Scalable Synthesis of Chiral 2,2'-Dipyrrolidine Ligands via Ellman Auxiliary Strategy
The development of efficient synthetic routes for C2-symmetric chiral diamines represents a critical advancement in modern organic synthesis, particularly for the preparation of sophisticated ligands used in catalytic transformations. Patent CN101838231B discloses a novel class of pyrrolidine derivatives and their application in the synthesis of 2,2'-dipyrrolidine, a privileged scaffold in asymmetric catalysis. This technology addresses the longstanding challenges associated with constructing C2-symmetric axes by leveraging the powerful stereocontrolling capabilities of Ellman's tert-butylsulfinamide auxiliary. Unlike traditional methods that rely on inefficient resolution or expensive chiral inducers, this approach utilizes readily available Boc-protected prolinol as a starting material, offering a streamlined pathway to high-value chiral building blocks. The significance of this methodology extends to the preparation of iron-based catalysts for selective C-H bond oxidation, as pioneered by White et al., where the steric and electronic properties of the 2,2'-dipyrrolidine ligand are paramount for achieving high regioselectivity and stereoselectivity in complex molecule functionalization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active 2,2'-dipyrrolidine has been fraught with significant inefficiencies that hinder its widespread industrial adoption. Early methodologies, such as the UV-light induced coupling reported by Crabtree et al., suffered from inherently low reaction efficiency, often requiring reaction times extending over dozens of days to reach completion. Furthermore, these photochemical processes frequently generated substantial amounts of meso compounds as byproducts, necessitating cumbersome separation protocols that drastically reduced the overall yield of the desired enantiomer. Another prominent approach involving zinc reagents, while offering improved stereoselectivity, relied heavily on the use of chiral methylbenzylamine as an inducing agent. This reliance introduced a major economic bottleneck, as the chiral amine is costly and its stoichiometric use inflates the raw material expenses, making the process less attractive for large-scale commercial manufacturing where cost margins are tightly controlled.
The Novel Approach
In stark contrast to these legacy methods, the novel synthetic route detailed in the patent utilizes a strategy centered on chiral tert-butylsulfinamide, which acts as a robust and removable chiral auxiliary. This approach fundamentally shifts the paradigm from resolution-based strategies to direct asymmetric synthesis, thereby theoretically doubling the maximum yield potential compared to racemic resolution. The process begins with the conversion of inexpensive Boc-L-prolinol into a chiral sulfinimine, which then undergoes a highly diastereoselective nucleophilic addition. This sequence ensures that the new chiral centers are established with high fidelity, minimizing the formation of unwanted diastereomers. By avoiding the use of precious metal catalysts or exotic chiral reagents in the initial steps, the process significantly lowers the barrier to entry for production, offering a cost-effective and operationally simple alternative that is highly amenable to scale-up in a GMP environment.
Mechanistic Insights into Ellman Auxiliary-Mediated Asymmetric Synthesis
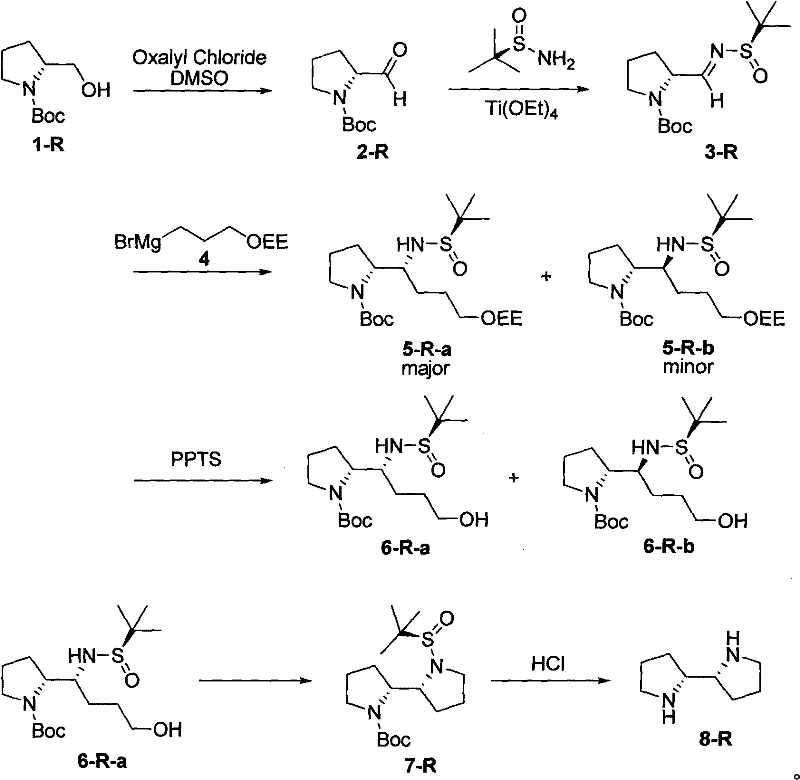
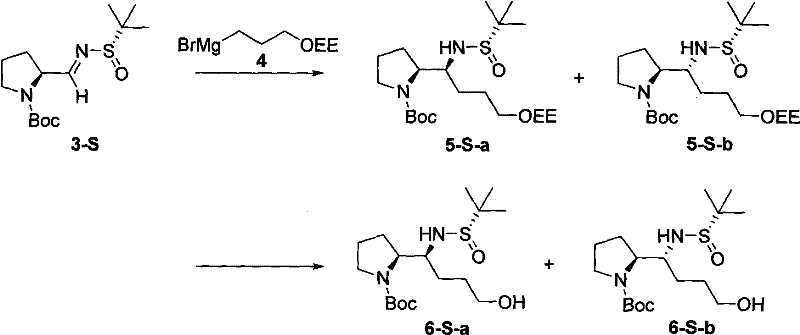
The core of this synthetic innovation lies in the precise manipulation of stereochemistry through the formation of a chiral N-tert-butanesulfinyl imine. The mechanism initiates with the Swern oxidation of Boc-protected prolinol, generating a reactive aldehyde intermediate under mild conditions that preserve the integrity of the protecting groups. Subsequently, the condensation with (S)-tert-butylsulfinamide in the presence of titanium tetraethoxide creates a rigid chiral environment around the imine nitrogen. This titanium complexation is crucial as it organizes the transition state, ensuring that the subsequent nucleophilic attack occurs from the less hindered face of the imine. The result is a highly diastereoselective addition of the Grignard reagent, where the bulky tert-butylsulfinyl group effectively shields one face of the molecule, directing the incoming nucleophile to establish the desired stereochemistry at the new carbon center with exceptional precision.
Following the establishment of the chiral center, the synthetic pathway proceeds through a carefully orchestrated deprotection and cyclization sequence. The removal of the 1-ethoxyethyl (OEE) protecting group using catalytic PPTS reveals a free hydroxyl group, which serves as the nucleophile for the ring-closing step. The activation of this hydroxyl group, typically via mesylation or tosylation, converts it into a good leaving group, facilitating an intramolecular nucleophilic substitution reaction. This cyclization step is critical as it forms the second pyrrolidine ring, locking the C2-symmetric architecture into place. The final acidic hydrolysis removes the Boc and sulfinyl groups simultaneously, unveiling the free diamine structure. This mechanistic elegance ensures that impurities arising from incomplete reactions or side pathways are minimized, resulting in a product profile that meets the stringent purity requirements demanded by the pharmaceutical industry for advanced intermediate applications.
How to Synthesize Chiral 2,2'-Dipyrrolidine Efficiently
The execution of this synthesis requires strict adherence to temperature controls and reagent stoichiometry to maximize the diastereomeric ratio and overall yield. The process is designed to be robust, utilizing common organic solvents such as dichloromethane and THF, which simplifies the logistical burden on the supply chain. The initial oxidation and imine formation steps set the stage for the critical carbon-carbon bond-forming event, where the choice of Grignard reagent and reaction temperature plays a pivotal role in determining the final stereochemical outcome. Operators must ensure that the reaction mixture is maintained at low temperatures during the addition phase to prevent thermal erosion of stereoselectivity, followed by a controlled warm-up to drive the reaction to completion. The subsequent workup procedures involve standard aqueous extractions and drying steps, which are easily transferable from laboratory glassware to industrial reactor vessels without requiring specialized equipment.
- Oxidize Boc-protected prolinol to the corresponding aldehyde using Swern oxidation conditions (DMSO, Oxalyl Chloride).
- Condense the aldehyde with (S)-tert-butylsulfinamide using Ti(OEt)4 to form the chiral sulfinimine intermediate.
- Perform a diastereoselective Grignard addition followed by acid-catalyzed deprotection and intramolecular cyclization to close the second pyrrolidine ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers compelling advantages by decoupling production costs from the volatility of expensive chiral pool materials. The primary starting material, Boc-protected prolinol, is derived from abundant natural amino acids, ensuring a stable and predictable supply chain that is not subject to the same geopolitical or manufacturing constraints as synthetic chiral amines. By eliminating the need for stoichiometric amounts of costly chiral inducers like methylbenzylamine, the raw material cost per kilogram of the final product is substantially reduced. Furthermore, the high stereoselectivity inherent in the Ellman auxiliary approach minimizes the generation of hard-to-separate diastereomeric impurities, which reduces the burden on downstream purification processes and lowers the consumption of silica gel and solvents typically associated with extensive chromatographic separations.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and the use of commodity chemicals for the backbone construction lead to a significant optimization of the bill of materials. The process avoids the theoretical 50% yield loss associated with racemic resolution, effectively doubling the material throughput from the same amount of starting feedstock. Additionally, the mild reaction conditions reduce energy consumption related to heating or extreme cooling, contributing to lower utility costs per batch. The simplicity of the workup procedures further reduces labor hours and solvent waste disposal costs, creating a leaner and more economically viable manufacturing model.
- Enhanced Supply Chain Reliability: Reliance on widely available reagents such as oxalyl chloride, DMSO, and magnesium turnkeys mitigates the risk of supply disruptions caused by single-source dependencies. The robustness of the chemistry allows for flexible sourcing of raw materials, as the specifications for reagents like Ti(OEt)4 and Grignard precursors are standard across the chemical industry. This flexibility empowers procurement teams to negotiate better terms with multiple vendors, ensuring continuity of supply even during market fluctuations. The scalability of the process means that production volumes can be ramped up quickly to meet surging demand without the need for lengthy process re-validation or equipment modification.
- Scalability and Environmental Compliance: The synthetic pathway is designed with green chemistry principles in mind, utilizing solvents that are easily recoverable and recyclable in a closed-loop system. The absence of heavy metal catalysts in the key bond-forming steps simplifies the waste stream treatment, reducing the environmental footprint and compliance costs associated with hazardous waste disposal. The high atom economy of the cyclization step ensures that the majority of the input mass is incorporated into the final product, minimizing waste generation. This alignment with environmental standards facilitates smoother regulatory approvals and enhances the sustainability profile of the supply chain, which is increasingly important for downstream pharmaceutical customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and benefits of adopting this route for commercial production. Understanding these details is essential for technical teams evaluating the integration of this intermediate into their broader manufacturing workflows.
Q: What is the primary advantage of using Ellman's auxiliary in this synthesis?
A: The use of tert-butylsulfinamide allows for high diastereoselectivity during the Grignard addition step, effectively controlling the stereochemistry of the new chiral center without the need for difficult chiral resolution processes.
Q: Can this process be scaled for industrial production?
A: Yes, the patent highlights that the raw materials such as Boc-prolinol are commercially available and inexpensive, and the reaction conditions utilize standard reagents suitable for large-scale manufacturing.
Q: What are the key purification methods mentioned?
A: The process primarily utilizes column chromatography for high-purity isolation in the lab scale, but the simplified workup procedures suggest potential for crystallization or extraction-based purification in larger scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Dipyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral ligands play in the development of next-generation pharmaceuticals and agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the enantiomeric excess and chemical purity of every batch. Our facility is equipped to handle the specific handling requirements of air- and moisture-sensitive reagents often used in such asymmetric syntheses, guaranteeing consistent product quality that meets global regulatory standards.
We invite you to collaborate with us to leverage this innovative synthetic technology for your specific application needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your overall production budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. We are dedicated to supporting your R&D and supply chain goals with reliable, high-performance chemical solutions that drive your projects forward.
