Scalable Synthesis of 4-Alkoxy-3-Hydroxypicolinic Acid Intermediates via Furfural Rearrangement
Introduction to Advanced Picolinic Acid Manufacturing
The pharmaceutical and agrochemical industries continuously demand high-purity heterocyclic building blocks, particularly functionalized picolinic acids which serve as critical scaffolds for fungicides and kinase inhibitors. Patent CN106573005B introduces a robust and tunable process for the preparation of 4-alkoxy-3-hydroxypicolinic acid derivatives, utilizing furfural as a cost-effective starting material. This technology represents a significant leap forward in process chemistry, replacing traditional, often hazardous or expensive pyridine synthesis routes with a streamlined sequence involving cyanamination, bromination-induced rearrangement, and selective functional group manipulation. For R&D directors and procurement specialists, understanding this pathway is essential for securing a reliable supply chain of these high-value intermediates. The method not only improves overall yield but also offers flexibility in introducing various alkoxy groups at the 4-position, catering to diverse structure-activity relationship (SAR) studies required in modern drug discovery.
The core innovation lies in the efficient conversion of a furan ring into a pyridine ring through a bromination-rearrangement mechanism. This transformation allows for the precise installation of substituents that are otherwise difficult to introduce regioselectively on a pre-formed pyridine core. By leveraging inexpensive commodity chemicals like furfural, ammonium salts, and cyanide sources, the process drastically lowers the raw material cost basis. Furthermore, the patent details specific solvent systems and reaction conditions that enhance safety and scalability, addressing common pain points in the commercial scale-up of complex heterocyclic intermediates. As a leading manufacturer, we recognize the potential of this route to stabilize the supply of critical API precursors while adhering to stringent environmental and quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 4-alkoxy-3-hydroxypicolinic acids often rely on the functionalization of pre-existing pyridine rings, such as 3-hydroxypicolinic acid or its esters. These conventional methods frequently suffer from poor regioselectivity, requiring multiple protection and deprotection steps that inflate production costs and reduce overall throughput. For instance, direct alkoxylation of halogenated pyridines can lead to mixtures of isomers, necessitating energy-intensive chromatographic separations that are impractical on a multi-ton scale. Additionally, many legacy processes utilize harsh reagents or extreme temperatures that pose significant safety risks and generate substantial hazardous waste streams. The reliance on precious metal catalysts for certain coupling reactions further exacerbates cost volatility and introduces the risk of heavy metal contamination, which is strictly regulated in pharmaceutical manufacturing. These inefficiencies create bottlenecks in the supply chain, leading to longer lead times and higher prices for downstream customers seeking reliable pharmaceutical intermediate suppliers.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes a furfural-based strategy that constructs the pyridine ring de novo with inherent regiocontrol. By starting with furfural, the process leverages a biomass-derived feedstock that is abundant and economically stable. The key breakthrough is the bromination-rearrangement step, which simultaneously expands the five-membered furan ring to a six-membered pyridine ring while installing bromine atoms at the 4 and 6 positions. This creates a highly reactive intermediate, 4,6-dibromo-3-hydroxypicolinonitrile, which serves as a versatile platform for further diversification. The subsequent nucleophilic substitution with alkali metal alkoxides proceeds with high selectivity at the 4-position due to the electronic activation provided by the adjacent nitrile and hydroxyl groups. This sequence eliminates the need for complex protecting group strategies and minimizes the formation of regioisomeric impurities. Consequently, the novel approach offers a cleaner, safer, and more cost-effective pathway that aligns perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing.
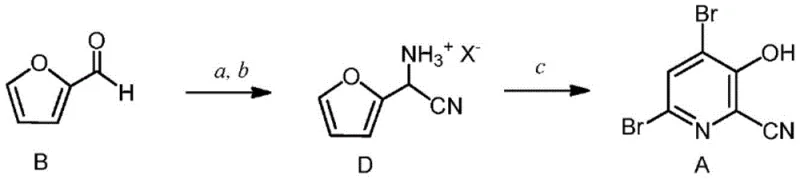
Mechanistic Insights into Bromination-Rearrangement and Substitution
The mechanistic elegance of this process centers on the transformation of the aminonitrile intermediate into the dibromopyridine core. Initially, furfural undergoes a Strecker-type reaction with an ammonia source and a cyanide source to form an alpha-aminonitrile. Upon treatment with a mineral acid, this forms a stable ammonium salt, which is crucial for handling and purification. The pivotal step involves the reaction of this salt with elemental bromine. Mechanistically, this likely proceeds through electrophilic bromination of the furan ring, followed by ring opening and recyclization to form the pyridine system. The presence of excess bromine ensures complete conversion to the 4,6-dibromo species, suppressing the formation of monobrominated byproducts. This rearrangement is highly exothermic and requires careful temperature control, typically maintained between 0°C and 40°C, to manage reaction kinetics and safety. The resulting 4,6-dibromo-3-hydroxypicolinonitrile is a crystalline solid that can be isolated in high purity, providing a robust checkpoint in the manufacturing process.
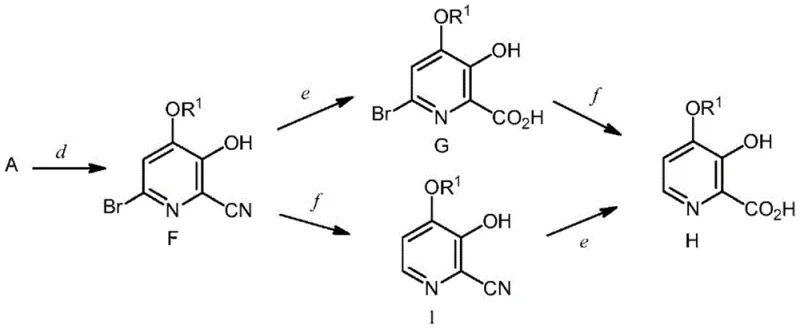
Following the formation of the dibromo intermediate, the process employs a nucleophilic aromatic substitution to introduce the desired alkoxy group. The electron-withdrawing nature of the nitrile group at the 2-position and the bromine at the 6-position activates the 4-position for attack by alkoxide nucleophiles. Using alkali metal alkoxides, such as sodium methoxide or ethoxide, in polar aprotic solvents like DMSO or DMF facilitates this displacement. The reaction conditions are tunable; for example, using a mixture of protic and aprotic solvents can optimize solubility and reaction rate. The patent highlights that this substitution is highly regioselective, leaving the 6-bromo atom intact for potential further functionalization or removal. Finally, the nitrile group is hydrolyzed to the carboxylic acid, and the remaining bromine can be removed via catalytic hydrogenation or metal reduction. This modular mechanism allows for the precise synthesis of various 4-alkoxy analogues, ensuring high purity specifications are met for sensitive applications.
How to Synthesize 4-Alkoxy-3-Hydroxypicolinic Acid Efficiently
The synthesis of 4-alkoxy-3-hydroxypicolinic acid is achieved through a logical sequence of reactions that maximize yield and minimize waste. The process begins with the condensation of furfural with ammonium acetate and potassium cyanide in a biphasic solvent system, forming the aminonitrile intermediate which is then converted to its hydrobromide salt. This salt is subsequently treated with bromine in an aqueous or mixed solvent system to effect the ring expansion and bromination, yielding the key 4,6-dibromo intermediate. The detailed standardized synthesis steps involve precise control of stoichiometry, temperature, and pH to ensure optimal conversion at each stage. For the final transformation, the dibromo intermediate is reacted with sodium methoxide to install the methoxy group, followed by hydrolysis of the nitrile and reductive debromination. This comprehensive route has been validated across multiple scales, from gram-level laboratory experiments to multi-kilogram pilot runs, demonstrating its robustness and reproducibility for commercial production.
- Perform Strecker synthesis on furfural with ammonia and cyanide sources to form aminonitrile intermediates, followed by salt formation.
- Execute bromination and rearrangement using elemental bromine to convert the furan derivative into 4,6-dibromo-3-hydroxypicolinonitrile.
- Conduct nucleophilic substitution with alkali metal alkoxides, followed by nitrile hydrolysis and debromination to yield the final picolinic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this furfural-based synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage is the drastic simplification of the supply chain by utilizing furfural, a widely available bio-based chemical, rather than relying on specialized, low-volume pyridine derivatives that are subject to market volatility. This shift to commodity feedstocks significantly mitigates the risk of supply disruptions and price spikes, ensuring a more stable and predictable cost structure for long-term projects. Furthermore, the high selectivity of the bromination-rearrangement and substitution steps reduces the burden on purification processes. By minimizing the generation of difficult-to-remove isomers and byproducts, the process lowers the consumption of solvents and chromatography media, which are major cost drivers in fine chemical manufacturing. This efficiency translates directly into improved margins and a more competitive pricing model for the final active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in favor of simple alkali metal alkoxides and zinc dust for reduction steps removes the need for costly metal scavenging and validation procedures. Additionally, the ability to perform the debromination and hydrolysis in a one-pot operation reduces unit operations, saving energy, labor, and equipment time. The use of biphasic systems simplifies workups, allowing for the easy separation of inorganic salts without extensive washing or extraction sequences. These cumulative efficiencies result in substantial cost savings throughout the production lifecycle, making the final intermediates more affordable for downstream API synthesis.
- Enhanced Supply Chain Reliability: By anchoring the synthesis on furfural, manufacturers can leverage a global supply network that is less susceptible to the geopolitical and logistical constraints often associated with specialized heterocyclic starting materials. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent mixtures, ensures consistent output even with minor variations in raw material quality. This resilience is critical for maintaining continuous production schedules and meeting tight delivery deadlines for pharmaceutical clients. The process scalability has been demonstrated up to multi-kilogram batches, proving its readiness for commercial scale-up of complex pharmaceutical intermediates without the typical teething problems associated with new chemistry.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by reducing the number of synthetic steps and avoiding the use of persistent organic pollutants. The aqueous workups and the potential for solvent recycling in the biphasic steps contribute to a lower environmental footprint. Moreover, the avoidance of heavy metal catalysts simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. The high yields reported in the patent examples indicate that material throughput is maximized, reducing the volume of waste generated per kilogram of product. This alignment with sustainability goals not only reduces disposal costs but also enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis route. They are derived from the specific embodiments and experimental data provided in the patent documentation, offering clarity on reaction parameters and product quality. Understanding these details is vital for technical teams evaluating the feasibility of integrating this chemistry into their existing manufacturing workflows. The answers reflect the optimized conditions discovered during the development of this process, ensuring that potential adopters can anticipate the performance and requirements of the technology.
Q: What are the key advantages of using furfural as a starting material for picolinic acids?
A: Furfural is a biomass-derived, inexpensive feedstock that allows for a convergent synthesis route. The patent demonstrates that furfural can be efficiently converted into the pyridine core through a bromination-induced ring expansion and rearrangement, bypassing more expensive traditional pyridine precursors.
Q: How does the biphasic solvent system improve the purification of intermediates?
A: The use of a biphasic system (e.g., water and MTBE or ethyl acetate) facilitates the separation of water-soluble inorganic salts from the organic-soluble aminonitrile intermediate. This simplifies workup procedures, reduces solvent consumption, and enhances the purity of the subsequent bromination step.
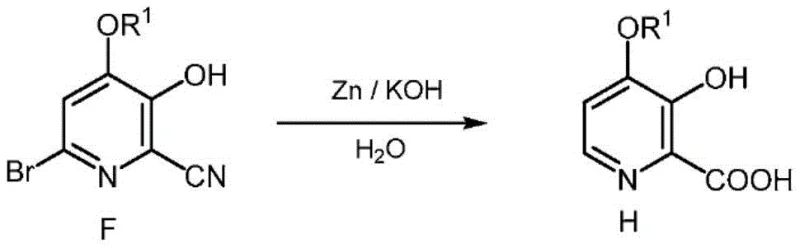
Q: Can the debromination and hydrolysis steps be combined?
A: Yes, the patent discloses a one-pot process where zinc metal and potassium hydroxide are used simultaneously to effect both the reductive removal of the bromine atom and the hydrolysis of the nitrile group, significantly streamlining the production of the final acid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Alkoxy-3-Hydroxypicolinic Acid Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this furfural-based chemistry, including the safe management of bromination reactions and the precise control needed for nucleophilic substitutions. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-alkoxy-3-hydroxypicolinic acid meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that we can provide consistent, high-purity intermediates that facilitate the rapid development of your final drug candidates.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with us, you gain access to our deep expertise in heterocyclic chemistry and our robust supply chain capabilities. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
