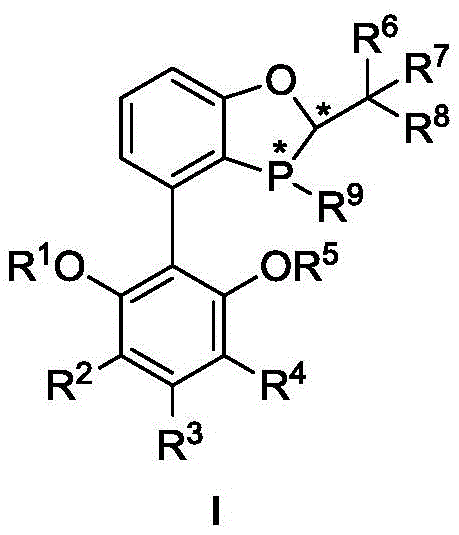
Advanced Chiral Phosphine Ligands for Efficient Synthesis of Ortho-Tetra-Substituted Biaryl Intermediates
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing axially chiral biaryl scaffolds, which serve as critical backbones in numerous bioactive natural products and drug candidates. Patent CN111662331B introduces a groundbreaking class of phosphine ligands designed specifically to overcome the steric and electronic challenges associated with synthesizing ortho-tetra-substituted biaryl compounds. These novel ligands, characterized by a unique benzodioxaphosphole core substituted with bulky alkyl or cycloalkyl groups, enable highly enantioselective Suzuki-Miyaura coupling reactions. The technology addresses a significant gap in current synthetic capabilities, offering a pathway to complex molecular architectures that were previously difficult to access with high optical purity. For R&D teams focused on developing next-generation therapeutics, this innovation represents a pivotal advancement in catalytic efficiency and substrate versatility.
Traditional approaches to synthesizing axially chiral biaryls have long been plagued by inherent inefficiencies and limitations that hinder large-scale production. Conventional methods often rely on the resolution of racemic mixtures, a process that theoretically caps the maximum yield at 50% unless dynamic kinetic resolution is employed, which adds further complexity. Alternatively, the use of stoichiometric chiral auxiliaries or reagents generates substantial chemical waste and increases the overall cost of goods due to the consumption of expensive chiral sources. Furthermore, existing asymmetric catalytic methods frequently suffer from narrow substrate scope, particularly struggling with the steric congestion found in ortho-tetra-substituted systems. Many prior art ligands fail to induce sufficient stereocontrol when bulky groups are present at the ortho positions, leading to poor enantiomeric excess values. The novel approach detailed in the patent circumvents these issues by utilizing a catalytic amount of a highly tailored chiral ligand that creates a precise steric environment around the palladium center.
The new methodology leverages the unique structural features of the disclosed phosphine ligands to facilitate asymmetric coupling under mild conditions. By incorporating bulky substituents such as tert-butyl, cyclopentyl, or isopropyl groups at strategic positions on the ligand framework, the catalyst effectively differentiates between the enantiotopic faces of the reacting species. This steric differentiation is crucial for achieving high enantioselectivity in the formation of the hindered biaryl bond. The process operates efficiently at moderate temperatures ranging from 40 to 100°C and tolerates a wide array of functional groups, including aldehydes, which are notoriously sensitive in cross-coupling reactions. This functional group tolerance eliminates the need for extensive protection and deprotection sequences, thereby streamlining the synthetic route and improving overall step economy. The ability to directly couple aldehyde-containing substrates opens new avenues for the rapid assembly of complex intermediates used in the synthesis of natural products like Korupensamine and Michellamine B.
Mechanistic Insights into Asymmetric Suzuki-Miyaura Coupling
The efficacy of this catalytic system stems from the intricate interplay between the chiral ligand and the palladium metal center during the catalytic cycle. The ligand, featuring a rigid benzodioxaphosphole backbone, coordinates to the palladium atom to form a chiral pocket that dictates the trajectory of the oxidative addition and reductive elimination steps. The bulky substituents on the ligand, such as the 3,5-dicyclopentylphenyl or 3,5-diisopropylphenyl moieties, project into the coordination sphere, creating significant steric hindrance that favors the formation of one atropisomer over the other. This steric bulk is essential for overcoming the rotational barrier of the biaryl product and locking it into a single chiral configuration. The mechanism proceeds through the standard Pd(0)/Pd(II) cycle, where the aryl halide undergoes oxidative addition, followed by transmetallation with the organoboron species, and finally reductive elimination to release the chiral biaryl product. The specific geometry imposed by the ligand ensures that the reductive elimination occurs with high fidelity, preserving the axial chirality established during the bond-forming event.
Impurity control is another critical aspect where this technology excels, particularly regarding the suppression of homocoupling byproducts and proto-dehalogenation side reactions. The optimized reaction conditions, utilizing mild inorganic bases like potassium phosphate in mixed solvent systems of toluene and water, minimize the degradation of sensitive functional groups. The high turnover frequency of the catalyst allows for low catalyst loadings, typically between 0.0005 and 0.01 molar equivalents, which reduces the residual metal content in the final product. This is a vital consideration for pharmaceutical manufacturing, where strict limits on heavy metal impurities must be met. The robustness of the catalytic system ensures consistent performance across different batches, reducing the risk of batch-to-batch variability in enantiomeric purity. Furthermore, the ligand's stability under reaction conditions prevents decomposition pathways that could lead to the formation of achiral byproducts, ensuring a clean reaction profile that simplifies downstream purification processes.
How to Synthesize Ortho-Tetra-Substituted Biaryl Efficiently
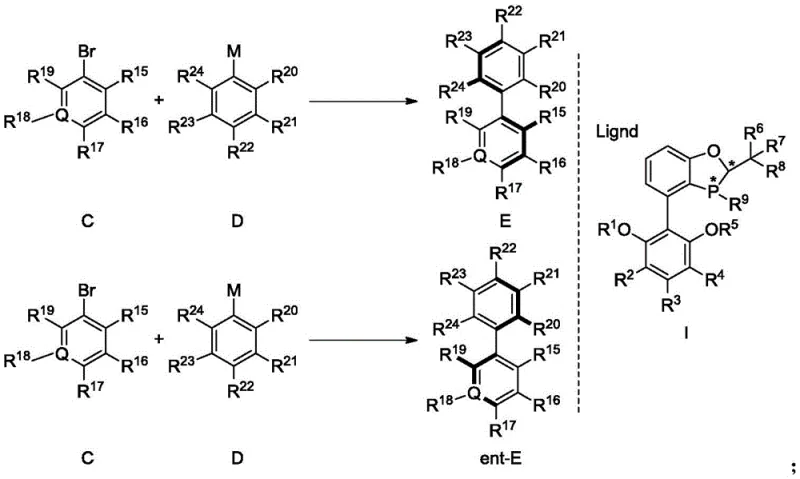
The preparation of these high-value chiral intermediates involves a straightforward yet highly optimized protocol that balances reactivity with selectivity. The process begins with the careful selection of the appropriate ligand variant from the disclosed family, such as the BaryPhos ligand (I-6), which has demonstrated exceptional performance across a broad range of substrates. The reaction setup requires an inert atmosphere to protect the active palladium species from oxidation, typically achieved using nitrogen or argon purging. The choice of solvent system is also critical, with mixtures of aromatic hydrocarbons and water proving most effective for solubilizing both organic substrates and inorganic bases. Detailed standardized synthesis steps for implementing this technology in a laboratory or pilot plant setting are provided below to ensure reproducibility and optimal outcomes.
- Prepare the reaction mixture by combining the aryl halide substrate, aryl boronic acid or potassium arylfluoroborate, and the chiral phosphine ligand (e.g., I-6) in a suitable solvent system.
- Add a palladium catalyst source such as tris(dibenzylideneacetone)dipalladium and an inorganic base like potassium phosphate under inert atmosphere.
- Heat the reaction mixture to temperatures between 40-100°C, monitor progress via HPLC or TLC, and purify the resulting chiral biaryl product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this catalytic technology offers substantial strategic benefits that extend beyond mere technical performance. The shift from stoichiometric chiral sources to catalytic asymmetric synthesis fundamentally alters the cost structure of manufacturing complex biaryl intermediates. By eliminating the need for expensive chiral resolving agents or auxiliaries that are consumed in equimolar amounts, the process significantly reduces raw material costs. The high yields and enantioselectivity reported in the patent data mean that less starting material is wasted, leading to a more efficient utilization of resources. This efficiency translates directly into improved margins and a more competitive pricing structure for the final active pharmaceutical ingredients. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to lower operational expenditures and a smaller carbon footprint for the manufacturing facility.
- Cost Reduction in Manufacturing: The transition to a catalytic process removes the financial burden of purchasing and disposing of stoichiometric chiral reagents. Since the ligand is used in sub-stoichiometric amounts, the cost per kilogram of the product is drastically lowered compared to resolution-based methods. The high atom economy of the Suzuki-Miyaura coupling further minimizes waste generation, reducing the costs associated with waste treatment and disposal. Moreover, the ability to tolerate sensitive functional groups like aldehydes without protection strategies shortens the synthetic sequence, saving both time and money on reagents and labor. These cumulative effects result in a leaner, more cost-effective manufacturing process that enhances overall profitability.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, such as aryl halides and arylboronic acids, are widely available commodities in the global chemical market. This availability ensures a stable supply chain that is less susceptible to disruptions caused by the scarcity of specialized chiral pool materials. The robustness of the catalyst system allows for consistent production schedules, minimizing the risk of delays due to failed batches or low yields. The scalability of the process from milligram to multi-kilogram scales has been demonstrated, providing confidence in the ability to meet increasing demand as a drug candidate progresses through clinical trials. This reliability is crucial for maintaining uninterrupted supply to downstream customers and meeting regulatory timelines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common solvents and reagents that are easy to handle on a large scale. The reduced generation of chemical waste aligns with increasingly stringent environmental regulations and corporate sustainability goals. By avoiding the use of hazardous chiral auxiliaries and minimizing solvent usage through higher concentrations, the process supports greener manufacturing practices. The low residual palladium levels in the product simplify the purification workflow, reducing the need for extensive metal scavenging steps. This ease of scale-up ensures that the technology can be seamlessly transferred from R&D to commercial production without significant re-engineering, facilitating faster time-to-market for new pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and practical application of this patented technology. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What enantiomeric excess (ee) can be achieved with these ligands?
A: The patented ligands consistently achieve high optical purity, with ee values typically exceeding 86% and often reaching above 90% for various ortho-tetra-substituted biaryl substrates.
Q: Are these ligands compatible with sensitive functional groups?
A: Yes, the ligands demonstrate excellent functional group tolerance, specifically accommodating aldehyde groups which are often challenging in traditional coupling protocols.
Q: What is the typical catalyst loading required?
A: The process is highly efficient, operating effectively with low palladium catalyst loadings ranging from 0.0005 to 0.01 molar equivalents relative to the substrate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BaryPhos Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and process development, possessing the technical expertise to leverage advanced catalytic technologies like those described in CN111662331B. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral intermediate meets the highest quality standards required by the global pharmaceutical industry. Our commitment to excellence ensures that you receive materials that are not only chemically pure but also possess the precise stereochemical integrity necessary for biological activity.
We invite you to collaborate with us to explore the full potential of these novel phosphine ligands for your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project's unique requirements. Please contact us to request specific COA data and route feasibility assessments that will demonstrate how our capabilities can accelerate your timeline and optimize your budget. Let us be your partner in turning complex chemical challenges into commercial successes.
