Revolutionizing X-Ray Contrast Agent Intermediates: A Deep Dive into Safe, Scalable Acid Chloride Synthesis
Introduction to Patent CN1154689A and Process Innovation
The pharmaceutical and diagnostic imaging industries rely heavily on the consistent supply of high-purity iodinated intermediates, specifically 5-amino-2,4,6-triiodo-1,3-phthaloyl chloride, which serves as a critical building block for non-ionic X-ray contrast agents. Patent CN1154689A introduces a transformative methodology that addresses long-standing safety and efficiency bottlenecks in the chlorination of 5-amino-2,4,6-triiodo-1,3-benzenedicarboxylic acid. Unlike traditional approaches that either risked thermal runaway by operating solvent-free or suffered from poor yields using ethyl acetate, this invention utilizes a heterogeneous reaction system mediated by catalytic tertiary amines in specific hydrocarbon or ether solvents. This technical breakthrough not only enhances operational safety by moderating reaction exotherms but also significantly improves material throughput, making it a cornerstone technology for any reliable pharmaceutical intermediate supplier aiming to optimize their production lines for complex iodinated compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this diacid chloride faced a dichotomy of risks and inefficiencies that plagued industrial scale-up efforts. Early literature described solvent-free conditions where solid starting materials were added directly to hot thionyl chloride, a practice that created severe industrial safety concerns due to the difficulty in controlling the addition rate and the potential for violent, unpredictable exothermic spikes. Alternatively, processes utilizing ethyl acetate as a solvent, while safer, resulted in dramatically reduced yields, often plummeting to the 50-60% range, which is economically unsustainable for high-value contrast agent precursors. These conventional methods forced manufacturers to choose between unacceptable safety hazards or prohibitive material costs, creating a significant barrier to entry for producing cost reduction in contrast agent manufacturing at a commercial scale.
The Novel Approach
The novel approach detailed in the patent data circumvents these issues by employing a carefully selected solvent system comprising C7-C16 linear or branched hydrocarbons, aromatic hydrocarbons like toluene or xylene, or specific ethers such as diglyme, all in the presence of a catalytic tertiary amine. This heterogeneous reaction environment allows for precise temperature control, typically maintained between 65-85°C, preventing thermal runaway while ensuring complete conversion of the carboxylic acid groups to acid chlorides. By shifting the reaction medium to these inert, high-boiling solvents, the process achieves yields consistently above 90%, eliminating the need for complex purification steps like recrystallization and thereby streamlining the workflow for the commercial scale-up of complex polymer additives and pharmaceutical intermediates alike.
Mechanistic Insights into Tertiary Amine-Catalyzed Chlorination
The core of this synthetic advancement lies in the mechanistic role of the tertiary amine catalyst, such as quinoline, N-methylmorpholine, or triethylamine, which activates the thionyl chloride for nucleophilic attack by the carboxylic acid oxygen. In this heterogeneous system, the solvent choice dictates the solubility profile of the reactants and intermediates; for instance, when using aliphatic hydrocarbons, the reaction proceeds through a distinct isolable intermediate, 5-sulfinylamino-2,4,6-triiodo-1,3-phthaloyl chloride (Formula II), which precipitates out of the solution. This precipitation drives the equilibrium forward and protects the sensitive amino group from side reactions, a critical factor in maintaining the high purity specifications required for injectable diagnostic agents. The ability to isolate this intermediate provides a unique quality control checkpoint, ensuring that any unreacted starting material or byproducts are removed before the final chlorination step is completed.
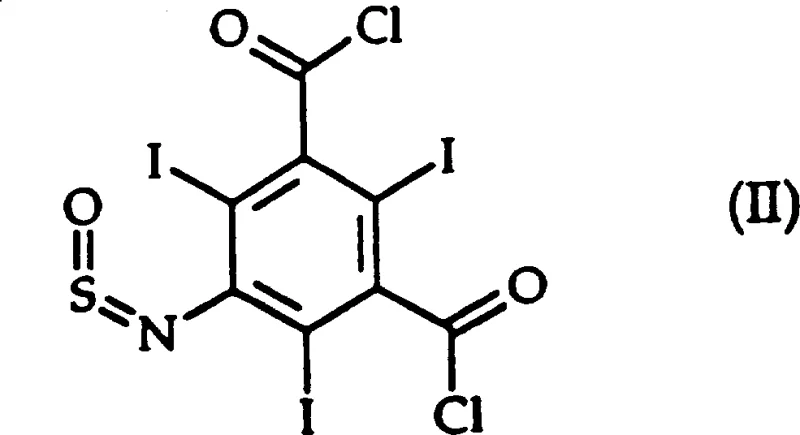
Furthermore, the mechanism involves the formation of an acyl ammonium salt species which facilitates the departure of the chloride ion, effectively lowering the activation energy for the chlorination of the sterically hindered triiodo-substituted benzene ring. The use of solvents like m-xylene or n-dodecane ensures that while the intermediate may precipitate, the final diacid chloride product remains manageable or can be easily processed upon the addition of diglyme and water in the workup phase. This nuanced understanding of solubility and reactivity allows for reducing lead time for high-purity pharmaceutical intermediates by minimizing downstream processing time, as the crude product often requires only simple filtration and washing to meet stringent analytical standards without the need for energy-intensive distillation or chromatography.
How to Synthesize 5-Amino-2,4,6-Triiodo-1,3-Phthaloyl Chloride Efficiently
To implement this robust synthesis protocol effectively, operators must adhere to strict temperature controls and reagent addition rates to maintain the heterogeneous balance of the reaction mixture. The process begins with the suspension of the triiodo-benzenedicarboxylic acid in the chosen solvent, followed by the addition of the amine catalyst and the gradual introduction of thionyl chloride to manage gas evolution. Detailed standard operating procedures regarding the specific stoichiometry of the amine catalyst and the precise temperature ramping profiles are essential for replicating the high yields reported in the patent examples. For a comprehensive guide on the exact experimental parameters, including specific solvent volumes and drying conditions, please refer to the standardized synthesis steps outlined below.
- Suspend 5-amino-2,4,6-triiodo-1,3-benzenedicarboxylic acid in a selected solvent such as m-xylene or n-dodecane.
- Add a catalytic amount of a tertiary amine (e.g., quinoline or N-methylmorpholine) and heat the mixture to 65-70°C.
- Gradually add thionyl chloride while maintaining temperature, then heat to 80-85°C to complete the reaction and isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this solvent-mediated chlorination process offers substantial benefits that extend far beyond simple yield improvements, fundamentally altering the cost structure of producing iodinated intermediates. By eliminating the safety risks associated with solvent-free operations, facilities can reduce insurance premiums and operational downtime related to safety incidents, while the higher yields directly translate to better raw material utilization and reduced waste disposal costs. The ability to use common, inexpensive solvents like kerosene, ligroin, or xylenes instead of specialized or hazardous reagents further drives down the variable costs of production, making the supply chain more resilient to market fluctuations in specialty chemical pricing.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps such as recrystallization significantly lowers the operational expenditure per kilogram of finished product. Since the process yields high-purity material directly from the reaction mixture through simple filtration, the consumption of additional solvents for purification and the energy required for heating and cooling cycles during crystallization are drastically reduced. This streamlined workflow means that manufacturing capacity is freed up, allowing for higher throughput without the need for capital investment in new equipment, thereby achieving significant cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: Utilizing widely available commodity solvents like n-octane, n-decane, or toluene ensures that production is not bottlenecked by the scarcity of exotic reagents. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without compromising product quality, reduces the risk of batch failures that could disrupt supply continuity. This reliability is crucial for maintaining long-term contracts with downstream contrast agent manufacturers who require guaranteed delivery schedules and consistent quality attributes for their regulatory filings.
- Scalability and Environmental Compliance: The heterogeneous nature of the reaction simplifies scale-up from pilot plant to multi-ton commercial production, as heat transfer and mixing issues are mitigated by the solvent volume. Furthermore, the process generates less hazardous waste compared to solvent-free methods where quenching exotherms is difficult, and the use of recoverable solvents aligns with modern green chemistry principles. This environmental compatibility facilitates easier permitting and compliance with increasingly strict global regulations on volatile organic compound emissions and chemical waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this chlorination process, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is vital for process engineers and quality assurance teams looking to integrate this methodology into their existing manufacturing suites. The answers provided reflect the specific conditions and outcomes observed in the patent examples, offering a realistic expectation of performance metrics.
Q: Why is the solvent-free method for this acid chloride considered unsafe?
A: Running the reaction without solvent leads to severe exotherms and unpredictable short-term reactions when mixing solid acid with hot thionyl chloride, posing significant industrial safety risks.
Q: What is the advantage of using aliphatic hydrocarbons over ethyl acetate?
A: Ethyl acetate processes historically suffer from dramatic yield drops (50-60%), whereas aliphatic hydrocarbons allow for yields exceeding 90% and enable the isolation of a stable intermediate.
Q: Can the intermediate 5-sulfinylamino derivative be isolated?
A: Yes, when using C7-C16 linear or branched aliphatic hydrocarbons, the intermediate (Formula II) precipitates and can be filtered before final conversion to the diacid chloride.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Amino-2,4,6-Triiodo-1,3-Phthaloyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced synthetic methodologies like the one described in CN1154689A to deliver superior quality intermediates for the global diagnostic imaging market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every batch meets stringent purity specifications through our rigorous QC labs equipped with state-of-the-art HPLC and spectroscopic analysis tools. We understand that the consistency of iodinated intermediates is paramount for the safety and efficacy of the final contrast agents, and our commitment to process excellence guarantees a supply partner you can trust for your most critical projects.
We invite you to contact our technical procurement team to discuss how we can tailor our production capabilities to your specific volume requirements and quality standards. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis routes can lower your total cost of ownership. We are ready to provide specific COA data and route feasibility assessments to support your R&D and supply chain planning, ensuring a seamless integration of our high-purity intermediates into your manufacturing workflow.
