Advanced Rhodium-Catalyzed Synthesis of Tetrahydronaphthalene Derivatives for Commercial Scale-up
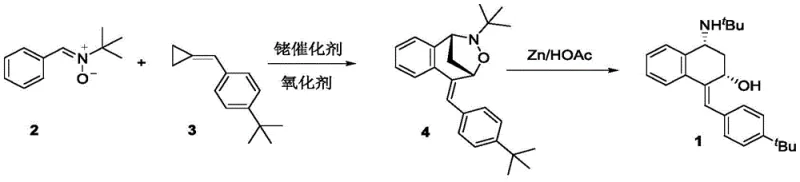
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to complex molecular scaffolds, particularly those containing fused ring systems essential for bioactive compounds. Patent CN107915649B introduces a significant advancement in this domain by disclosing a novel preparation method for 1-(Z-4-tert-butyl benzylidene)-4-tert-butylamine-1,2,3,4-tetrahydronaphthalene-2-ol. This specific tetrahydronaphthalene derivative serves as a critical building block in the synthesis of various medicinal agents and agrochemical intermediates. The disclosed methodology leverages a sophisticated rhodium-catalyzed strategy to construct the core skeleton with high precision. Unlike traditional approaches that often struggle with regioselectivity and harsh conditions, this invention utilizes a nitrone and methylene cyclopropane coupling strategy. The process is characterized by its exceptional atom economy and operational simplicity, making it a highly attractive candidate for industrial adoption. By addressing the inherent difficulties in synthesizing bridged ring compounds, this technology offers a robust solution for producing high-purity intermediates required in modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of compounds containing bridged ring structures, such as the oxazolidine intermediates required for this target molecule, has been fraught with significant technical challenges. Conventional methodologies often rely on multi-step sequences that suffer from low overall efficiency and poor atom economy. These traditional routes frequently necessitate the use of hazardous reagents or extreme reaction conditions, which not only increase safety risks but also complicate waste management and environmental compliance. Furthermore, the construction of the specific stereochemistry required for bioactive tetrahydronaphthalene derivatives is often difficult to control using older techniques, leading to mixtures of isomers that require costly and time-consuming purification steps. The low flexibility of these prior art methods means that scaling up production often results in diminished yields and inconsistent quality, creating bottlenecks in the supply chain for downstream pharmaceutical manufacturers. Consequently, there has been a persistent demand for a more streamlined and reliable synthetic protocol that can overcome these structural and economic hurdles.
The Novel Approach
The innovative method described in the patent fundamentally shifts the paradigm by employing a direct addition reaction between a nitrone and a methylene cyclopropane. This approach bypasses the need for cumbersome precursor synthesis and allows for the rapid assembly of the complex bridged oxazolidine structure in a single catalytic step. The use of a rhodium catalyst in conjunction with a silver-based oxidant facilitates this transformation under remarkably mild conditions, typically ranging from 30°C to 50°C. This gentle thermal profile preserves the integrity of sensitive functional groups and minimizes the formation of degradation byproducts. Following the initial cyclization, the process employs a straightforward zinc-mediated reductive ring-opening to unveil the final tetrahydronaphthalene-2-ol core. This two-step sequence represents a drastic simplification compared to legacy methods, offering superior selectivity and significantly reducing the number of unit operations required. The result is a highly efficient pathway that delivers the target molecule with excellent purity and yield, directly addressing the inefficiencies of the past.
Mechanistic Insights into Rhodium-Catalyzed Cyclization and Reductive Opening
The core of this technological breakthrough lies in the precise orchestration of the rhodium-catalyzed addition reaction. The mechanism initiates with the activation of the methylene cyclopropane by the trivalent rhodium species, likely forming a reactive metallacycle intermediate. This activated species then engages with the nitrone dipole in a highly regioselective manner to construct the oxazolidine ring system with a bridged architecture. The choice of ligand, specifically the pentamethylcyclopentadienyl group on the rhodium center, plays a crucial role in stabilizing the active catalyst and directing the stereochemical outcome of the reaction. The presence of the silver oxidant is essential for regenerating the active rhodium species, ensuring the catalytic cycle continues efficiently without deactivation. This mechanistic elegance allows for the tolerance of various substituents on the aromatic rings, providing versatility for synthesizing diverse analogues. The subsequent step involves the cleavage of the nitrogen-oxygen bond within the bridged intermediate. By utilizing zinc powder in an acidic medium, the system achieves a chemoselective reduction that opens the ring without affecting other sensitive functionalities, such as the olefinic double bond or the tert-butyl groups.
Controlling the impurity profile is paramount for any intermediate intended for pharmaceutical use, and this mechanism inherently supports high purity standards. The high selectivity of the rhodium catalyst minimizes the formation of regioisomers and oligomeric byproducts that are common in non-catalyzed cycloadditions. Furthermore, the mild reaction conditions prevent thermal decomposition, which is a frequent source of colored impurities and tars in organic synthesis. The workup procedure, involving simple filtration through celite and standard column chromatography, effectively removes metal residues and inorganic salts. The patent data indicates that the intermediate oxazolidine can be obtained with purity greater than 95%, and the final product maintains similar high-quality specifications. This level of control over the impurity spectrum reduces the burden on downstream purification processes, ensuring that the final API or advanced intermediate meets stringent regulatory requirements. The robustness of the catalytic system against moisture and air, as noted in the examples, further enhances its reliability for consistent batch-to-batch production.
How to Synthesize 1-(Z-4-tert-butyl benzylidene)-4-tert-butylamine-1,2,3,4-tetrahydronaphthalene-2-ol Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the catalyst and oxidant to maximize turnover numbers. The process begins with the preparation of the catalytic solution in a suitable solvent like trifluoroethanol, followed by the sequential addition of substrates under an inert atmosphere. Maintaining the temperature within the optimal window of 30°C to 50°C is critical for balancing reaction rate and selectivity. Once the bridged intermediate is formed and isolated, the reductive ring-opening step must be conducted with sufficient zinc powder to ensure complete conversion. Detailed standardized operating procedures for this specific transformation are outlined below to guide process chemists in replication and scale-up.
- Prepare the reaction mixture by combining Cp*Rh(OAc)2 catalyst and silver acetate oxidant in trifluoroethanol solvent under nitrogen protection.
- Add nitrone and methylene cyclopropane substrates to the mixture and stir at 30-50°C for 24 hours to form the bridged oxazolidine intermediate.
- Perform reductive ring-opening by treating the isolated intermediate with zinc powder and acetic acid in THF to yield the final tetrahydronaphthalene alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed route offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the substantial simplification of the manufacturing process, which directly translates to reduced operational complexity and lower overhead costs. By condensing the synthesis into fewer steps with higher yields, manufacturers can significantly decrease the consumption of solvents and reagents per kilogram of product. This efficiency gain is compounded by the use of readily available starting materials, such as nitrones and methylene cyclopropanes, which are commercially accessible and do not require custom synthesis. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and reduced utility expenses. These factors collectively enhance the economic viability of producing this complex intermediate on a large scale.
- Cost Reduction in Manufacturing: The elimination of harsh reagents and the reduction in reaction steps lead to a streamlined production workflow that minimizes waste generation. Since the process avoids the use of expensive transition metals beyond the catalytic amount of rhodium, and utilizes common silver salts as oxidants, the raw material costs are optimized. The high selectivity reduces the need for extensive purification, saving both time and chromatography media. Furthermore, the ability to operate at near-ambient temperatures lowers the energy intensity of the process, resulting in significant long-term savings on utility bills. These cumulative efficiencies allow for a more competitive pricing structure for the final intermediate without compromising on quality margins.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially available reagents ensures a resilient supply chain that is less susceptible to disruptions. Unlike processes dependent on exotic or unstable precursors, this method utilizes robust starting materials that can be sourced from multiple vendors. The robustness of the reaction conditions, which tolerate air and moisture to some extent, reduces the risk of batch failures due to minor environmental fluctuations. This reliability ensures consistent delivery schedules and helps maintain steady inventory levels for downstream customers. Additionally, the scalability of the process from gram-scale to multi-kilogram production provides flexibility to meet fluctuating market demands without requiring major process re-engineering.
- Scalability and Environmental Compliance: The atom economy of this reaction is superior to many traditional alternatives, meaning a higher proportion of reactant atoms end up in the final product rather than as waste. This aligns well with green chemistry principles and simplifies the management of chemical waste streams. The use of zinc and acetic acid in the second step generates benign byproducts that are easier to treat and dispose of compared to heavy metal sludge from stoichiometric oxidants. The process is designed to be scalable, with examples demonstrating success at meaningful scales, indicating readiness for commercial manufacturing. This ease of scale-up reduces the time-to-market for new drugs utilizing this scaffold and ensures regulatory compliance regarding environmental impact assessments.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and embodiments provided in the patent documentation to ensure accuracy and relevance for process development teams. Understanding these nuances is essential for successful technology transfer and commercialization.
Q: What specific rhodium catalyst is preferred for this synthesis?
A: The patent specifies trivalent rhodium salts, particularly pentamethylcyclopentadienyl rhodium diacetate (Cp*Rh(OAc)2), as the optimal catalyst for achieving high yields.
Q: What are the optimal reaction conditions for the addition step?
A: The addition reaction proceeds best at mild temperatures between 30°C and 50°C in a trifluoroethanol solvent, utilizing silver salts as oxidants.
Q: How is the bridged ring structure opened to form the final product?
A: The bridged oxazolidine intermediate undergoes a reductive ring-opening reaction using zinc powder and acetic acid, which cleaves the nitrogen-oxygen bond efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(Z-4-tert-butyl benzylidene)-4-tert-butylamine-1,2,3,4-tetrahydronaphthalene-2-ol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic routes play in accelerating drug development timelines. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN107915649B and is fully prepared to leverage this rhodium-catalyzed methodology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of tetrahydronaphthalene intermediate delivered meets the highest international standards. We are committed to providing a reliable supply of high-quality intermediates that support your R&D and commercial goals.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through advanced process chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments for this compound or related derivatives. By partnering with NINGBO INNO PHARMCHEM, you gain access to a wealth of technical expertise and a dedicated supply partner committed to your success in the competitive pharmaceutical landscape.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
