Advanced Synthesis of Coumarin-Based Beta-Diketone Boron Fluoride Fluorescent Dyes for Commercial Scale-Up
Advanced Synthesis of Coumarin-Based Beta-Diketone Boron Fluoride Fluorescent Dyes for Commercial Scale-Up
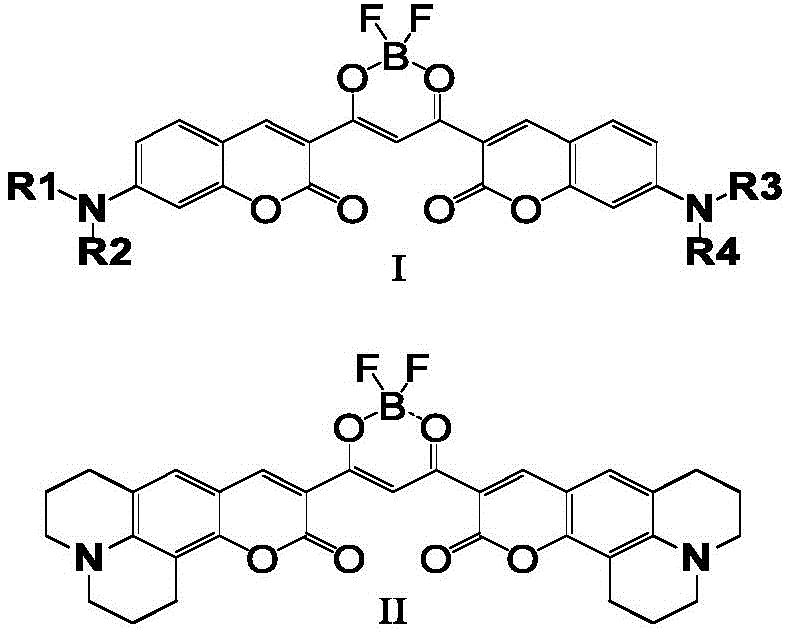
The rapid evolution of the optoelectronic and biotechnology sectors demands materials that offer superior stability and tunable photophysical properties. Patent CN110105381B introduces a groundbreaking methodology for synthesizing a class of beta-diketone boron fluoride fluorescent dyes utilizing a coumarin skeleton. This innovation addresses critical limitations found in traditional organic luminescent compounds, specifically targeting the need for long-wavelength emission and enhanced Stokes shifts. By strategically modifying the coumarin framework through Claisen condensation and subsequent boron coordination, this technology enables the production of dyes with narrow absorption and emission spectra. For R&D directors and procurement specialists seeking reliable OLED material supplier partnerships, this patent represents a significant leap forward in designing near-infrared fluorescent probes and organic light-emitting diodes. The structural versatility allows for extensive derivatization, ensuring that these materials can be adapted for diverse applications ranging from cell imaging to advanced photoelectric devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional coumarin dyes have long been valued for their high fluorescence quantum yields and biocompatibility; however, their utility is often restricted by inherent photophysical constraints. A primary drawback of substituent-free coumarins is their exceptionally small Stokes displacement, which limits their effectiveness in applications requiring distinct separation between excitation and emission signals. Furthermore, achieving a red shift in absorption and emission typically requires complex molecular engineering that can compromise synthetic feasibility. Conventional methods often struggle to extend the conjugated chain length sufficiently without introducing instability or toxicity. For supply chain heads managing the commercial scale-up of complex polymer additives or electronic chemicals, these limitations translate into higher costs and inconsistent batch quality. The inability to easily tune the push-pull electron structure in standard coumarin derivatives means that manufacturers are frequently locked into suboptimal performance profiles, hindering the development of next-generation biosensors and organic probes.
The Novel Approach
The methodology disclosed in the patent overcomes these hurdles by employing a strategic two-step synthesis that fundamentally alters the electronic landscape of the coumarin molecule. By first preparing a series of coumarins with substituents introduced at the 3-position, the process establishes a foundation for extended conjugation. The subsequent Claisen condensation between the coumarin-containing ester and a corresponding carbonyl compound generates a beta-diketone acid ligand, effectively lengthening the conjugated system. Finally, the coordination of this ligand with boron trifluoride creates an electron-deficient center that dramatically enhances the system's push-pull electron capability. This novel approach not only achieves a significant red shift in emission wavelength but also ensures stable optical properties essential for rigorous industrial applications. For procurement managers focused on cost reduction in electronic chemical manufacturing, this route is particularly attractive due to its reliance on simple, high-yield reactions that utilize commercially accessible starting materials.
Mechanistic Insights into Claisen Condensation and Boron Coordination
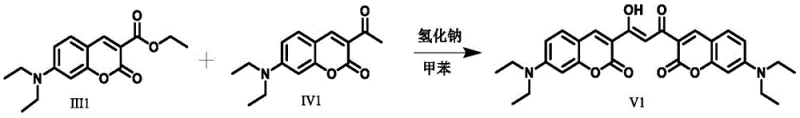
The core of this synthetic strategy lies in the precise execution of the Claisen condensation reaction, which serves as the bridge between simple precursors and complex functional dyes. In the presence of a strong base such as sodium hydride, the alpha-hydrogen of the carbonyl compound is deprotonated to form a nucleophilic enolate. This enolate then attacks the ester carbonyl of the coumarin derivative, leading to the formation of a beta-keto ester intermediate which subsequently eliminates an alkoxide to form the beta-diketone ligand. This step is crucial as it extends the pi-conjugation system across the molecule, directly influencing the energy gap between the highest occupied molecular orbital and the lowest unoccupied molecular orbital. The reaction conditions, typically involving reflux in non-polar solvents like toluene, ensure high conversion rates while minimizing side reactions. Understanding this mechanism allows R&D teams to optimize reaction parameters such as molar ratios and temperature to maximize the yield of the intermediate ligand, which is the precursor to the final high-performance dye.
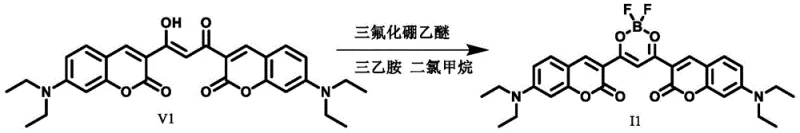
Following the formation of the beta-diketone ligand, the final and most critical transformation involves the coordination with boron trifluoride. The beta-diketone acts as a bidentate ligand, chelating the boron atom to form a rigid, planar six-membered ring structure. This coordination introduces a strong electron-withdrawing effect due to the electron-deficient nature of the boron difluoride group. When combined with the electron-donating amino groups typically present on the coumarin skeleton, a powerful intramolecular charge transfer (ICT) system is established. This push-pull architecture is responsible for the observed red shift in both absorption and emission wavelengths, pushing the dye's performance into the near-infrared region. The rigidity imparted by the boron coordination also reduces non-radiative decay pathways, thereby enhancing the fluorescence quantum yield. For technical teams evaluating the feasibility of scaling this process, the use of mild Lewis acids like boron trifluoride etherate in dichloromethane ensures that the reaction proceeds smoothly under controlled conditions without requiring extreme pressures or temperatures.
How to Synthesize Coumarin-Based Beta-Diketone Boron Fluoride Dyes Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these advanced fluorescent materials with high reproducibility. The process begins with the preparation of the beta-diketone intermediate through the condensation of specific coumarin esters and ketones, followed by the complexation step with boron reagents. This streamlined approach minimizes the number of purification steps required, which is a key factor in maintaining cost efficiency during production. Detailed standardized synthesis steps see the guide below for specific reaction conditions and workup procedures tailored for laboratory and pilot-scale operations.
- Perform Claisen condensation between a coumarin-containing ester and a corresponding carbonyl compound using sodium hydride in toluene to form the beta-diketone acid ligand intermediate.
- React the resulting beta-diketone intermediate with boron trifluoride diethyl etherate in dichloromethane in the presence of triethylamine.
- Purify the final boron-coordinated fluorescent dye product through extraction, drying, and recrystallization to achieve high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations tasked with securing a reliable supply of high-purity OLED material, the economic and logistical benefits of this patented synthesis route are substantial. The process is designed to utilize raw materials that are cheap and easily available on the global market, reducing dependency on exotic or supply-constrained reagents. By eliminating the need for expensive transition metal catalysts often found in cross-coupling reactions, the method significantly lowers the overall cost of goods sold. Furthermore, the simplicity of the reaction conditions means that existing chemical infrastructure can be adapted for production with minimal capital expenditure. This accessibility translates directly into reduced lead times for high-purity electronic chemicals, allowing manufacturers to respond more agilely to market demands. The high yields reported in the experimental examples suggest that waste generation is minimized, aligning with modern environmental compliance standards and reducing the burden on waste treatment facilities.
- Cost Reduction in Manufacturing: The synthetic route avoids the use of precious metal catalysts such as palladium or platinum, which are subject to volatile pricing and supply chain disruptions. Instead, the process relies on abundant bases like sodium hydride and triethylamine, alongside common solvents like toluene and dichloromethane. This substitution drastically simplifies the bill of materials and removes the need for costly metal scavenging steps during purification. Consequently, the overall production cost is significantly lowered, making the final fluorescent dyes more competitive in price-sensitive markets like consumer electronics and disposable diagnostic sensors.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted coumarins and simple ketones, are commodity chemicals with established global supply chains. This ensures that production schedules are not jeopardized by the scarcity of niche intermediates. Additionally, the robustness of the Claisen condensation and boron coordination steps means that the process is tolerant to minor variations in raw material quality, further stabilizing the supply output. For supply chain heads, this reliability is paramount for maintaining continuous operation of downstream assembly lines that depend on consistent deliveries of optoelectronic components.
- Scalability and Environmental Compliance: The reaction conditions described, such as refluxing in toluene or stirring in dichloromethane at room temperature, are inherently scalable from gram to ton quantities without requiring specialized high-pressure equipment. The absence of heavy metals in the final product simplifies regulatory compliance for applications in biological imaging and medical devices. Moreover, the high atom economy of the condensation reaction reduces the volume of chemical waste generated per kilogram of product. This environmental efficiency not only lowers disposal costs but also supports corporate sustainability goals, making the technology attractive for green chemistry initiatives within the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorescent dye technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners.
Q: What are the primary optical advantages of these coumarin-based boron fluoride dyes?
A: Unlike traditional coumarins which suffer from small Stokes shifts, these novel dyes exhibit long absorption and emission wavelengths with narrow spectra. The introduction of the electron-deficient boron group significantly enhances the push-pull electron capability, resulting in stable optical properties suitable for bioimaging and OLED applications.
Q: How does the synthesis route ensure scalability for industrial production?
A: The synthesis relies on robust Claisen condensation reactions using readily available raw materials like coumarin esters and ketones. The process avoids complex transition metal catalysts, utilizing common bases like sodium hydride and triethylamine, which simplifies downstream processing and waste treatment for large-scale manufacturing.
Q: Can the emission wavelength be tuned for specific electronic applications?
A: Yes, the patent describes a modular approach where substituents at the 3-position of the coumarin skeleton can be varied. By extending the conjugated chain length and modifying the electron-donating groups, manufacturers can precisely tune the emission wavelength into the near-infrared region for specialized optoelectronic devices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin-Based Boron Fluoride Dyes Supplier
As the demand for advanced optoelectronic materials continues to surge, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of fluorescent dye meets the exacting standards required for OLED displays and biological probes. We understand the critical nature of supply continuity in the high-tech sector and are committed to delivering high-purity OLED material that drives your product performance forward.
We invite you to collaborate with us to explore the full potential of these coumarin-based dyes for your specific applications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a competitive advantage through superior material science and reliable manufacturing excellence.
