Advanced Synthesis of Dialkyl Phosphinates: A Commercial Breakthrough for Flame Retardants and Extractants
Advanced Synthesis of Dialkyl Phosphinates: A Commercial Breakthrough for Flame Retardants and Extractants
The global demand for high-performance organophosphorus compounds, particularly dialkyl phosphinates and their metal salts, has surged due to their critical role as halogen-free flame retardants and efficient rare earth extractants. A pivotal advancement in this sector is detailed in patent CN112321638A, which discloses a novel, mild, and highly efficient preparation method for dialkyl monoalkylphosphinates. This technology represents a significant departure from traditional synthetic routes, addressing long-standing challenges related to reaction severity, cost, and product purity. By utilizing a silicon-hydrogen containing reducing agent to transform dialkyl monoalkyl phosphonates, this process offers a robust pathway for producing high-value intermediates. For R&D directors and procurement strategists, understanding this shift is essential for optimizing supply chains and reducing the total cost of ownership in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dialkyl phosphinic acids and their derivatives has relied heavily on three primary methodologies, each fraught with significant industrial drawbacks. The Friedel-Crafts reaction, while conceptually simple, suffers from low efficiency and generates substantial amounts of aluminum chloride waste, complicating post-treatment and environmental compliance. Alternatively, the Grignard reagent method, though capable of producing pure products, imposes severe operational constraints; it requires strictly anhydrous and anaerobic conditions, expensive magnesium activation, and poses significant safety risks during scale-up. Furthermore, the radical addition method, often employed for industrial production, necessitates high-temperature and high-pressure reactors to handle gaseous olefins like ethylene. This approach frequently leads to the formation of unwanted monoalkylated by-products and oligomers, which degrade the thermal stability of the final flame retardant and reduce extraction efficiency in hydrometallurgical applications.
The Novel Approach
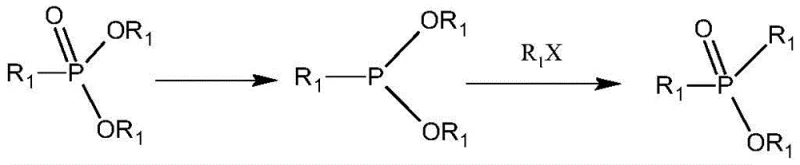
In stark contrast, the methodology outlined in patent CN112321638A introduces a transformative strategy that circumvents these traditional bottlenecks. By employing a silicon-hydrogen containing reducing agent, such as phenylsilane or trichlorosilane, the process converts dialkyl monoalkyl phosphonates into dialkyl monoalkyl phosphinites under remarkably mild conditions. This reduction step avoids the extreme pressures of radical chemistry and the moisture sensitivity of Grignard reagents. Subsequent catalytic isomerization allows for the precise rearrangement of the phosphorus skeleton to yield the target dialkyl phosphonates with exceptional selectivity. This two-step sequence not only simplifies the reaction engineering but also drastically reduces the formation of impurities, ensuring a product profile that meets the stringent specifications required for high-end electronic and pharmaceutical applications.
Mechanistic Insights into Silicon-Hydrogen Reduction and Isomerization
The core of this technological breakthrough lies in the selective reduction of the phosphoryl bond using silane chemistry. Unlike traditional hydride reducers that might lack selectivity or require cryogenic conditions, the silicon-hydrogen reducing agents utilized in this patent facilitate a controlled deoxygenation of the phosphonate starting material. The reaction proceeds through a coordinated mechanism where the silicon center activates the phosphorus-oxygen double bond, allowing for the insertion of hydrogen and the formation of a trivalent phosphorus intermediate (phosphinite). This step is crucial as it sets the stage for the subsequent skeletal rearrangement. The use of organic amine catalysts, such as triethylamine, further enhances this process by forming transient complexes with the silane, lowering the activation energy and enabling the reaction to proceed efficiently at temperatures ranging from 70°C to 120°C.

Following the reduction, the process leverages a catalytic isomerization to reconstruct the phosphorus valency. The dialkyl monoalkyl phosphinite intermediate undergoes a rearrangement in the presence of a halogenated hydrocarbon catalyst, such as methyl iodide or bromo-isooctane. This step effectively migrates an alkyl group from the oxygen to the phosphorus atom, regenerating the pentavalent state but with a different substitution pattern characteristic of phosphinates. This isomerization is thermodynamically driven and occurs smoothly at moderate temperatures (100-150°C), avoiding the thermal degradation often seen in high-pressure radical processes. The result is a clean conversion to the desired dialkyl alkyl phosphonate structure, which can then be easily hydrolyzed to the free acid or converted into metal salts.
How to Synthesize Dialkyl Phosphinates Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing fine chemical manufacturing facilities. The process begins with the careful addition of the phosphonate substrate to the reducing agent mixture under an inert atmosphere, ensuring safety and reproducibility. Following the reduction and distillation of the intermediate, the isomerization step requires precise temperature control to maximize yield while minimizing side reactions. The final hydrolysis and salt formation steps are straightforward aqueous workups that leverage standard filtration and drying equipment. For a detailed breakdown of the specific reaction parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- React dialkyl monoalkyl phosphonate with a silicon-hydrogen containing reducing agent (e.g., phenylsilane or trichlorosilane) under inert gas protection at mild temperatures (0-30°C initial, heating to 70-120°C).
- Perform catalytic isomerization of the resulting dialkyl monoalkyl phosphinite using a halogenated hydrocarbon catalyst at elevated temperatures (100-150°C) to form the dialkyl alkyl phosphonate.
- Hydrolyze the ester under acidic conditions and react with metal salts (e.g., Aluminum, Zinc) via hydrothermal reaction to obtain the final metal dialkyl phosphinate salts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silicon-hydrogen reduction technology offers compelling economic and logistical benefits. By eliminating the dependency on volatile raw materials like ethylene gas and expensive Grignard reagents, manufacturers can achieve significant cost reduction in flame retardant manufacturing. The process operates at near-atmospheric pressures and moderate temperatures, which reduces the capital expenditure required for specialized high-pressure reactors and enhances overall plant safety. Furthermore, the simplified post-treatment workflow minimizes solvent usage and waste generation, aligning with increasingly strict environmental regulations and reducing the burden on wastewater treatment facilities.
- Cost Reduction in Manufacturing: The elimination of high-cost Grignard reagents and the avoidance of energy-intensive high-pressure radical reactions lead to substantial savings in raw material and utility costs. The mild reaction conditions also extend the lifespan of reactor vessels and reduce maintenance downtime, contributing to a lower total cost of production per kilogram of active ingredient.
- Enhanced Supply Chain Reliability: By utilizing readily available silane reducing agents and stable phosphonate precursors, this method mitigates the supply risks associated with specialized organometallic reagents. The robustness of the process ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed reactions or difficult purifications, thereby securing a steady flow of high-purity organophosphorus intermediates.
- Scalability and Environmental Compliance: The absence of hazardous high-pressure gases and the reduction of heavy metal catalyst residues simplify the scale-up process from pilot to commercial tonnage. The cleaner reaction profile results in less hazardous waste, facilitating easier compliance with environmental discharge standards and reducing the costs associated with waste disposal and regulatory reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel phosphinate synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: How does the Si-H reduction method improve upon traditional Grignard synthesis?
A: The Si-H reduction method eliminates the need for expensive, moisture-sensitive Grignard reagents and harsh anhydrous conditions. It operates under milder temperatures and avoids the complex post-treatment associated with aluminum chloride catalysts, significantly lowering production costs and improving operational safety.
Q: What is the purity level achievable with this new phosphinate synthesis route?
A: The patented process achieves product purity levels as high as 98-99% without requiring additional purification steps. This high purity is critical for applications in rare earth extraction and high-performance flame retardants where impurity profiles directly impact performance.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for easy industrialization. It avoids high-pressure reactors required for radical addition methods and uses readily available raw materials. The mild reaction conditions and simple distillation workup make it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dialkyl Phosphinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Si-H reduction pathway for producing next-generation flame retardants and extractants. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to market-ready product is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of organophosphorus chemistry, maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical and agrochemical intermediates.
We invite you to collaborate with us to optimize your supply chain for dialkyl phosphinates and related derivatives. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global market for high-performance fine chemicals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
