Advanced Synthesis of N,N'-Dialkylalkanediamines for Industrial Pharmaceutical Applications
Advanced Synthesis of N,N'-Dialkylalkanediamines for Industrial Pharmaceutical Applications
The global demand for high-purity diamine intermediates continues to surge, driven by their critical role in the synthesis of complex pharmaceutical active ingredients and advanced agrochemical formulations. A pivotal advancement in this sector is detailed in patent CN1296472A, which outlines a novel preparation method for N,N'-dialkylalkanediamines that fundamentally addresses the long-standing challenges of by-product formation and purification complexity. This technology represents a significant leap forward for manufacturers seeking reliable N,N'-dialkylalkanediamines supplier partnerships, as it shifts the paradigm from simple stoichiometric mixing to precise kinetic control. By leveraging specific reaction engineering principles, this process inhibits the formation of hazardous cyclic and triamine impurities, thereby delivering a product stream that requires far less downstream processing. For R&D directors and procurement specialists alike, understanding the mechanistic underpinnings of this patent is essential for evaluating the feasibility of scaling these intermediates for commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of N,N'-dialkylalkanediamines has been plagued by inefficient reaction kinetics that favor the generation of structurally similar impurities. Traditional batch processes, where dihaloalkanes and alkylamines are mixed simultaneously or without strict concentration controls, often result in the accumulation of reactive mono-substituted intermediates. As described in the background art of the patent, these conventional methods typically achieve yields in the range of 80-85%, but they concurrently generate substantial quantities of cyclic dialkyldiamines (1-5%) and trialkylalkanetriamines (9-13%). These by-products are not merely minor contaminants; their physical properties often closely mirror the target diamine, making separation via standard distillation energy-intensive and technically demanding. Furthermore, older methodologies sometimes relied on transition metal catalysts like nickel or copper compounds, introducing the risk of heavy metal contamination that necessitates expensive removal steps to meet stringent pharmaceutical purity specifications. The economic burden of disposing of these chemical wastes and the capital expenditure required for complex purification trains have long hindered the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The breakthrough presented in the patent data lies in the realization that the reaction pathway can be steered away from side reactions by manipulating the concentration of the haloalkaneamine intermediate in real-time. Instead of allowing this intermediate to accumulate, the novel approach dictates that its residual amount in the reaction system must be maintained at 0.002 mol or less per 1 mol of alkylamine. This is achieved through sophisticated reactor designs, such as tubular continuous reactors or multi-stage container systems, where the dihaloalkane is added continuously or in controlled batches into a large excess of the alkylamine solvent. 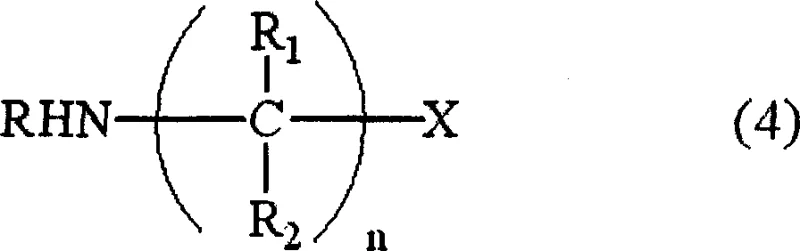 This strategic addition ensures that the probability of two intermediate molecules colliding (which forms cyclic by-products) or an intermediate colliding with the final product (which forms triamines) is statistically minimized. The result is a dramatic improvement in selectivity, with experimental data showing yields climbing to over 90%, effectively transforming the economic model of diamine production by simplifying the purification workflow and maximizing raw material utilization.
This strategic addition ensures that the probability of two intermediate molecules colliding (which forms cyclic by-products) or an intermediate colliding with the final product (which forms triamines) is statistically minimized. The result is a dramatic improvement in selectivity, with experimental data showing yields climbing to over 90%, effectively transforming the economic model of diamine production by simplifying the purification workflow and maximizing raw material utilization.
Mechanistic Insights into Kinetic Control of Alkylation
To fully appreciate the technical superiority of this method, one must delve into the competitive reaction mechanisms governing the alkylation of amines. The primary reaction involves the nucleophilic attack of the alkylamine on the dihaloalkane to form the desired diamine, proceeding through a mono-haloalkylamine intermediate. However, parallel side reactions compete for this intermediate. If the concentration of the mono-haloalkylamine becomes too high, it acts as an electrophile towards another molecule of itself, leading to cyclization, or towards the already formed diamine product, leading to oligomerization (triamines). The patent elucidates that the rate of the desired second alkylation step (intermediate to diamine) is surprisingly slower than the initial formation of the intermediate. 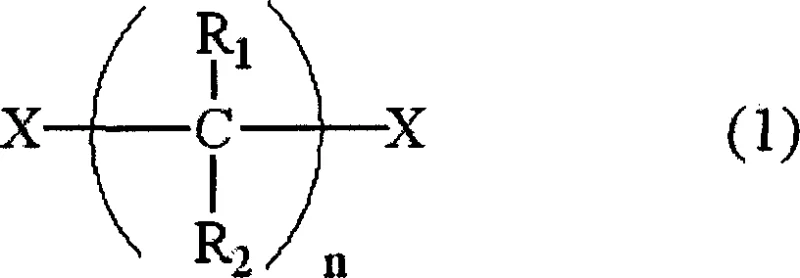 Therefore, without intervention, the intermediate accumulates. By enforcing a strict upper limit on the intermediate concentration—specifically maintaining the ratio [HA]m/[AM]m at ≤ 0.002—the process forces the kinetics to favor the reaction between the intermediate and the vast excess of free alkylamine. This kinetic suppression of bimolecular side reactions involving the intermediate is the core innovation, ensuring that the reaction trajectory remains locked on the formation of the linear diamine rather than diverging into cyclic or branched impurities.
Therefore, without intervention, the intermediate accumulates. By enforcing a strict upper limit on the intermediate concentration—specifically maintaining the ratio [HA]m/[AM]m at ≤ 0.002—the process forces the kinetics to favor the reaction between the intermediate and the vast excess of free alkylamine. This kinetic suppression of bimolecular side reactions involving the intermediate is the core innovation, ensuring that the reaction trajectory remains locked on the formation of the linear diamine rather than diverging into cyclic or branched impurities.
Furthermore, the control of impurity profiles is intrinsically linked to the reactor hydrodynamics and thermal management. The patent specifies a reaction temperature window of 50-250°C, with a preferred range of 80-140°C. Operating within this optimized thermal window is crucial because temperature influences the relative rate constants of the competing pathways. Too low a temperature might stall the reaction, while too high a temperature could accelerate the undesired cyclization pathways despite concentration controls. Additionally, the use of continuous flow technology enhances heat transfer efficiency, preventing local hot spots that could degrade selectivity. This level of mechanistic understanding allows process chemists to design robust manufacturing protocols that consistently deliver high-purity N,N'-dialkylalkanediamines, mitigating the risks associated with batch-to-batch variability often seen in legacy processes.
How to Synthesize N,N'-Dialkylalkanediamines Efficiently
Implementing this synthesis route requires a shift from traditional batch thinking to continuous processing logic. The core principle involves establishing a reaction environment where the dihaloalkane is the limiting reagent at any given point in the reaction zone, while the alkylamine serves as both the reactant and the solvent medium. Detailed operational parameters, such as feed rates, reactor dimensions, and specific injection port configurations, are critical to maintaining the requisite intermediate concentration thresholds. The following guide outlines the standardized approach derived from the patent examples to achieve optimal yields and purity profiles.
- Prepare a continuous flow reactor system, preferably a tubular reactor, and charge it with a large excess of lower alkylamine acting as both reactant and solvent.
- Introduce the dihaloalkane reactant continuously or in multiple batches at specific injection points to maintain the haloalkaneamine intermediate concentration below 0.002 mol per mole of amine.
- Maintain reaction temperature between 80-140°C and monitor the effluent via gas chromatography to ensure minimal formation of cyclic or triamine by-products before final distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method translates directly into tangible operational efficiencies and risk mitigation. The primary advantage lies in the drastic simplification of the downstream purification process. By suppressing the formation of trialkylalkanetriamines and cyclic diamines at the source, the crude product stream is significantly cleaner, reducing the load on distillation columns and minimizing the volume of heavy ends waste. This reduction in processing complexity inherently lowers the energy consumption per kilogram of finished product, contributing to substantial cost savings in fine chemical manufacturing without compromising on quality standards. Moreover, the elimination of transition metal catalysts, which were common in older methods, removes the need for expensive metal scavenging steps and the associated regulatory testing for heavy metal residues, further streamlining the path to market for pharmaceutical grade intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the significant increase in yield and the reduction of waste disposal costs. Conventional methods often lose nearly 20% of potential product mass to irrecoverable by-products, whereas this controlled method pushes yields above 90%. This improvement in atom economy means that less raw material is required to produce the same amount of final product, directly lowering the variable cost of goods sold. Additionally, the simplified purification train reduces the capital intensity of the plant, as fewer distillation stages are needed to achieve the same purity specification, allowing for a more lean and agile manufacturing footprint.
- Enhanced Supply Chain Reliability: The shift towards continuous flow reactors, such as the tubular reactors recommended in the patent, offers superior scalability compared to batch processing. Continuous systems can run for extended periods with consistent output, reducing the downtime associated with charging, heating, cooling, and cleaning batch vessels. This operational continuity ensures a more stable supply of high-purity N,N'-dialkylalkanediamines, mitigating the risk of stockouts for downstream clients. The robustness of the kinetic control also means the process is less sensitive to minor fluctuations in raw material quality, enhancing the overall reliability of the supply chain against upstream variability.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a greener profile. The reduction in by-product formation means less chemical waste requiring treatment or incineration, aligning with increasingly strict global environmental regulations. The ability to scale this process using modular continuous reactors allows manufacturers to incrementally increase capacity to meet market demand without the massive capital outlay required for new batch plants. This scalability ensures that the supply of these critical intermediates can grow in tandem with the pharmaceutical and agrochemical sectors, securing long-term supply continuity for strategic partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and benefits of adopting this technology for industrial production. Understanding these details is vital for stakeholders evaluating the integration of this process into their existing manufacturing portfolios.
Q: How does this novel process reduce trialkylalkanetriamine impurities?
A: By strictly controlling the residual concentration of the haloalkaneamine intermediate to below 0.002 mol relative to the alkylamine, the probability of the intermediate reacting with the formed product (which creates triamines) is kinetically suppressed.
Q: What reactor types are suitable for this synthesis?
A: The patent specifies that tubular continuous reactors or multi-stage container-type continuous reactors are optimal, as they allow for precise control over reagent addition rates and intermediate concentrations compared to traditional batch reactors.
Q: What yields can be expected compared to conventional methods?
A: While conventional methods typically achieve 80-85% yields with significant impurities, this controlled process demonstrates yields exceeding 90%, with specific examples reaching up to 93% purity for N,N'-dimethyl-1,2-ethylenediamine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-Dialkylalkanediamines Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the kinetic advantages demonstrated in patent CN1296472A can be fully realized on an industrial scale. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of N,N'-dialkylalkanediamines meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to process excellence means we can deliver the high-purity intermediates your R&D teams need to accelerate drug development timelines.
We invite you to collaborate with us to leverage these advanced synthesis capabilities for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can reduce your overall cost of goods. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your supply chain objectives and quality mandates.
