Scalable Synthesis of 4-Isothiocyanatopyrazolones for Advanced Drug Discovery
Scalable Synthesis of 4-Isothiocyanatopyrazolones for Advanced Drug Discovery
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic scaffolds that serve as privileged structures in medicinal chemistry. Patent CN107382867B introduces a significant advancement in this domain by disclosing a novel class of 4-isothiocyanatopyrazolones, characterized by the general Formula I. These compounds are not merely academic curiosities but serve as critical building blocks for constructing multi-pharmacophore systems, specifically pyrazolone spiro[ethylenethiourea]spirooxindoles. The innovation lies in a streamlined three-step synthetic protocol that transforms simple 4-unsubstituted pyrazolones into highly functionalized isothiocyanate derivatives. This methodology addresses the longstanding challenge of introducing reactive handles at the 4-position of the pyrazolone ring without compromising the integrity of the sensitive lactam moiety. For R&D directors and process chemists, this represents a reliable pathway to access diverse chemical space for lead optimization programs targeting enzymes such as human carboxylesterase 1 (hCE1).
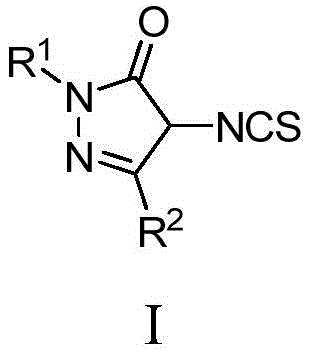
The strategic value of this technology extends beyond the immediate compounds. By establishing a foundation for spiro-cyclization reactions, this patent enables the rapid assembly of complex molecular architectures that combine the biological activities of pyrazolones, ethylene thioureas, and oxindoles. Such multi-target ligands are increasingly relevant in treating complex diseases where single-target therapies often fail. The versatility of the isothiocyanate group allows for further derivatization, making these intermediates indispensable for modern drug discovery pipelines focused on neuroprotective agents and antipsychotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of pyrazolone rings, particularly at the 4-position, has been fraught with synthetic difficulties. Conventional methods often rely on harsh electrophilic substitutions that suffer from poor regioselectivity, leading to mixtures of 3-substituted and 4-substituted isomers that are difficult to separate. Furthermore, existing routes to introduce nitrogen-containing functionalities frequently require multiple protection and deprotection steps, significantly increasing the step count and reducing overall atom economy. The use of heavy metal catalysts in some traditional cross-coupling approaches also introduces concerns regarding residual metal contamination, which is a critical quality attribute for pharmaceutical intermediates. These limitations result in higher production costs, longer lead times, and environmental burdens associated with waste disposal, making conventional processes less attractive for large-scale manufacturing of complex spiro-oxindole precursors.
The Novel Approach
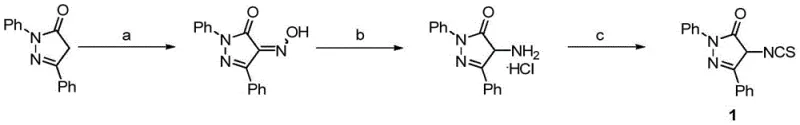
In stark contrast, the methodology disclosed in CN107382867B offers a remarkably efficient solution. The process initiates with the oximation of 4-unsubstituted pyrazolones using sodium nitrite and hydrochloric acid in ethanol at low temperatures (0°C), a reaction that proceeds with high conversion to yield 4-oximino intermediates. This is followed by a catalytic hydrogenation step using palladium on carbon (Pd/C) to reduce the oxime to the corresponding amine, which is isolated as a stable hydrochloride salt. The final transformation involves reacting the amine with carbon disulfide and triethylamine, followed by activation with Boc anhydride to generate the target 4-isothiocyanatopyrazolone. This three-step sequence is operationally simple, avoids chromatographic purification in the early stages, and utilizes inexpensive, commercially available reagents. The ability to perform these reactions under mild conditions demonstrates a clear advantage in terms of safety and scalability for industrial applications.
Mechanistic Insights into Organocatalytic Asymmetric Spiro-Cyclization
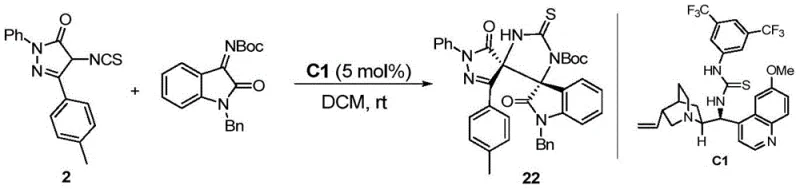
Beyond the synthesis of the linear precursors, the patent details a sophisticated organocatalytic strategy for converting these 4-isothiocyanatopyrazolones into chiral spiro-compounds. The core of this transformation is a [3+2] cycloaddition reaction between the 4-isothiocyanatopyrazolone and an isatin-derived N-Boc imine. This reaction is mediated by a chiral quinine-derived thiourea catalyst (such as Catalyst C1), which plays a dual role in activating both the nucleophile and the electrophile through hydrogen bonding interactions. The thiourea moiety activates the imine electrophile, while the basic quinoline nitrogen likely deprotonates the acidic methine proton of the pyrazolone, generating a reactive enolate species. This bifunctional activation ensures precise spatial arrangement of the reactants within the chiral pocket of the catalyst, leading to excellent stereocontrol.
The mechanistic elegance of this approach lies in its ability to construct two contiguous stereocenters and a spiro-center in a single operation with high enantiomeric excess (ee values up to 96% are reported). For R&D teams, understanding this mechanism is crucial for optimizing reaction parameters such as solvent polarity and temperature to maximize stereoselectivity. Furthermore, the absence of transition metals in this key bond-forming step simplifies the downstream purification process, eliminating the need for expensive scavengers to remove metal residues. This purity profile is essential for meeting the stringent regulatory requirements of pharmaceutical manufacturing, ensuring that the final active pharmaceutical ingredients (APIs) are free from genotoxic metal impurities.
How to Synthesize 4-Isothiocyanatopyrazolones Efficiently
The preparation of these valuable intermediates follows a logical progression designed to maximize yield and minimize waste. The process begins with the careful control of temperature during the diazotization-like oximation step to prevent side reactions. Subsequent reduction requires standard hydrogenation equipment but benefits from the stability of the intermediate amine salt, which can be stored or transported if necessary. The final thiocarbonylation step is rapid and exothermic, requiring controlled addition of reagents to maintain safety. Detailed standardized operating procedures for each stage, including specific molar ratios and workup protocols, are essential for reproducible results in a GMP environment.
- React 4-unsubstituted pyrazolone with sodium nitrite and hydrochloric acid in ethanol at 0°C to form the 4-oximino intermediate.
- Reduce the oxime using Pd/C and hydrogen in methanol, followed by treatment with HCl to obtain the amine hydrochloride salt.
- Treat the amine salt with carbon disulfide and triethylamine, followed by Boc anhydride activation to yield the final 4-isothiocyanatopyrazolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost structure and supply reliability. The reliance on commodity chemicals such as sodium nitrite, ethanol, and carbon disulfide means that raw material costs are inherently low and subject to minimal market volatility compared to specialized organometallic reagents. The simplicity of the three-step sequence reduces the requirement for complex reactor configurations, allowing production to be scaled up in standard stainless steel vessels without the need for exotic materials of construction. This flexibility enhances supply chain resilience, as the manufacturing process can be easily transferred between different facilities or contract manufacturing organizations (CMOs) without significant re-engineering.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in purification steps directly translate to lower manufacturing costs. By avoiding chromatography for intermediate isolation and utilizing simple filtration and crystallization techniques, the process significantly reduces solvent consumption and labor hours. This efficiency gain allows for substantial cost savings in the production of high-purity pharmaceutical intermediates, making the final drug candidates more economically viable to develop and commercialize.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent quality and yield, minimizing the risk of batch failures that can disrupt supply chains. The use of stable intermediates allows for the strategic stocking of key precursors, buffering against potential disruptions in the supply of starting materials. Furthermore, the short reaction times and mild conditions contribute to faster cycle times, enabling manufacturers to respond more quickly to fluctuating demand from downstream drug development partners.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reagents and conditions that are compatible with large-scale industrial operations. The avoidance of heavy metals aligns with green chemistry principles and simplifies waste treatment, reducing the environmental footprint of the manufacturing process. This compliance with environmental regulations mitigates the risk of regulatory penalties and supports the sustainability goals of modern pharmaceutical companies, enhancing the overall corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-isothiocyanatopyrazolones. These answers are derived from the detailed experimental data and structural analysis provided in the patent documentation, offering clarity on the feasibility and advantages of this technology for potential licensees and manufacturing partners.
Q: What is the primary advantage of the 3-step synthesis route described in CN107382867B?
A: The primary advantage is operational simplicity and high efficiency. Starting from readily available 4-unsubstituted pyrazolones, the route avoids harsh conditions and complex purification steps, utilizing standard reagents like sodium nitrite and carbon disulfide to achieve high yields suitable for industrial scaling.
Q: How does the organocatalytic approach improve the synthesis of spiro-oxindole derivatives?
A: The use of quinine-derived thiourea catalysts enables highly enantioselective [3+2] cycloadditions. This eliminates the need for expensive transition metal catalysts and difficult chiral separations, directly providing optically active scaffolds critical for biological activity against targets like hCE1.
Q: Are these intermediates stable for long-term storage and shipping?
A: Yes, the 4-isothiocyanatopyrazolones described exhibit good stability as solid compounds. The patent data indicates they can be isolated as white or pale yellow solids with defined melting points, facilitating safe transport and inventory management for downstream drug development projects.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Isothiocyanatopyrazolones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN107382867B for the development of next-generation therapeutics. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from laboratory discovery to global market supply. Our state-of-the-art facilities are equipped to handle the specific requirements of heterocyclic chemistry, including dedicated lines for hydrogenation and moisture-sensitive reactions, guaranteeing stringent purity specifications and rigorous QC labs to validate every batch against your exacting standards.
We invite you to collaborate with us to leverage this innovative synthetic route for your drug discovery programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and efficient supply chain for your critical pharmaceutical intermediates.
