Advanced One-Pot Synthesis of 1,3-Oxazine-1,3-Oxazole Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to construct complex heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. A significant breakthrough in this domain is detailed in patent CN114163452A, which discloses a novel preparation method for 1,3-oxazine-1,3-oxazole derivatives. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, copper-catalyzed one-pot multicomponent reaction. By leveraging the synergistic effects of a copper compound catalyst and a specific amine ligand, this method enables the direct convergence of an oxirane compound, a terminal alkynone compound, a sulfonyl azide compound, and a nitrile compound. The result is the formation of the target 1,3-oxazine-1,3-oxazole derivative in a single operational step, offering substantial improvements in atom economy and process efficiency. For R&D teams and procurement strategists alike, this innovation addresses critical pain points regarding synthetic complexity and raw material utilization, positioning it as a highly attractive route for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fused heterocyclic systems like 1,3-oxazine-1,3-oxazoles has been fraught with synthetic challenges that hinder efficient manufacturing. Conventional methodologies often rely on sequential stepwise reactions, requiring the isolation and purification of unstable intermediates between each transformation. These traditional routes frequently suffer from harsh reaction conditions, such as extreme temperatures or the use of hazardous reagents, which complicate safety protocols and increase operational costs. Furthermore, many existing methods exhibit poor substrate applicability, meaning that slight modifications to the molecular structure can lead to drastic drops in yield or complete reaction failure. The cumulative effect of these limitations is a prolonged development timeline, increased waste generation due to low atom economy, and difficulties in securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality at scale. The complexity of these legacy processes often results in impurity profiles that are difficult to control, posing significant risks during regulatory filings.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the patented one-pot methodology introduces a robust and versatile synthetic strategy that dramatically simplifies the production landscape. This novel approach utilizes a copper-catalyzed cascade reaction that seamlessly integrates four distinct building blocks into the final fused ring system without the need for intermediate isolation. The reaction proceeds under relatively mild conditions, typically ranging from 25°C to 120°C, and demonstrates exceptional tolerance for various functional groups on the aromatic rings and alkyl chains. As illustrated in the general reaction scheme below, the convergence of the oxirane, alkynone, sulfonyl azide, and nitrile components is highly efficient, leading to single product selectivity and yields that can exceed 90% in optimized examples. This level of efficiency not only reduces the consumption of solvents and reagents but also significantly shortens the overall processing time, thereby facilitating cost reduction in pharmaceutical intermediates manufacturing while maintaining high standards of chemical purity.
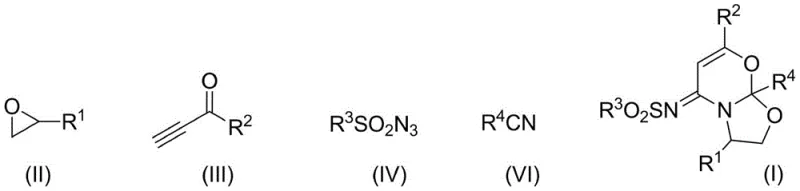
Mechanistic Insights into Copper-Catalyzed Multicomponent Cyclization
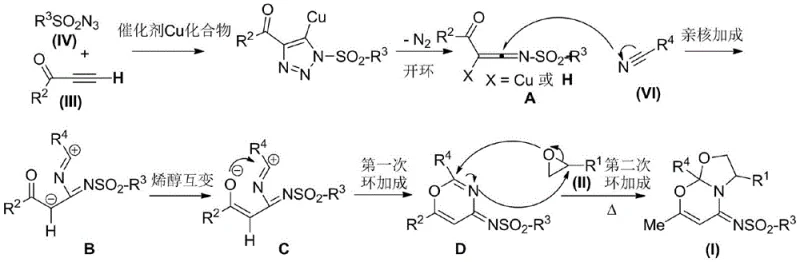
Understanding the underlying mechanistic pathway is crucial for R&D directors aiming to optimize this process for specific analogues. The reaction initiates with a copper-catalyzed 1,3-dipolar cycloaddition between the sulfonyl azide compound and the terminal alkynone compound. This initial step generates a copper-triazole intermediate, which subsequently undergoes a ring-opening rearrangement to form a reactive ketene imine species, designated as intermediate A in the mechanistic cycle. This electrophilic intermediate then engages in a nucleophilic addition reaction with the nitrile compound, forming intermediate B. Following this addition, an enol tautomerization occurs to yield intermediate C, which possesses the necessary electronic configuration for the subsequent cyclization events. The elegance of this mechanism lies in its ability to orchestrate multiple bond-forming events in a precise sequence, driven by the catalytic activity of the copper species and the stabilizing influence of the amine ligand.
The final stages of the mechanism involve intricate intramolecular cyclizations that construct the fused ring system characteristic of the target molecule. Intermediate C undergoes a first intramolecular cycloaddition to generate a carbon-nitrogen double bond within a fused intermediate D. Concurrently, the oxirane compound undergoes ring-opening to form a 1,3-dipole, which then participates in a secondary cycloaddition with the carbon-nitrogen double bond in intermediate D. This final closure forms the stable 1,3-oxazine-1,3-oxazole framework. The detailed catalytic cycle, depicted below, highlights the role of the copper catalyst in facilitating these transformations and the critical function of the ligand in maintaining catalyst stability and activity throughout the reaction course. This deep mechanistic understanding allows for precise tuning of reaction parameters to minimize side reactions and ensure the production of high-purity OLED material precursors or pharmaceutical intermediates with minimal impurity burdens.
How to Synthesize 1,3-Oxazine-1,3-Oxazole Derivatives Efficiently
The practical execution of this synthesis is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment. The process begins by combining the four key reactants—the oxirane compound, the terminal alkynone, the sulfonyl azide, and the nitrile compound (which often serves as both reactant and solvent, such as acetonitrile)—along with the organic ligand in the reaction vessel. A copper compound, preferably cuprous iodide (CuI), is added to initiate the catalytic cycle. The mixture is then heated, typically to around 80°C, and stirred for a period ranging from 1 to 24 hours depending on the specific substrate reactivity. Upon completion, the reaction mixture is cooled, and a simple workup procedure involving extraction with ethyl acetate and water, followed by drying and concentration, yields the crude product. Final purification is achieved through standard techniques such as recrystallization or silica gel column chromatography, ensuring the isolation of the target derivative with high purity. For a comprehensive guide on the specific molar ratios and purification details, please refer to the standardized protocol below.
- Mix oxirane compound, terminal alkynone compound, sulfonyl azide compound, nitrile compound, and organic ligand in an organic solvent.
- Add a copper compound catalyst and react at temperatures between 25-120°C for 1-24 hours.
- Perform purification via extraction, drying, concentration, and chromatography or recrystallization to obtain the high-purity derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the priorities of procurement managers and supply chain heads. The transition from multi-step batch processes to a telescoped one-pot reaction fundamentally alters the cost structure of manufacturing these complex heterocycles. By eliminating the need for intermediate isolation and purification steps, the process drastically reduces the consumption of solvents, filtration media, and labor hours associated with handling multiple batches. Furthermore, the high atom economy inherent in this multicomponent reaction ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal costs and environmental impact. These efficiencies translate into a more competitive pricing model for the final intermediate, allowing downstream manufacturers to optimize their own cost of goods sold without compromising on quality or supply reliability.
- Cost Reduction in Manufacturing: The elimination of transition metal removal steps often required in other catalytic processes, combined with the use of inexpensive and readily available copper salts like CuI, leads to significant operational savings. The high yields observed across various substrates mean that less raw material is required to produce the same amount of product, effectively lowering the unit cost. Additionally, the ability to use the nitrile compound as both a reactant and a solvent simplifies the bill of materials and reduces solvent procurement costs. These factors collectively contribute to a leaner manufacturing process that is economically superior to traditional synthetic routes.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including substituted oxiranes, terminal alkynones, and sulfonyl azides, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of functional groups and substituents, ensures consistent production outcomes even with slight variations in raw material quality. This resilience is critical for maintaining continuous supply lines for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the likelihood of batch failures or delays caused by sensitive reaction requirements. The scalability of the process further ensures that supply can be ramped up quickly to meet fluctuating market demands.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which relies on standard liquid-liquid extraction and crystallization rather than complex distillation or specialized chromatography, makes this process highly amenable to scale-up from kilogram to tonne quantities. The reduced solvent usage and higher overall yield inherently lower the E-factor (mass of waste per mass of product), aligning with increasingly stringent environmental regulations and corporate sustainability goals. The use of common solvents like acetonitrile and ethyl acetate facilitates easier recycling and waste management, ensuring that the manufacturing process remains compliant with global environmental standards while maintaining high throughput capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific project needs.
Q: What are the key advantages of this one-pot synthesis method?
A: The method offers single product selectivity, high yield (up to 92%), high purity, and excellent atom economy by combining four components in a single reaction vessel.
Q: Which catalyst system provides the best results?
A: Cuprous iodide (CuI) combined with tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine (TBTA) as a ligand demonstrates superior catalytic activity and yield compared to other copper salts.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes readily available raw materials, mild reaction conditions, and simple workup procedures like extraction and crystallization, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Oxazine-1,3-Oxazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed one-pot synthesis for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented methodology from the laboratory bench to full-scale commercial production. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot runs to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and purity at every stage of the process.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. By partnering with us, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in your drug development pipeline.
