Optimizing Febuxostat Synthesis: A Technical Breakthrough in Intermediate Purification and Scalability
Introduction to Advanced Febuxostat Synthesis
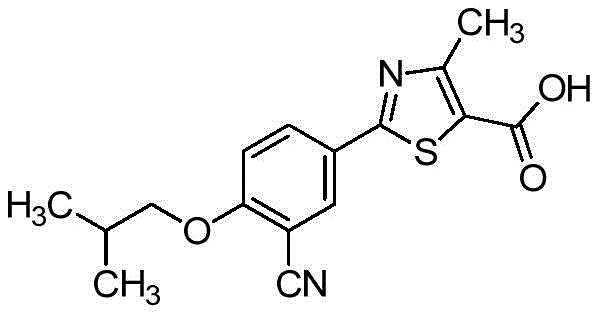
The pharmaceutical landscape for gout treatment has been significantly shaped by the development of non-purine xanthine oxidase inhibitors, with Febuxostat standing out as a premier therapeutic agent. As detailed in patent CN109912531B, recent advancements have focused not merely on the synthesis of the active pharmaceutical ingredient (API) itself, but critically on the optimization of its key intermediates. The structural integrity and purity of these intermediates directly dictate the safety profile and efficacy of the final drug substance. This technical insight report analyzes a breakthrough preparation method that addresses long-standing challenges in yield and impurity control, offering a robust pathway for commercial-scale production.
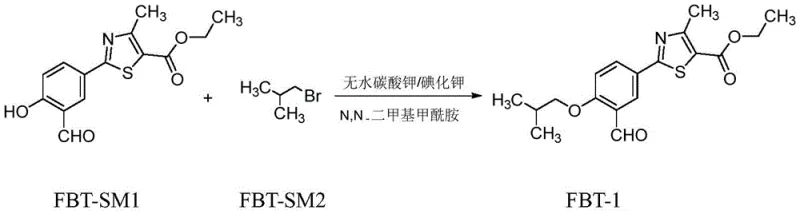
The core innovation lies in the post-treatment processing of the etherification reaction product, specifically the intermediate known as FBT-1 (2-(3-formyl-4-isobutoxy-phenyl)-4-methyl-thiazole-5-carboxylic acid ethyl ester). Traditional synthetic routes often suffer from low conversion rates in the initial steps and the persistence of raw material impurities that are notoriously difficult to separate. By implementing a rigorous three-stage purification protocol involving controlled crystallization, pH-adjusted pulping, and specialized solvent recrystallization, this new method achieves a purity of 99.80% and a total yield exceeding 90%. For R&D directors and supply chain managers, this represents a pivotal shift towards more sustainable and cost-efficient manufacturing paradigms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Febuxostat intermediates have historically been plagued by inefficiencies that compromise both economic viability and environmental compliance. For instance, earlier patents such as CN103058950A disclosed refining processes utilizing DMF, which resulted in a meager refined product yield of only 57%. Furthermore, subsequent steps required additional refinement with ethanol, generating substantial volumes of hazardous waste liquid. Other approaches, like those in CN104529935, relied on complex multi-solvent washing sequences involving DMF, water, and methanol. While these methods attempted to address purity, they invariably increased the environmental burden through excessive waste liquid generation and failed to consistently deliver high yields. The fundamental issue across these legacy routes is the inability to effectively separate unreacted starting materials (FBT-SM1) from the desired intermediate (FBT-1) without sacrificing significant amounts of product to the mother liquor.
The Novel Approach
The methodology presented in CN109912531B introduces a paradigm shift by optimizing the post-reaction workup rather than altering the core reaction chemistry itself. The process begins with the substitution reaction between FBT-SM1 and isobutyl bromide, followed immediately by a strategic addition of purified water to induce primary crystallization. Unlike the harsh solvent washes of the past, this method employs a gentle pulping step with purified water where the pH is meticulously adjusted to a range of 6.5 to 8. This specific pH window is critical for solubilizing and removing inorganic salts such as potassium bromide and potassium bicarbonate without dissolving the organic product. The final and most crucial step involves recrystallization using a composite solvent system of ethanol and formic acid. This specific solvent combination, utilized at a mass ratio of 1.2 to 2.0, provides the ideal solubility gradient to exclude trace organic impurities while maximizing crystal recovery.
Mechanistic Insights into Etherification and Recrystallization Dynamics
The success of this synthesis route hinges on the precise thermodynamic and kinetic control exercised during the purification phases. The initial etherification reaction, typically conducted at temperatures between 75°C and 85°C for 5 to 8 hours, generates a crude mixture containing the target ester, unreacted phenol, and inorganic salts. The novelty of the patented process is the recognition that simple filtration is insufficient for high-purity requirements. By cooling the reaction mass to 20-30°C and introducing water, the solubility of the organic product is drastically reduced, forcing precipitation. However, the true mechanistic advantage is realized during the pulping stage. At a neutral to slightly alkaline pH (6.5-8), the carboxylate and phenolic impurities remain soluble or are converted to water-soluble salts, whereas the target ester remains stable and insoluble. This selective solubility acts as a preliminary filter, removing the bulk of inorganic contaminants before the final recrystallization.
The recrystallization mechanism using the ethanol-formic acid system is equally sophisticated. Formic acid acts not just as a solvent component but potentially as a modifier that influences the crystal lattice formation of the thiazole derivative. The mass ratio of ethanol to formic acid (optimized at 1.4-1.5) ensures that the solvent power is sufficient to dissolve the crude solid at elevated temperatures (60-70°C) but drops sharply upon cooling to 15-30°C. This sharp solubility differential drives the formation of highly ordered crystals, effectively excluding structurally similar impurities like the starting material FBT-SM1 which might otherwise co-crystallize. Comparative data indicates that deviating from this specific solvent ratio or neglecting the pH control during pulping leads to a marked decrease in purity, confirming that the removal of impurities is a result of this specific synergistic solvent interaction rather than simple solubility differences.
How to Synthesize FBT-1 Efficiently
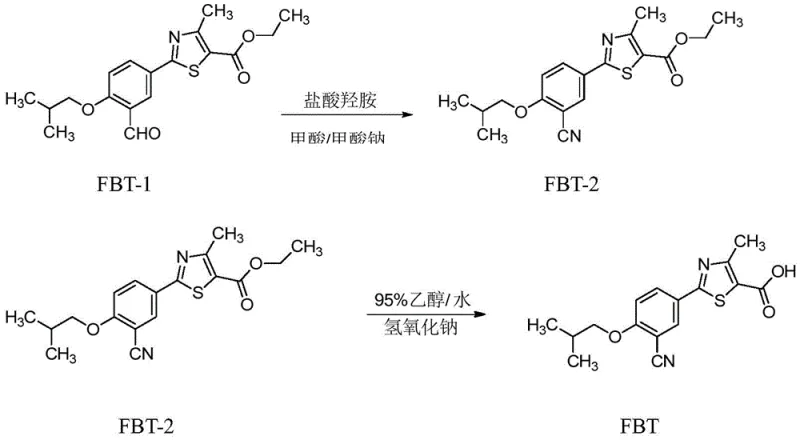
The synthesis of the critical intermediate FBT-1 requires strict adherence to the optimized post-treatment parameters to ensure the high purity necessary for downstream pharmaceutical applications. The process integrates standard etherification chemistry with a novel, multi-stage purification workflow that eliminates the need for hazardous solvent washes. Operators must pay particular attention to the temperature gradients during crystallization and the precise pH monitoring during the pulping phase, as these are the critical control points (CCPs) that determine the final quality of the intermediate. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring reproducibility and scalability for commercial manufacturing teams.
- Conduct the substitution reaction between FBT-SM1 and isobutyl bromide at 75-85°C, then cool to 20-30°C and add purified water to precipitate primary crystals.
- Pulp the primary crystals with purified water, strictly controlling the pH between 6.5 and 8 to remove inorganic salt impurities like potassium bromide.
- Recrystallize the secondary crystals using an ethanol-formic acid solution (mass ratio 1.2-2.0) to obtain the final high-purity FBT-1 intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this optimized synthesis route translates directly into tangible operational efficiencies and risk mitigation. The primary advantage lies in the dramatic improvement of process yield. By recovering product that would traditionally be lost to mother liquors or degraded during harsh washing steps, the overall throughput of the manufacturing line is significantly enhanced. This yield improvement reduces the quantity of raw materials required per kilogram of finished API, thereby lowering the direct material costs associated with production. Furthermore, the simplification of the purification steps—from complex multi-solvent washes to a streamlined water pulping and single recrystallization—reduces the consumption of expensive organic solvents like DMF, leading to substantial cost savings in solvent procurement and recovery.
- Cost Reduction in Manufacturing: The elimination of extensive DMF washing and the reduction of solvent usage in the recrystallization step drastically lowers the operational expenditure (OPEX) related to solvent management. Additionally, the high yield (>90%) means less raw material waste, directly impacting the cost of goods sold (COGS). The process avoids the need for multiple refining stages that characterize older methods, streamlining the production timeline and reducing labor and utility costs associated with extended processing times.
- Enhanced Supply Chain Reliability: The robustness of this purification method ensures a consistent supply of high-quality intermediates. Older methods often suffered from batch-to-batch variability due to the difficulty in separating starting materials, leading to potential production delays. By guaranteeing a purity of 99.80% with maximum single impurities below 0.07%, this method minimizes the risk of batch rejection and rework. This reliability allows for tighter inventory planning and reduces the need for safety stock buffers, optimizing working capital.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior. The reduction in DMF usage—a solvent with significant toxicity and regulatory scrutiny—simplifies waste treatment protocols and lowers the cost of hazardous waste disposal. The use of ethanol and formic acid, which are more environmentally benign and easier to recover, aligns with green chemistry principles. This facilitates easier regulatory approval and supports corporate sustainability goals, making the supply chain more resilient against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity Febuxostat intermediate synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology.
Q: How does the new purification method improve the yield of Febuxostat intermediates compared to prior art?
A: Unlike prior art methods such as CN103058950A which achieved only 57% yield after refinement, the optimized process in patent CN109912531B utilizes a specific ethanol-formic acid recrystallization system. This approach minimizes product loss in mother liquors and effectively removes difficult-to-separate raw materials, boosting total yields to over 90%.
Q: What is the critical role of pH control during the pulping stage?
A: Controlling the pH between 6.5 and 8 during the water pulping stage is essential for removing inorganic salt impurities, specifically potassium bicarbonate and potassium bromide generated during the reaction. Failure to control pH, as seen in comparative examples, results in lower purity intermediates despite potentially higher crude yields.
Q: Why is the ethanol-formic acid solvent system preferred over DMF or methanol washes?
A: Traditional washing methods using DMF and methanol generate significant hazardous waste and often fail to completely separate unreacted starting materials. The ethanol-formic acid composite solvent (ratio 1.2-2.0) offers superior solubility characteristics that allow for effective recrystallization, achieving purities up to 99.80% with maximum single impurities below 0.07%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Febuxostat Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high-purity standards demonstrated in patent CN109912531B can be reliably maintained at an industrial level. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch, guaranteeing that our Febuxostat intermediates meet the exacting demands of global pharmaceutical regulators.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to evaluate how our optimized manufacturing processes can drive efficiency and quality in your Febuxostat production programs.
