimental conditions, temperatures, and workup procedures validated in the patent literature, please refer to the standardized synthesis guide below.
- Oxidize 5-alkoxy[1,2,4]triazolo[1,5-c]pyrimidine-2(3H)-thione compounds using hydrogen peroxide in an inert medium to form 2,2'-dithio-bis intermediates.
- Subject the resulting dithio-bis intermediate to chloroxidation conditions using chlorine gas in a suitable aqueous-organic solvent system.
- Isolate the final 2-chlorosulfonyl-5-alkoxy[1,2,4]triazolo[1,5-c]pyrimidine compound via phase separation and solvent removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers substantial strategic benefits beyond mere technical feasibility. The elimination of expensive and hazardous reagents traditionally used in thio-ether protection strategies translates directly into reduced raw material costs and simplified waste disposal protocols. By streamlining the number of unit operations, manufacturers can achieve higher throughput rates and reduce the overall cycle time for producing critical herbicide intermediates. This efficiency gain is particularly valuable in a volatile market where speed to market and cost competitiveness are paramount for maintaining margin integrity.
- Cost Reduction in Manufacturing: The new process significantly lowers production costs by removing the need for complex protection and deprotection steps involving benzyl or alkyl halides. This reduction in synthetic steps decreases the consumption of solvents and reagents, leading to a leaner manufacturing footprint. Additionally, the improved yield and purity profile reduce the burden on downstream purification units, minimizing product loss and maximizing the effective output per batch. The use of commodity chemicals like hydrogen peroxide and chlorine further ensures that raw material pricing remains stable and predictable, shielding the supply chain from the volatility associated with specialty reagents.
- Enhanced Supply Chain Reliability: By establishing a more robust and direct synthetic pathway, manufacturers can mitigate the risks associated with supply disruptions of niche intermediates. The ability to produce stable disulfide intermediates allows for the creation of strategic stockpiles, ensuring continuity of supply even during periods of high demand or logistical constraints. The simplified process flow also reduces the dependency on specialized equipment or highly skilled operators for complex transformations, making the technology more transferable across different manufacturing sites and enhancing overall supply chain resilience against regional disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing reaction conditions that are easily managed in large-scale reactors without requiring exotic high-pressure or cryogenic equipment. The generation of benign byproducts such as water and salt facilitates easier wastewater treatment and compliance with increasingly stringent environmental regulations. This alignment with green chemistry principles not only reduces the environmental impact but also lowers the costs associated with effluent treatment and regulatory reporting, positioning the manufacturer as a responsible and sustainable partner in the global agrochemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical characteristics of the 5-alkoxy[1,2,4]triazolo[1,5-c]pyrimidine derivatives. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: What are the primary advantages of the disulfide intermediate route over conventional benzylthio methods?
A: The disulfide intermediate route eliminates the need for complex benzylthio protection and deprotection steps, significantly reducing waste generation and improving the overall purity of the final chlorosulfonyl product by avoiding side reactions associated with alkylthio groups.
Q: How does the new oxidation method impact the stability of the thione compounds?
A: While the free thione compounds can be unstable during storage, converting them immediately into the 2,2'-dithio-bis intermediates provides a stable, crystalline solid form that is easier to handle, store, and transport for subsequent chloroxidation reactions.
Q: Is this process scalable for industrial production of sulfonylurea herbicides?
A: Yes, the process utilizes common industrial reagents like hydrogen peroxide and chlorine gas, and the reaction conditions (temperature and pressure) are manageable, making it highly suitable for commercial scale-up from pilot plants to multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Alkoxy Triazolopyrimidine Thione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation herbicides. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 5-alkoxy[1,2,4]triazolo[1,5-c]pyrimidine thione and its derivatives performs consistently in your final formulations.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific agrochemical projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and drive down your overall production costs.
