Advanced Synthesis of High-Purity Fluoroalkylbenzene Derivatives for Commercial Scale-Up
The chemical industry's relentless pursuit of ultra-high purity intermediates has found a significant breakthrough in the methodologies described within patent CN1653029A, which outlines a sophisticated process for producing high-purity (fluoroalkyl)benzene derivatives. This technology addresses a longstanding challenge in fine chemical synthesis: the persistent contamination of final products with residual transition metals and halogens, which are unacceptable in sensitive applications like semiconductor manufacturing and active pharmaceutical ingredient (API) production. By implementing a rigorous pre-purification step on the starting alkylbenzene derivatives to reduce transition metal content to below 500ppm, followed by controlled photohalogenation and excess hydrofluoric acid treatment, manufacturers can achieve purity levels exceeding 99.9% with residual halogens reduced to less than 1wt%. This approach not only enhances the quality of the final electronic chemical or pharmaceutical intermediate but also streamlines the downstream purification burden, offering a robust pathway for reliable high-purity fluoroalkylbenzene supplier operations globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for introducing fluorine atoms into aromatic systems often rely on direct halogen-fluorine substitution using standard industrial alkylbenzene feedstocks without adequate pre-treatment. A critical flaw in these conventional processes is the presence of trace transition metals, such as iron, zinc, or tin, which are frequently introduced during the initial synthesis of the alkylbenzene precursors or through equipment corrosion. When these metal-contaminated substrates undergo halogenation, the metals act as Lewis acid catalysts that promote electrophilic aromatic substitution on the benzene ring itself, rather than the desired free-radical substitution on the alkyl side chain. This results in a complex mixture of nuclear-halogenated isomers that are structurally similar to the target molecule and extremely difficult to separate via standard distillation or crystallization. Furthermore, these metal impurities carry through the subsequent fluorination steps, ending up in the final product where they can cause catastrophic failures in electronic devices or toxicity issues in medicinal compounds, necessitating expensive and time-consuming post-synthesis scavenging procedures.
The Novel Approach
The innovative strategy disclosed in the patent fundamentally shifts the purification locus to the beginning of the synthetic route, ensuring that the starting alkylbenzene derivative contains less than 500ppm of Group 3 to 12 transition metals before any halogenation occurs. By enforcing this strict metal threshold, potentially lowering iron content to below 50ppm through adsorption or distillation, the process effectively suppresses the catalytic activity that leads to nuclear halogenation side reactions. This allows the subsequent photohalogenation step to proceed with high selectivity exclusively on the alkyl side chain, generating the necessary haloalkyl precursor with minimal structural impurities. When this purified intermediate is then subjected to halogen-fluorine substitution using a large excess of anhydrous hydrogen fluoride (at least 10 moles relative to the substrate), the reaction drives to completion with exceptional efficiency, yielding (fluoroalkyl)benzene derivatives with drastically reduced residual halogen and metal content, thereby eliminating the need for complex downstream metal removal technologies.
Mechanistic Insights into Photohalogenation and Halogen-Fluorine Substitution
The core of this synthesis relies on a precise understanding of radical chemistry and the inhibitory effects of transition metals on radical propagation. In the first stage, the purified alkylbenzene derivative, represented generally by Formula (1), is subjected to photochlorination using chlorine gas under irradiation from a mercury or tungsten lamp. 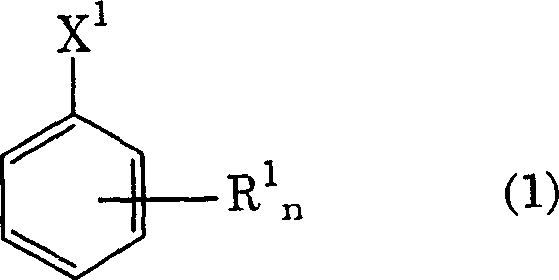 The presence of light initiates the homolytic cleavage of chlorine molecules into chlorine radicals, which abstract hydrogen atoms from the benzylic position of the alkyl side chain. Because the starting material has been rigorously stripped of transition metals, there are no competing Lewis acid pathways to activate the chlorine for electrophilic attack on the aromatic ring. This ensures that the radical chain reaction propagates efficiently along the side chain, converting methyl or ethyl groups into trichloromethyl or pentachloroethyl groups without compromising the integrity of the benzene core, a selectivity that is paramount for maintaining the structural fidelity required in high-value functional chemicals.
The presence of light initiates the homolytic cleavage of chlorine molecules into chlorine radicals, which abstract hydrogen atoms from the benzylic position of the alkyl side chain. Because the starting material has been rigorously stripped of transition metals, there are no competing Lewis acid pathways to activate the chlorine for electrophilic attack on the aromatic ring. This ensures that the radical chain reaction propagates efficiently along the side chain, converting methyl or ethyl groups into trichloromethyl or pentachloroethyl groups without compromising the integrity of the benzene core, a selectivity that is paramount for maintaining the structural fidelity required in high-value functional chemicals.
Following the successful generation of the haloalkyl intermediate, the process moves to the critical halogen-fluorine substitution phase, transforming the structure into Formula (2). 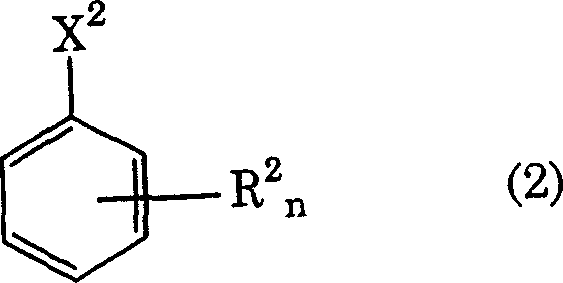 This step utilizes anhydrous hydrogen fluoride (HF) in a molar excess of 10 to 500 times relative to the haloalkyl substrate, typically conducted at elevated temperatures between 150°C and 200°C for carboxylic acid derivatives. The thermodynamic driving force provided by the massive excess of HF ensures that the equilibrium is shifted overwhelmingly towards the formation of carbon-fluorine bonds, displacing the chlorine atoms completely. The use of such a significant excess of HF is mechanistically vital; if the molar ratio drops below 10, the substitution becomes incomplete, leaving residual chlorine atoms that are difficult to remove and degrade the purity profile. The combination of low-metal starting materials and aggressive fluorination conditions results in a final product where residual halogens are suppressed to below 1wt% and transition metals are undetectable by ICP analysis, meeting the stringent specifications for electronic grade materials.
This step utilizes anhydrous hydrogen fluoride (HF) in a molar excess of 10 to 500 times relative to the haloalkyl substrate, typically conducted at elevated temperatures between 150°C and 200°C for carboxylic acid derivatives. The thermodynamic driving force provided by the massive excess of HF ensures that the equilibrium is shifted overwhelmingly towards the formation of carbon-fluorine bonds, displacing the chlorine atoms completely. The use of such a significant excess of HF is mechanistically vital; if the molar ratio drops below 10, the substitution becomes incomplete, leaving residual chlorine atoms that are difficult to remove and degrade the purity profile. The combination of low-metal starting materials and aggressive fluorination conditions results in a final product where residual halogens are suppressed to below 1wt% and transition metals are undetectable by ICP analysis, meeting the stringent specifications for electronic grade materials.
How to Synthesize 3,5-Bis(trifluoromethyl)benzoic Acid Efficiently
The practical implementation of this patented technology for the synthesis of key intermediates like 3,5-bis(trifluoromethyl)benzoic acid involves a tightly controlled sequence of unit operations designed to maximize yield while minimizing impurity ingress. The process begins with the analytical verification of the starting 3,5-dimethylbenzoyl chloride to ensure transition metal levels are within the specified sub-500ppm limit, followed by photochlorination at approximately 140°C until gas chromatography confirms complete conversion to the trichloromethyl derivative. Detailed standardized synthesis steps see the guide below for the specific operational parameters regarding pressure, light intensity, and HF handling protocols that ensure safety and reproducibility at scale.
- Purify the starting alkylbenzene derivative to reduce Group 3-12 transition metal content to below 500ppm, preferably under 100ppm, to prevent nuclear halogenation side reactions.
- Perform side-chain photohalogenation using chlorine gas under light irradiation at 80-200°C to convert alkyl groups into haloalkyl groups without affecting the aromatic ring.
- Conduct halogen-fluorine substitution using at least 10 moles of anhydrous HF per mole of haloalkyl derivative at 150-200°C to ensure complete fluorination and minimal residual halogen impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this low-metal synthesis route offers profound strategic advantages beyond mere technical specification compliance. By eliminating the root cause of impurity formation at the raw material stage, the process drastically simplifies the downstream purification train, removing the need for expensive heavy metal scavengers, complex ion-exchange columns, or multiple recrystallization cycles that traditionally inflate production costs and extend lead times. This streamlined workflow translates directly into substantial cost savings in electronic chemical manufacturing, as the yield loss associated with aggressive purification is minimized, and the throughput of the production facility is significantly enhanced. Furthermore, the robustness of the process against side reactions ensures consistent batch-to-batch quality, reducing the risk of supply disruptions caused by out-of-specification batches that require reprocessing or disposal.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the prevention of nuclear halogenation side reactions mean that the costly and time-consuming steps required to remove heavy metal residues from the final product are rendered unnecessary. This qualitative shift in process design removes the dependency on expensive purification resins and reduces solvent consumption associated with multiple washing and recrystallization steps, leading to a leaner and more economically efficient production model that improves overall margin potential without compromising on the high-purity standards demanded by the pharmaceutical and semiconductor sectors.
- Enhanced Supply Chain Reliability: By utilizing industrially available alkylbenzene derivatives that are simply pre-treated to lower metal content, the supply chain becomes less vulnerable to the fluctuations and shortages often associated with specialized high-purity reagents. The process relies on commodity chemicals like chlorine and hydrogen fluoride, which are widely sourced, ensuring that production continuity is maintained even during periods of raw material volatility. This stability allows for more accurate forecasting and inventory management, reducing the lead time for high-purity fluoroalkylbenzene derivatives and enabling suppliers to respond more agilely to sudden spikes in demand from downstream electronics or drug manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the use of exotic catalysts or hazardous reagents that pose significant handling challenges at large volumes. The ability to recycle excess hydrogen fluoride through distillation further enhances the environmental profile of the operation by minimizing waste generation and reducing the consumption of fresh reagents. This alignment with green chemistry principles facilitates easier regulatory approval and permits for commercial scale-up of complex pharmaceutical intermediates, ensuring that the manufacturing facility remains compliant with increasingly stringent environmental regulations regarding halogenated waste and heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this low-metal fluorination technology, focusing on the critical parameters that define product quality and process safety. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how specific variables like metal content and HF ratios influence the final outcome.
Q: Why is the transition metal content in the starting material critical for this synthesis?
A: Transition metals above 500ppm catalyze unwanted nuclear halogenation on the benzene ring instead of the desired side-chain reaction, leading to difficult-to-remove isomeric impurities and higher residual metal content in the final API intermediate.
Q: What is the required molar ratio of HF for effective fluorination?
A: The patent specifies using at least 10 moles of HF relative to the haloalkyl benzene derivative, with a preferred range of 20 to 200 moles, to drive the equilibrium towards complete halogen-fluorine substitution and minimize residual chlorine.
Q: What are the primary applications for these high-purity fluoroalkyl compounds?
A: These derivatives serve as critical intermediates in the manufacture of pharmaceuticals and electronic materials, such as photoresists, where trace metal and halogen impurities can cause device failure or biological toxicity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Bis(trifluoromethyl)benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory-scale innovation to industrial reality requires more than just a patent; it demands deep process engineering expertise and a commitment to uncompromising quality standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of metal purification and fluorination chemistry described in CN1653029A is executed with precision at every batch size. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced ICP-MS and GC-MS instrumentation to verify that every shipment of fluoroalkyl intermediates meets the exacting requirements for residual metals and halogens, providing our partners with the confidence needed for their own critical manufacturing processes.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this low-impurity methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities as a reliable agrochemical intermediate supplier and partner in electronic materials can drive value and efficiency in your upcoming projects.
