Advanced Chiral Amidophosphine Ligands: Enabling Scalable Asymmetric Hydrogenation for Global Pharma Supply Chains
Introduction to Novel Chiral Catalyst Technology
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for higher enantiomeric excess and more robust catalytic systems in the production of high-value fine chemicals. Patent CN1098856C introduces a significant advancement in this field through the disclosure of novel chiral amidophosphine trivalent phosphonate compounds. These molecules, existing in either (R) or (S) configurations, represent a sophisticated class of ligands designed to coordinate with Group VIII transition metals such as rhodium, iridium, ruthenium, and palladium. The structural innovation lies in the specific arrangement of phosphorus and nitrogen atoms around a chiral carbon center, creating a rigid yet tunable environment that dictates the stereochemical outcome of hydrogenation reactions. For R&D directors and process chemists, this technology offers a compelling alternative to conventional ligand systems, particularly for substrates that have historically shown poor selectivity. The ability to achieve high optical yields in the synthesis of complex pharmaceutical intermediates positions this technology as a critical asset for modern medicinal chemistry pipelines.
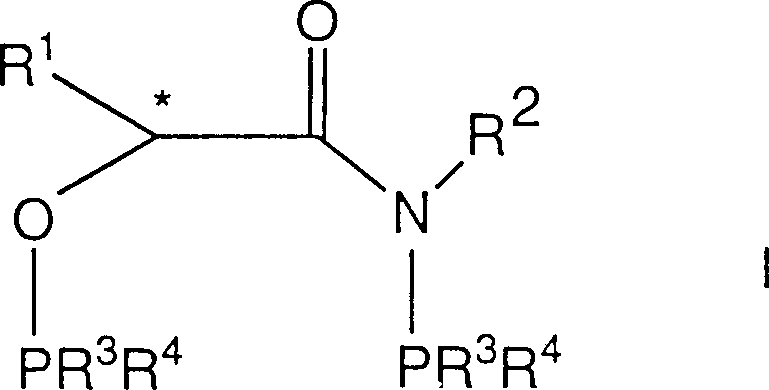
Beyond the theoretical novelty, the practical implications for supply chain stability are profound. The synthesis of these ligands relies on readily accessible starting materials, including substituted chlorophosphines and alpha-hydroxy amides, which can be sourced from established global chemical suppliers. This accessibility reduces the risk of raw material bottlenecks that often plague the production of exotic chiral catalysts. Furthermore, the versatility of the ligand scaffold allows for extensive derivatization; by modifying the R-groups on the phosphorus atoms, manufacturers can fine-tune the steric bulk and electronic properties to match specific substrate requirements. This adaptability ensures that the technology remains relevant across a wide spectrum of applications, from the production of cough suppressants like dextromethorphan to the synthesis of nutraceuticals such as zeaxanthin. As the industry moves towards more sustainable and efficient manufacturing processes, the adoption of such high-performance ligands becomes a strategic imperative for maintaining competitive advantage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional asymmetric hydrogenation often relies on well-known ligand families such as BINAP or DuPhos, which, while effective for many substrates, frequently encounter limitations when applied to sterically hindered or electronically deactivated olefins. In many cases, achieving the necessary enantiomeric excess requires excessive catalyst loading or extreme reaction conditions that are not economically viable on a commercial scale. Additionally, the stability of some conventional catalysts under process conditions can be problematic, leading to catalyst decomposition and the contamination of the final product with heavy metal residues. This necessitates costly and time-consuming purification steps, such as scavenging or recrystallization, which erode profit margins and extend lead times. For procurement managers, the reliance on single-source suppliers for these specialized ligands creates a vulnerability in the supply chain, where any disruption can halt production lines. The inability to easily modify the ligand structure to optimize performance for new drug candidates further restricts the flexibility of R&D teams during the process development phase.
The Novel Approach
The approach detailed in CN1098856C overcomes these hurdles by introducing a distinct amidophosphine trivalent phosphonate architecture that combines the soft donor properties of phosphorus with the hard donor characteristics of nitrogen and oxygen. This hybrid coordination sphere facilitates a more stable interaction with the metal center, reducing the likelihood of catalyst degradation during the reaction cycle. The patent data indicates that these ligands can operate effectively at moderate temperatures and pressures, significantly lowering the energy consumption associated with high-pressure hydrogenation processes. Moreover, the synthetic route to these ligands is straightforward and scalable, involving a nucleophilic substitution reaction that can be performed in common solvents like tetrahydrofuran or diethyl ether. This simplicity translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it eliminates the need for complex multi-step syntheses often required for other chiral ligands. By providing a platform technology that can be adapted through simple structural modifications, this novel approach empowers manufacturers to rapidly develop optimized processes for a diverse range of chiral building blocks.
Mechanistic Insights into Asymmetric Hydrogenation Catalysis
The efficacy of the amidophosphine trivalent phosphonate ligands stems from their ability to form well-defined, optically active complexes with transition metals. When coordinated with rhodium or iridium, the ligand creates a chiral pocket that effectively discriminates between the prochiral faces of the substrate molecule. The mechanism likely involves the formation of a cationic or neutral metal-dihydride species, where the substrate coordinates to the metal center prior to the migratory insertion step. The steric bulk provided by substituents such as tert-butyl or phenyl groups on the phosphorus atoms plays a crucial role in blocking one face of the substrate, thereby enforcing high enantioselectivity. Electronic effects are also significant; the electron-donating or withdrawing nature of the R-groups modulates the electron density at the metal center, influencing the rate of oxidative addition and reductive elimination steps. For technical teams, understanding these mechanistic nuances is essential for troubleshooting reaction issues and optimizing parameters such as solvent polarity and hydrogen pressure to maximize turnover numbers.
Impurity control is another critical aspect where this technology excels. The robust nature of the P-N-P ligand framework minimizes the formation of off-cycle species that often lead to racemic background reactions. In the examples provided, the catalysts demonstrate remarkable consistency in producing the desired enantiomer with minimal formation of the opposite isomer, even at high substrate-to-catalyst ratios. This high fidelity is vital for meeting the stringent purity specifications required by regulatory bodies for pharmaceutical ingredients. Furthermore, the stability of the catalyst complex reduces the leaching of metal into the product stream, simplifying downstream processing. From a quality assurance perspective, this means fewer batches are rejected due to out-of-specification metal content or optical purity, leading to a more predictable and reliable manufacturing output. The ability to maintain high selectivity across different substrate classes, from imines to enones, underscores the versatility of the underlying catalytic mechanism.
How to Synthesize Chiral Amidophosphine Ligands Efficiently
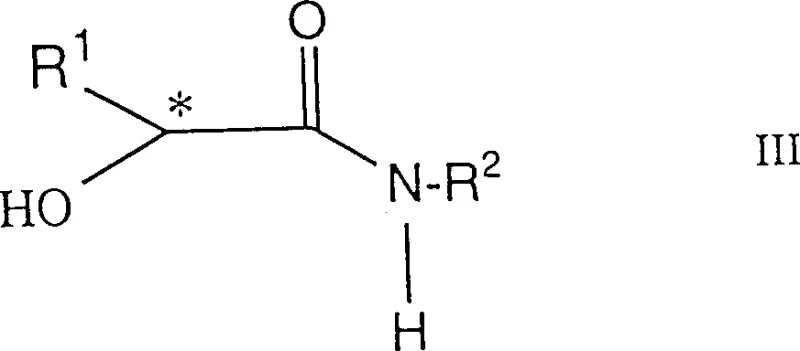
The preparation of these high-value ligands follows a precise protocol designed to preserve the integrity of the chiral center while ensuring complete conversion of the starting materials. The process begins with the activation of the hydroxyl group on the chiral amide precursor using a strong base under strictly anhydrous conditions. This step generates a reactive alkoxide intermediate that is highly susceptible to nucleophilic attack by the chlorophosphine reagent. Maintaining an inert atmosphere throughout the reaction is paramount to prevent oxidation of the phosphorus centers, which would render the ligand inactive. Following the coupling reaction, the crude product is typically isolated via filtration to remove salt byproducts and then purified through crystallization or chromatography to achieve the required optical purity. Detailed standardized synthesis steps for this process are provided in the guide below.
- Dissolve the chiral alpha-hydroxy amide precursor in anhydrous tetrahydrofuran under an inert argon atmosphere and add a secondary amine base such as diisopropylamine.
- Cool the reaction mixture to cryogenic temperatures (approximately -78°C) and slowly add an alkyllithium reagent like butyllithium to generate the reactive alkoxide species.
- Introduce the disubstituted chlorophosphine reagent dropwise while maintaining low temperature, then gradually warm to room temperature to complete the phosphorylation and isolate the crude ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain leaders, the adoption of this ligand technology offers tangible benefits that extend beyond mere technical performance. The primary advantage lies in the simplification of the sourcing strategy; because the ligands can be synthesized from commodity chemicals, there is less dependence on niche suppliers who may have limited capacity or long lead times. This decentralization of raw material sources enhances supply chain resilience, ensuring that production schedules are not disrupted by external market fluctuations. Additionally, the high efficiency of the catalysts means that lower quantities are required to achieve the same output, effectively reducing the inventory carrying costs associated with expensive catalytic materials. The robustness of the reaction conditions also allows for the use of standard reactor equipment, eliminating the need for capital investment in specialized high-pressure or cryogenic infrastructure. These factors collectively contribute to a more agile and cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The streamlined synthesis of the ligands themselves, combined with their high catalytic activity, drives down the overall cost of goods sold. By eliminating the need for expensive transition metal scavengers and reducing the number of purification steps, manufacturers can realize substantial cost savings. The ability to run reactions at lower catalyst loadings without sacrificing yield or selectivity further amplifies these economic benefits. Moreover, the reduced energy consumption associated with milder reaction conditions contributes to lower utility costs, enhancing the overall profitability of the production process. These efficiencies make the technology particularly attractive for large-scale commercial manufacturing where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: The use of widely available starting materials for ligand synthesis mitigates the risk of supply disruptions. Unlike proprietary ligands that are sole-sourced from a single vendor, the components for these amidophosphine compounds can be procured from multiple global suppliers. This redundancy ensures continuity of supply even in the event of geopolitical instability or logistical bottlenecks. Furthermore, the scalability of the synthesis process means that production volumes can be ramped up quickly to meet surging demand without the long lead times typically associated with complex chiral catalysts. This reliability is crucial for maintaining just-in-time inventory levels and meeting tight delivery deadlines for key customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The process described in the patent is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram pilot runs and eventually to ton-scale commercial production. The use of common solvents and the absence of hazardous reagents simplify waste management and disposal, aligning with increasingly stringent environmental regulations. The high atom economy of the reaction minimizes waste generation, supporting sustainability goals and reducing the environmental footprint of the manufacturing facility. This compliance with green chemistry principles not only avoids regulatory fines but also enhances the corporate reputation of the manufacturer as a responsible producer of fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this chemistry into their existing workflows. It clarifies the scope of application and the specific operational requirements needed to achieve optimal results.
Q: What represents the primary advantage of amidophosphine trivalent phosphonate ligands over traditional bis-phosphines?
A: These ligands offer a unique P-N-P coordination environment that provides superior steric tunability and electronic modulation compared to standard bis-phosphines, resulting in enhanced enantioselectivity for challenging substrates like isoquinolines and enones.
Q: Can these ligands be utilized for the synthesis of active pharmaceutical ingredients (APIs)?
A: Yes, the patent explicitly demonstrates their efficacy in catalyzing the asymmetric hydrogenation of precursors for dextromethorphan and levorphanol, which are critical intermediates in cough suppressant and analgesic drug manufacturing.
Q: What are the typical reaction conditions required for ligand synthesis?
A: The synthesis requires strict moisture and oxygen exclusion, typically utilizing anhydrous solvents like THF or diethyl ether, cryogenic cooling to -78°C, and strong bases such as butyllithium to ensure high optical purity and yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in the development of next-generation pharmaceuticals and fine chemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of chiral ligand meets the highest standards of quality and consistency. Our commitment to excellence extends beyond mere supply; we act as a strategic partner, offering technical support to optimize reaction conditions and troubleshoot any challenges that may arise during scale-up.
We invite you to explore the potential of these novel amidophosphine ligands for your specific applications. Whether you are looking to improve the enantioselectivity of an existing process or develop a new route for a complex intermediate, our experts are ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this technology can enhance your competitive position in the global market.
