Advanced Phase-Transfer Catalysis for High-Purity Anti-Inflammatory Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for active pharmaceutical ingredients (APIs) and their key precursors, particularly for the class of non-steroidal anti-inflammatory drugs (NSAIDs). Patent CN104230C discloses a significant technological breakthrough in the synthesis of 2-(4-R-aminophenyl) propionic acid derivatives, which serve as critical intermediates for potent analgesic and antiphlogistic agents. This innovation addresses long-standing challenges in organic synthesis regarding the selective monoalkylation of aromatic amines, a transformation that is notoriously difficult to control due to the increased nucleophilicity of the product compared to the starting material. By leveraging a novel phase-transfer catalysis (PTC) system in a heterogeneous aqueous-organic medium, this method achieves high selectivity and yield without the need for cumbersome protection-deprotection sequences. For R&D directors and process chemists, this patent represents a pivotal shift towards more atom-economical and environmentally benign manufacturing protocols for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of amino-substituted phenylpropionic acid derivatives has been fraught with significant chemical and economic inefficiencies, as highlighted in prior art such as United States Patent 3957850. The fundamental issue lies in the inherent reactivity of the primary amine group; once a single alkyl group is introduced, the resulting secondary amine often becomes more nucleophilic than the original substrate, leading to rapid and uncontrolled dialkylation. This side reaction generates NN-dialkylated byproducts which are not only structurally distinct but often possess drastically reduced pharmacological activity or increased toxicity profiles compared to the desired monoalkylated target. Furthermore, separating these closely related dialkyl impurities from the desired monoalkyl product typically requires complex and costly purification steps, such as repeated recrystallizations or preparative chromatography, which severely impact the overall process yield and commercial viability. The necessity to employ protecting groups to mask the amine functionality during alkylation adds additional synthetic steps, increasing raw material consumption, waste generation, and production time, thereby creating a substantial burden on the manufacturing supply chain.
The Novel Approach
In stark contrast to these traditional limitations, the method described in CN104230C introduces a streamlined, one-pot heterogeneous reaction system that effectively suppresses dialkylation while promoting high-yield monoalkylation. The core of this innovation involves dissolving the 2-(4-aminophenyl) propionic acid derivative, represented structurally in the following diagram, in a non-polar, water-immiscible solvent such as toluene. Under the influence of a specialized phase-transfer catalyst and an aqueous alkaline reagent, the substrate reacts with an alkylating agent in a biphasic system. This unique environment modulates the reactivity of the amine, allowing for precise control over the degree of substitution. The process eliminates the need for temporary protecting groups, as the reaction conditions inherently favor the formation of the monoalkylated product, which remains in the organic phase for easy recovery. This approach not only simplifies the synthetic route but also significantly enhances the purity profile of the crude product, reducing the downstream purification burden and aligning perfectly with the goals of green chemistry and cost-effective manufacturing.
Under the influence of a specialized phase-transfer catalyst and an aqueous alkaline reagent, the substrate reacts with an alkylating agent in a biphasic system. This unique environment modulates the reactivity of the amine, allowing for precise control over the degree of substitution. The process eliminates the need for temporary protecting groups, as the reaction conditions inherently favor the formation of the monoalkylated product, which remains in the organic phase for easy recovery. This approach not only simplifies the synthetic route but also significantly enhances the purity profile of the crude product, reducing the downstream purification burden and aligning perfectly with the goals of green chemistry and cost-effective manufacturing.
Mechanistic Insights into Phase-Transfer Catalyzed Alkylation
The success of this synthetic strategy relies heavily on the intricate mechanistic role played by the phase-transfer catalyst, typically a quaternary ammonium or phosphonium salt such as Aliquat 336 (methyl tricaprylammonium chloride). In this heterogeneous system, the catalyst functions as a molecular shuttle, transporting reactive anionic species across the interface between the aqueous and organic phases. Specifically, the catalyst facilitates the transfer of hydroxide ions or the deprotonated amine species into the organic layer where the alkylating agent (e.g., methallyl chloride) is dissolved. This interfacial activation ensures that the reaction proceeds rapidly at moderate temperatures, typically between 80°C and 85°C, avoiding the thermal degradation often associated with harsher homogeneous conditions. The choice of catalyst is critical; experimental data within the patent indicates that different quaternary salts (such as TEBA or tetrabutylammonium bromide) can influence the reaction rate and selectivity, with Aliquat 336 demonstrating superior performance in maintaining the integrity of the amino acid backbone while driving the alkylation to completion.
Furthermore, the mechanism provides a built-in safeguard against impurity formation through careful stoichiometric control and phase behavior. By maintaining a specific molar ratio of alkylating agent to substrate (approximately 1:1.1 to 1:1.4), the system limits the availability of the electrophile once the monoalkylation event has occurred. Additionally, the product, 2-(4-alkylaminophenyl) propionic acid, exhibits favorable partitioning behavior, remaining largely in the organic phase as the free acid or a manageable salt form, rather than forming insoluble precipitates that could trap impurities. The final product structure, shown below, highlights the successful introduction of the alkyl group (R) at the para-position of the phenyl ring.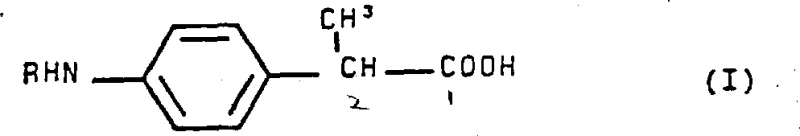 Post-reaction workup involves a straightforward acidification and crystallization sequence, which leverages the pH-dependent solubility of the amino acid derivative to isolate the high-purity target compound, effectively leaving behind any minor dialkylated byproducts in the mother liquor or aqueous washings.
Post-reaction workup involves a straightforward acidification and crystallization sequence, which leverages the pH-dependent solubility of the amino acid derivative to isolate the high-purity target compound, effectively leaving behind any minor dialkylated byproducts in the mother liquor or aqueous washings.
How to Synthesize 2-(4-Alkylaminophenyl) Propionic Acid Efficiently
Implementing this patented methodology requires strict adherence to the specified reaction parameters to ensure optimal selectivity and yield. The process begins with the preparation of a biphasic mixture containing the amino acid substrate, a water-immiscible solvent like toluene, and a catalytic amount of a quaternary ammonium salt. An aqueous solution of sodium hydroxide is then introduced to generate the reactive nucleophile in situ. The addition of the alkylating agent, such as methallyl chloride, is carefully controlled under an inert nitrogen atmosphere to prevent oxidation and manage the exotherm. Heating the mixture to 80-85°C for a duration of 2 to 4 hours drives the reaction to completion, after which the phases are separated. The detailed standardized operating procedures, including specific mixing rates, temperature ramping profiles, and crystallization protocols required for GMP compliance, are outlined in the technical guide below.
- Dissolve 2-(4-aminophenyl) propionic acid or its ester in a water-immiscible non-polar solvent such as toluene.
- Add a phase-transfer catalyst (e.g., Aliquat 336) and an aqueous alkaline reagent (e.g., sodium hydroxide) to the mixture.
- React with an alkylating agent (e.g., methallyl chloride) at 80-85°C under nitrogen, then separate and purify the product via acidification and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phase-transfer catalysis technology offers transformative advantages in terms of cost structure and operational reliability. The elimination of protection and deprotection steps fundamentally reshapes the cost model by removing the need for expensive protecting group reagents and the associated waste disposal costs. This streamlining of the synthetic route directly translates to a significant reduction in raw material expenditure and a decrease in the overall processing time per batch. Moreover, the use of commodity chemicals such as toluene, sodium hydroxide, and methallyl chloride ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce reagents. The robustness of the process also implies fewer batch failures and a more consistent output quality, which is crucial for maintaining uninterrupted production schedules in a high-demand pharmaceutical market.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the synthetic pathway. By avoiding the use of protecting groups, manufacturers save substantially on reagent costs and reduce the number of unit operations required, such as filtration and drying steps associated with intermediate isolation. The high selectivity of the reaction minimizes the formation of hard-to-remove impurities, thereby reducing the consumption of solvents and adsorbents during the purification phase. This lean manufacturing approach results in a lower cost of goods sold (COGS) and improved margin potential for the final API, making it a highly attractive option for generic drug producers seeking competitive pricing advantages.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk-grade chemicals enhances the security of supply for this intermediate. Unlike processes that depend on custom-synthesized catalysts or unstable reagents, this method utilizes standard industrial inputs that can be sourced from multiple global suppliers, reducing the risk of single-source bottlenecks. The reaction conditions are mild and do not require specialized high-pressure or cryogenic equipment, allowing for production in a broader range of manufacturing facilities. This flexibility ensures that production capacity can be easily scaled or shifted between sites to meet fluctuating market demands without significant capital investment or requalification efforts.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the heterogeneous nature of the reaction facilitates easier handling and safer scale-up. The use of aqueous alkali reduces the need for strong, hazardous bases in organic solvents, lowering the risk of exothermic runaways. Furthermore, the simplified workup generates less hazardous waste compared to traditional methods involving heavy metal catalysts or complex organic transformations. The ability to recover and recycle the organic solvent and potentially the phase-transfer catalyst adds another layer of sustainability, aligning with increasingly stringent global environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: How does this PTC method prevent the formation of inactive dialkylated byproducts?
A: The method utilizes a heterogeneous phase system with precise stoichiometric control (1:1.1 to 1:1.4 molar ratio) and specific phase-transfer catalysts like Aliquat 336. This environment favors monoalkylation kinetics while minimizing the reactivity of the secondary amine towards further alkylation, significantly reducing the formation of NN-dialkyl derivatives that plague conventional methods.
Q: What are the primary advantages of using Aliquat 336 over traditional alkylation catalysts?
A: Aliquat 336 (methyl tricaprylammonium chloride) offers superior solubility in both organic and aqueous phases, facilitating efficient ion transport across the interface. This allows the reaction to proceed under milder conditions (80-85°C) compared to harsh traditional methods, resulting in higher selectivity, easier product isolation, and reduced thermal degradation of the sensitive amino acid backbone.
Q: Is this process scalable for industrial production of anti-inflammatory drug intermediates?
A: Yes, the process is highly scalable. It employs common industrial solvents like toluene and readily available reagents such as sodium hydroxide and methallyl chloride. The workup involves standard unit operations like phase separation, washing, and crystallization, making it ideal for large-scale manufacturing without requiring complex chromatography or exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Alkylaminophenyl) Propionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and manufacture of life-saving anti-inflammatory medications. Our team of expert process chemists has extensively analyzed the technology disclosed in CN104230C and possesses the technical capability to adapt and optimize this phase-transfer catalysis route for commercial production. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-(4-alkylaminophenyl) propionic acid meets the highest industry standards for impurity profiles and physical properties.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can drive efficiency and value for your organization.
