Revolutionizing Nefopam Production: A Scalable Ethanol-Based Resolution Strategy for High-Purity API Intermediates
Introduction to Advanced Chiral Resolution Technologies
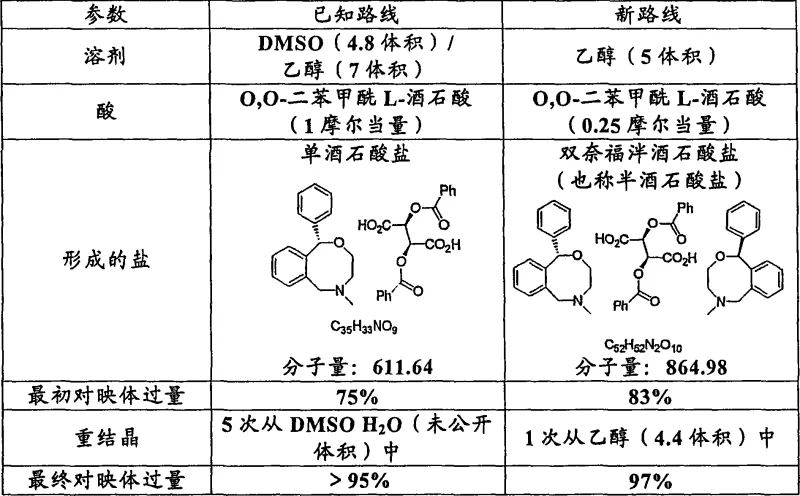
The pharmaceutical industry's relentless pursuit of high-purity single-enantiomer drugs has driven significant innovation in chiral separation technologies, particularly for analgesic agents like Nefopam. Patent CN1894224A discloses a groundbreaking method for increasing the optical purity of Nefopam enantiomers, addressing critical bottlenecks in traditional resolution processes. This technology leverages the formation of a novel bisnefopam hemitartrate salt using substantially single enantiomers of O,O-diaroyltartaric acid as resolving agents. For R&D directors and process chemists, this represents a paradigm shift from labor-intensive, solvent-heavy protocols to a streamlined, economically viable manufacturing route. The ability to produce high-purity API intermediates with reduced environmental impact and lower material consumption is paramount for maintaining competitiveness in the global generic and specialty pharma markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of Nefopam has relied on methods such as those described by Blaschke et al., which utilize a 1:1 molar ratio of the resolving agent to the substrate. These conventional pathways are fraught with inefficiencies that hinder large-scale commercial production. Specifically, the reliance on dimethyl sulfoxide (DMSO) as a co-solvent presents significant downstream processing challenges, including difficult solvent removal and potential toxicity concerns that require rigorous control strategies. Furthermore, achieving the requisite optical purity often necessitates multiple recrystallization steps—typically up to five cycles—which results in substantial yield losses and extended production timelines. The high loading of expensive chiral resolving agents (1.0 molar equivalent) further exacerbates the cost structure, making the final API intermediate less competitive in price-sensitive markets.
The Novel Approach
In stark contrast, the novel approach detailed in the patent introduces a highly efficient resolution strategy that fundamentally alters the stoichiometry and solvent system of the reaction. By forming a unique bisnefopam hemitartrate salt, the process requires only 0.25 molar equivalents of the resolving agent, representing a drastic reduction in raw material consumption. The substitution of the DMSO/ethanol mixture with pure ethanol not only simplifies the solvent recovery infrastructure but also aligns with green chemistry principles favored by modern regulatory bodies. Most notably, this method achieves superior enantiomeric excess (>95%) after merely a single recrystallization step, thereby maximizing yield throughput and minimizing operational complexity. This technological leap transforms the resolution of Nefopam from a bottleneck operation into a robust, scalable unit process suitable for multi-ton production.
Mechanistic Insights into Diastereomeric Salt Formation
The core innovation of this process lies in the specific stoichiometry of the salt formed between the basic Nefopam molecule and the acidic O,O-dibenzoyl-L-tartaric acid. Unlike traditional monotartrate salts which require equimolar amounts of acid and base, this system stabilizes a 'hemitartrate' or 'bis-salt' structure where two molecules of Nefopam interact with one molecule of the tartaric acid derivative. This 2:1 binding ratio is the mechanistic driver behind the reduced loading of the resolving agent. From a thermodynamic perspective, the solubility differences between the diastereomeric salts in ethanol are sufficiently large to allow for selective crystallization of the desired enantiomer complex. The crystal lattice energy of the bis-salt appears to favor the incorporation of the target enantiomer, effectively excluding the unwanted antipode during the nucleation and growth phases.
Furthermore, the mechanism facilitates exceptional impurity control through a simplified purification loop. In conventional resolutions, impurities often co-crystallize or remain trapped in the lattice, necessitating repeated dissolution and precipitation cycles to purge them. However, the unique crystal habit of the bisnefopam hemitartrate salt allows for the rejection of impurities and the minor enantiomer in the mother liquor during the initial crystallization. The subsequent single recrystallization from ethanol acts as a powerful polishing step, leveraging the sharp solubility curve of the salt to push the enantiomeric excess from an initial 83% to over 95%, and potentially up to 99%. This high fidelity in chiral recognition ensures that the final free base, upon liberation, meets the stringent purity specifications required for clinical applications without the need for chromatographic separation.
How to Synthesize (+)-Nefopam Efficiently
The synthesis of high-purity (+)-Nefopam via this patented route involves a straightforward sequence of salt formation, crystallization, and base liberation that is amenable to standard reactor setups. The process begins with the dissolution of racemic Nefopam free base in ethanol, followed by the controlled addition of a sub-stoichiometric amount of O,O-dibenzoyl-L-tartaric acid. The mixture is allowed to stand or stir until the bis-salt precipitates, which is then collected and subjected to a single recrystallization to ensure optical purity. Finally, the purified salt is treated with aqueous base to liberate the free amine, which can be isolated or converted into its hydrochloride salt form. For detailed operational parameters, safety data, and precise stoichiometric calculations, please refer to the standardized synthesis guide below.
- Dissolve racemic Nefopam free base in ethanol and treat with 0.25 molar equivalents of O,O-dibenzoyl-L-tartaric acid to induce crystallization of the bisnefopam hemitartrate salt.
- Collect the precipitated solid via filtration and perform a single recrystallization from ethanol to achieve high optical purity (>95% ee).
- Liberate the pure (+)-Nefopam free base from the purified salt using aqueous sodium hydroxide followed by extraction and conversion to the hydrochloride salt if required.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel resolution technology offers compelling economic and logistical benefits that directly impact the bottom line. The most immediate advantage is the dramatic reduction in the consumption of the chiral resolving agent. By lowering the requirement from 1.0 molar equivalent to just 0.25 molar equivalent, the direct material cost associated with this critical reagent is slashed significantly. Since chiral acids like O,O-dibenzoyl-L-tartaric acid are high-value inputs, this four-fold reduction in usage translates into substantial cost savings per kilogram of API produced. Additionally, the ability to recover the resolving agent in high purity from the mother liquors creates a closed-loop system that further mitigates raw material expenses and reduces waste disposal costs.
- Cost Reduction in Manufacturing: The elimination of DMSO from the solvent system removes the need for specialized high-temperature vacuum drying equipment and extensive wastewater treatment protocols associated with sulfur-containing solvents. Ethanol is a commodity chemical with a well-established global supply chain, offering lower procurement costs and easier handling compared to DMSO. Moreover, the reduction in recrystallization cycles from five down to one drastically cuts utility consumption, including steam for heating, chilled water for cooling, and electricity for agitation and filtration. These cumulative efficiencies result in a leaner manufacturing process with a significantly lower cost of goods sold (COGS), enhancing the margin profile for the final pharmaceutical product.
- Enhanced Supply Chain Reliability: Relying on ethanol as the primary solvent enhances supply chain resilience, as it is readily available in bulk quantities from multiple regional suppliers, reducing the risk of logistics disruptions. The simplified process flow, characterized by fewer unit operations and shorter cycle times, increases the overall throughput capacity of existing manufacturing facilities. This means that production schedules can be accelerated to meet sudden spikes in demand without the need for capital-intensive plant expansions. The robustness of the crystallization process also ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by out-of-specification results that often plague complex multi-step resolutions.
- Scalability and Environmental Compliance: The transition to a greener solvent system aligns perfectly with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste. Ethanol is biodegradable and easier to incinerate or recycle than DMSO, simplifying compliance with environmental permits. The high scalability of the process is evidenced by its simplicity; the crystallization kinetics are favorable for large-scale reactors, avoiding the mixing and heat transfer issues common in viscous DMSO solutions. This ease of scale-up ensures that the technology can be seamlessly transferred from pilot plant to commercial production scales of 100 MT or more, securing a stable long-term supply of high-quality Nefopam intermediates for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process feasibility. Understanding these details is crucial for technical teams assessing the integration of this method into their existing production workflows.
Q: What is the primary advantage of the new Nefopam resolution method over the Blaschke method?
A: The new method utilizes only 0.25 molar equivalents of the resolving agent compared to 1.0 equivalent in the prior art, significantly reducing material costs. Furthermore, it replaces toxic DMSO with ethanol and requires only one recrystallization step instead of five, drastically improving process efficiency and environmental compliance.
Q: What level of enantiomeric excess (ee) can be achieved with this process?
A: The process initially yields a salt with approximately 83% ee, which is upgraded to greater than 95% ee, and potentially up to 99% ee, after a single recrystallization from ethanol, meeting stringent pharmaceutical purity standards.
Q: Is the resolving agent recoverable in this new protocol?
A: Yes, a key advantage of this invention is that the resolving agent can be recovered in a high-purity state from the mother liquors or after liberation of the base, allowing for reuse in subsequent batches and further enhancing the economic viability of the process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nefopam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain a competitive edge in the pharmaceutical intermediate sector. Our technical team has extensively analyzed the methodology disclosed in CN1894224A and possesses the expertise to implement this efficient ethanol-based resolution process at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing environments. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of Nefopam intermediate meets the highest international standards for chiral purity and chemical integrity.
We invite potential partners to engage with our technical procurement team to discuss how this optimized process can drive value for your specific supply chain. By leveraging our capabilities, you can secure a reliable source of high-purity Nefopam that offers both cost efficiency and supply security. We encourage you to request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us collaborate to bring this superior chiral technology to your commercial pipeline, ensuring timely delivery and uncompromised quality for your analgesic drug formulations.
