Advanced Synthesis of Thermostable N-n-butylbenzene Sulphonamide for Industrial Polymer Applications
Advanced Synthesis of Thermostable N-n-butylbenzene Sulphonamide for Industrial Polymer Applications
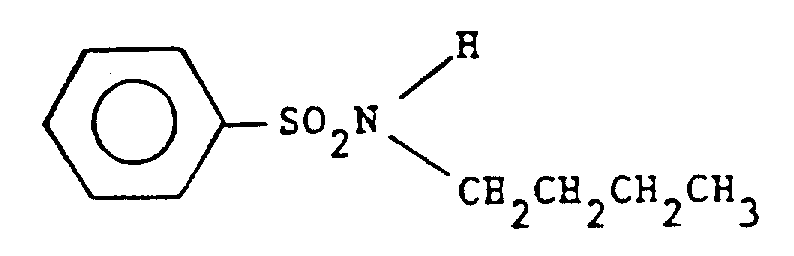
The chemical industry constantly seeks methods to enhance the thermal performance of polymer additives, particularly for high-performance engineering plastics. Patent CN1037894A, filed in late 1989, introduces a groundbreaking method for synthesizing arylsulfonylalkylamides, specifically targeting the production of N-n-butylbenzene sulphonamide (BBSA). This compound serves as a critical softening agent or plasticizer for polyamides, such as Nylon 11 and Nylon 12, which are widely used in automotive and industrial applications. The core innovation lies in a streamlined reaction protocol that contacts aryl sulfonyl halides with an excess of alkylamine and an aqueous alkaline reagent. By meticulously managing the phase separation and subsequent removal of volatiles, this process yields a product with exceptional thermostability, capable of withstanding processing temperatures between 200°C and 250°C without degradation. This technical advancement eliminates the generation of acidic byproducts that typically compromise polymer mechanical performance and cause discoloration, marking a significant leap forward in additive manufacturing technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of thermostable aryl sulfonyl alkylamides has been plagued by complex purification requirements and the risk of thermal degradation during polymer processing. Prior art, such as the method disclosed in European Patent Application EP 7623, necessitated a distinct purification step using alkaline reagents at elevated temperatures around 200°C to achieve the desired stability. This traditional approach not only increased energy consumption and operational complexity but also introduced potential points of failure where the product could degrade or form colored impurities. Furthermore, conventional acylation reactions often struggle with the complete removal of hydrohalic acid byproducts, which can catalyze the decomposition of the final plasticizer when exposed to the high shear and heat of extrusion processes. These limitations result in inconsistent product quality, where batches may vary in their ability to protect the polymer matrix, leading to premature failure of the final plastic components in demanding environments.
The Novel Approach
The methodology described in CN1037894A fundamentally reengineers the synthesis workflow to bypass these historical bottlenecks through a clever manipulation of reaction stoichiometry and phase behavior. Instead of relying on post-synthesis alkaline purification, the novel approach utilizes a large excess of alkylamine relative to the aryl sulfonyl halide during the initial reaction phase, coupled with an aqueous alkaline solution to neutralize the generated acid immediately. This creates a biphasic system where the desired aryl sulfonyl alkylamide partitions efficiently into the organic phase, while salts and water-soluble impurities remain in the aqueous layer. The subsequent steps involve simply dehydrating the organic phase and stripping off the excess amine, effectively purifying the product without harsh chemical treatments. This integrated synthesis-and-purification strategy ensures that the final residue is inherently thermostable, ready for direct use as a polyamide plasticizer without the need for the rigorous secondary treatments mandated by older patents.
Mechanistic Insights into Biphasic Acylation and Phase Separation
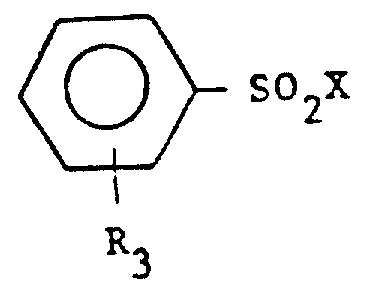
The chemical mechanism driving this synthesis is a variation of the Schotten-Baumann reaction, optimized for industrial scalability and impurity control. The process begins with the nucleophilic attack of the alkylamine nitrogen on the sulfur atom of the aryl sulfonyl halide. In this specific patent embodiment, the use of benzenesulfonyl chloride as the electrophile and n-butylamine as the nucleophile is preferred. The presence of an aqueous alkaline reagent, such as sodium hydroxide or potassium hydroxide, plays a dual role: it neutralizes the hydrogen chloride byproduct formed during the acylation, driving the equilibrium towards product formation, and it helps maintain the integrity of the amine by preventing its protonation in the aqueous phase. The reaction is exothermic, and careful temperature control between 0°C and 50°C is maintained to prevent side reactions, ensuring that the sulfonyl chloride reacts selectively with the amine rather than hydrolyzing with water.
Following the reaction, the system separates into two distinct phases, a critical feature that underpins the purity of the final product. The organic phase contains the target N-n-butylbenzene sulphonamide along with unreacted excess amine and trace water. The efficiency of this phase separation is paramount; it allows for the bulk removal of inorganic salts (such as sodium chloride) which would otherwise act as contaminants in the polymer matrix. The patent specifies that the organic phase obtained after sedimentation contains a high concentration of the desired amide, often exceeding 96% purity before final distillation. This high initial purity reduces the burden on downstream processing equipment and minimizes the risk of thermal degradation during the final isolation step, ensuring the product retains its colorless and odorless characteristics essential for high-grade plastic applications.
How to Synthesize N-n-butylbenzene Sulphonamide Efficiently
Implementing this synthesis route requires precise control over reagent ratios and distillation parameters to maximize yield and thermal stability. The process is designed to be flexible, allowing for both batch and continuous operations, which makes it highly adaptable to different manufacturing scales. The key to success lies in the initial contact of the aryl sulfonyl halide with the amine and base mixture, followed by a controlled heating period to complete the reaction without decomposing the sensitive sulfonamide bond. Once the reaction is complete, the focus shifts to the efficient removal of volatiles. The patent outlines a specific distillation protocol where water and excess n-butylamine are stripped off under vacuum at temperatures below 180°C, typically between 130°C and 170°C. This gentle removal prevents the onset of thermal degradation that could lead to discoloration. The detailed standardized synthetic steps for replicating this high-efficiency process are provided in the guide below.
- Contact aryl sulfonyl halide with excess alkylamine and aqueous alkaline reagent to form the crude amide in the organic phase.
- Separate the organic phase and remove water and excess alkylamine via distillation under reduced pressure.
- Isolate the final high-purity aryl sulfonyl alkylamide residue through vacuum distillation or thin-film evaporation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers profound strategic benefits beyond mere technical specification compliance. The elimination of complex purification steps found in prior art translates directly into a simplified manufacturing workflow, which significantly reduces capital expenditure on specialized purification equipment and lowers operational overhead. By avoiding the need for high-temperature alkaline treatment post-synthesis, manufacturers can reduce energy consumption and minimize the handling of hazardous hot caustic solutions, thereby enhancing workplace safety and reducing insurance liabilities. Furthermore, the reliance on commodity chemicals such as benzenesulfonyl chloride and n-butylamine ensures a robust and resilient supply chain, as these raw materials are widely available from multiple global suppliers, mitigating the risk of single-source bottlenecks that often plague specialty chemical production.
- Cost Reduction in Manufacturing: The streamlined process architecture drastically cuts production costs by integrating reaction and initial purification into a single phase-separation event. By removing the necessity for the expensive and energy-intensive purification steps required by older technologies like EP 7623, the overall cost of goods sold is substantially lowered. The ability to recover and recycle the excess alkylamine through distillation further enhances economic efficiency, turning a potential waste stream into a reusable asset. Additionally, the high selectivity of the reaction minimizes the formation of byproducts that would require costly disposal or treatment, leading to a leaner and more profitable manufacturing operation that can offer competitive pricing in the global polymer additives market.
- Enhanced Supply Chain Reliability: The use of standard, non-proprietary raw materials ensures that production schedules are not held hostage by the availability of exotic catalysts or reagents. Since the process operates effectively at near-atmospheric pressure and moderate temperatures, it can be executed in standard glass-lined or stainless steel reactors (such as 304L or 316L grades) found in most multipurpose chemical plants. This compatibility with existing infrastructure means that scale-up can be achieved rapidly without the need for bespoke engineering or long lead-time equipment fabrication. Consequently, suppliers can guarantee shorter lead times and more consistent delivery schedules, providing downstream polymer manufacturers with the certainty they need to plan their own production runs without fear of raw material shortages.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method offers a cleaner profile that aligns with modern green chemistry principles. The aqueous waste stream primarily consists of salt water and trace organics, which is far easier to treat than the complex sludge generated by multi-step purification processes. The closed-loop nature of the distillation steps minimizes volatile organic compound (VOC) emissions, helping facilities meet stringent environmental regulations. Moreover, the simplicity of the process makes it highly scalable; moving from pilot plant quantities to multi-tonne commercial production involves straightforward linear scaling of reactor volumes and distillation columns, ensuring that quality remains consistent regardless of batch size, which is crucial for maintaining long-term supply contracts with major automotive and industrial clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this arylsulfonylalkylamide synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific polymer formulations.
Q: Why is thermal stability critical for polyamide plasticizers?
A: Polyamides like PA11 and PA12 are processed at high temperatures (200-250°C). Conventional plasticizers may degrade, releasing acidic products that damage polymer chains and machinery. This patented method ensures the sulfonamide remains stable without generating color or acidic byproducts.
Q: How does this method improve upon previous purification techniques?
A: Unlike prior art such as EP 7623 which required complex alkaline purification steps, this process integrates purification into the workup by simply removing excess amine and water from the organic phase, drastically simplifying the workflow.
Q: What are the key raw materials for this synthesis?
A: The primary reactants are aryl sulfonyl halides (preferably benzenesulfonyl chloride) and alkylamines (specifically n-butylamine), reacted in the presence of a basic aqueous solution like sodium hydroxide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-n-butylbenzene Sulphonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance plasticizers play in the durability and functionality of modern engineering plastics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of N-n-butylbenzene sulphonamide meets the exacting thermal stability standards required for polyamide processing. Our commitment to quality assurance means that our clients receive a product that consistently performs under high-heat conditions, protecting their polymer matrices from degradation and ensuring the longevity of their final products.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. By leveraging our optimized synthesis protocols, we can help you reduce total landed costs while securing a stable supply of this essential additive. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your supply chain goals.
