Advanced Synthesis of Near-Infrared Coumarin Boron Difluoride Complexes for High-Performance Optoelectronics
Advanced Synthesis of Near-Infrared Coumarin Boron Difluoride Complexes for High-Performance Optoelectronics
The landscape of organic fluorescent materials is undergoing a significant transformation driven by the demand for near-infrared (NIR) emitters with superior photophysical properties. Patent CN110143977B introduces a groundbreaking class of coumarin heteroboron difluoride complexes that address the critical limitations of traditional fluorophores. By strategically integrating an electron-deficient boron difluoride-beta-dicarbonyl six-membered ring at the 3-position of a 7-diethylaminocoumarin scaffold, this technology creates a robust push-pull electronic system. This structural innovation not only extends the conjugated chain length but also dramatically shifts absorption and emission wavelengths into the desirable red and near-infrared regions. For R&D directors and procurement specialists in the optoelectronic and biomedical sectors, this represents a pivotal advancement in designing stable, high-quantum-yield fluorescent probes.
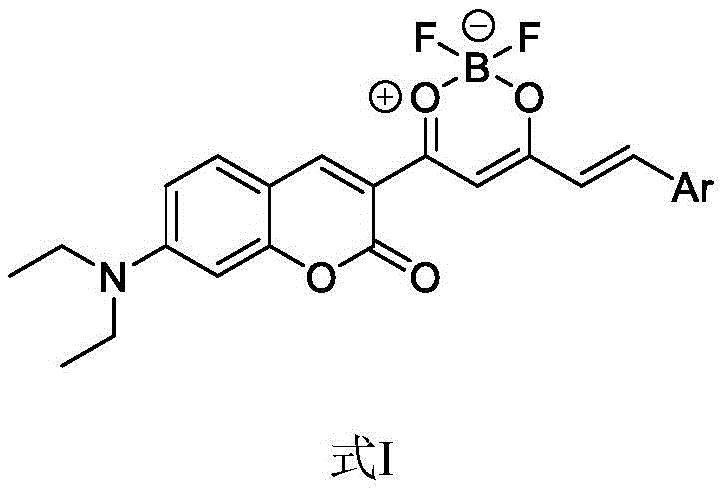
The core innovation lies in the specific molecular architecture depicted in the patent, where the boron difluoride moiety acts as a potent electron-withdrawing group. This configuration effectively overcomes the short emission wavelength limitations (typically 400-520 nm) inherent to standard coumarin dyes. Furthermore, unlike conventional BODIPY dyes which often suffer from small Stokes shifts leading to self-excitation issues, these novel complexes exhibit large Stokes shifts. This characteristic is paramount for applications requiring high signal-to-noise ratios, such as real-time monitoring in living cells and tissues. The versatility of the structure allows for further tuning by varying the aromatic aldehyde component, enabling a broad spectrum of optical properties tailored to specific device requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing near-infrared fluorescent dyes often rely on complex cyanine structures or heavily modified BODIPY cores that present significant manufacturing challenges. Conventional coumarin derivatives, while chemically stable, are fundamentally restricted by their electronic structure, limiting their utility in deep-tissue imaging or long-wavelength optoelectronic devices. Attempts to red-shift their emission frequently involve extending conjugation through unstable linkers or introducing substituents that compromise photostability. Additionally, many existing NIR dyes suffer from aggregation-caused quenching or require rigorous purification steps to remove toxic heavy metal catalysts used in cross-coupling reactions. These factors collectively increase the cost of goods sold and introduce supply chain vulnerabilities for manufacturers of high-purity electronic chemicals and biological reagents.
The Novel Approach
The methodology outlined in CN110143977B offers a streamlined alternative that bypasses these historical bottlenecks. By leveraging a Knoevenagel condensation reaction between a pre-formed boron-complexed coumarin intermediate and various aromatic aldehydes, the synthesis achieves high yields under mild conditions. This approach eliminates the need for expensive palladium or other transition metal catalysts, thereby simplifying the downstream purification process and reducing the risk of metal contamination in the final product. The use of readily available starting materials, such as 7-diethylaminocoumarin and substituted benzaldehydes, ensures a reliable supply chain foundation. Moreover, the resulting compounds demonstrate exceptional quantum yields and thermal stability, making them ideal candidates for mass production in the competitive display and sensor markets.
Mechanistic Insights into Boron Complexation and Knoevenagel Condensation
The synthesis mechanism is a sophisticated interplay of Lewis acid-base chemistry and carbon-carbon bond formation. Initially, the 7-diethylaminocoumarin reacts with boron trifluoride diethyl etherate to form a stable six-membered chelate ring. This step is crucial as it rigidifies the molecular framework, reducing non-radiative decay pathways and enhancing fluorescence efficiency. The methyl group on this newly formed ring is activated by the strong electron-withdrawing effect of the boron center, rendering the alpha-protons highly acidic. This activation is the key driver for the subsequent condensation step, allowing the intermediate to react efficiently with a wide range of aromatic aldehydes.
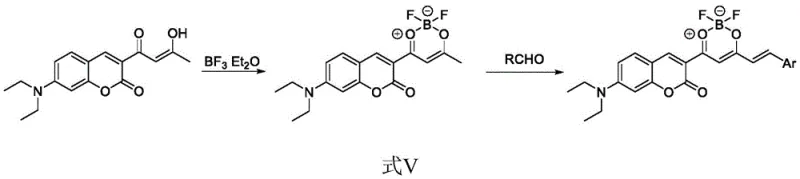
In the second stage, the activated methyl group undergoes Knoevenagel condensation with aromatic aldehydes in the presence of a mild base like piperidine or triethylamine. This reaction extends the pi-conjugation system across the molecule, which is directly responsible for the observed bathochromic shift in absorption and emission spectra. The patent data indicates that this process tolerates a diverse array of substituents on the aromatic aldehyde, including electron-donating groups like methoxy and dimethylamino, as well as electron-withdrawing groups like chloro and hydroxy. This tolerance allows for fine-tuning of the HOMO-LUMO gap, providing chemists with a powerful toolkit to engineer specific optical properties without altering the core synthetic strategy. The result is a series of structurally diverse dyes with consistent high performance.
How to Synthesize Coumarin Boron Difluoride Complex Efficiently
The practical execution of this synthesis is designed for operational simplicity and reproducibility, making it highly attractive for process chemistry teams looking to scale up production. The protocol involves dissolving the coumarin precursor in a solvent such as dichloromethane or acetic anhydride, followed by the addition of the boron source at controlled temperatures. Once the intermediate complex is isolated, it is subjected to reflux in ethanol with the chosen aldehyde and base catalyst. The reaction progress is easily monitored via TLC, and the final products typically precipitate out of the solution as colored solids, facilitating straightforward isolation by filtration. This operational ease translates directly into reduced labor costs and shorter batch cycle times in a commercial setting.
- React 7-diethylaminocoumarin with boron trifluoride diethyl etherate in an organic solvent like dichloromethane to form the intermediate boron complex.
- Perform a reflux reaction of the intermediate complex with various aromatic aldehydes in ethanol using a base catalyst such as piperidine.
- Isolate the final fluorescent dye product through filtration and washing, achieving high purity suitable for optical applications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical performance. The elimination of precious metal catalysts removes a major cost driver and supply risk associated with fluctuating prices of metals like palladium or iridium. Furthermore, the reliance on commodity chemicals such as aromatic aldehydes and ethanol ensures that raw material sourcing remains stable and cost-effective, even during periods of global supply chain disruption. The high yields reported in the patent examples suggest that waste generation is minimized, aligning with increasingly stringent environmental regulations and reducing disposal costs.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of expensive transition metal catalysts and harsh reaction conditions, which significantly lowers the overall cost of production. By utilizing a simple reflux setup and common solvents, capital expenditure on specialized reactor equipment is minimized. The high conversion rates and straightforward workup procedures reduce the consumption of energy and purification media, leading to substantial operational savings. Additionally, the absence of heavy metals simplifies the quality control workflow, as there is no need for costly and time-consuming metal residue testing, further streamlining the manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, including 7-diethylaminocoumarin and various substituted benzaldehydes, are widely available from multiple global suppliers. This commoditization of raw materials mitigates the risk of single-source dependency and ensures business continuity. The robustness of the reaction conditions means that production can be maintained consistently without sensitive adjustments for minor variations in reagent quality. Consequently, lead times for delivering high-purity fluorescent dye intermediates can be significantly shortened, allowing downstream customers to maintain leaner inventory levels and respond more agilely to market demands.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram or ton-scale commercial production without requiring fundamental changes to the chemistry. The use of ethanol as a primary solvent in the second step is particularly advantageous from an environmental, health, and safety (EHS) perspective, as it is less toxic and easier to recover than many chlorinated solvents. The high atom economy of the Knoevenagel condensation minimizes the generation of hazardous byproducts, simplifying waste treatment protocols. This alignment with green chemistry principles not only reduces regulatory compliance burdens but also enhances the sustainability profile of the final product for eco-conscious end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorescent dye technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating these materials into their product lines. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the key optical advantages of this coumarin boron difluoride complex compared to traditional dyes?
A: The patented structure introduces a push-pull electron system that significantly red-shifts absorption and emission wavelengths into the near-infrared region while maintaining a large Stokes shift, reducing self-excitation interference common in standard BODIPY dyes.
Q: Is the synthesis process scalable for industrial production of fluorescent probes?
A: Yes, the method utilizes simple reflux conditions and commercially available aromatic aldehydes, avoiding expensive transition metal catalysts, which facilitates easy scale-up from laboratory to commercial manufacturing volumes.
Q: How does the structural modification affect the stability of the fluorescent material?
A: The formation of the six-membered boron difluoride ring rigidifies the molecular structure, enhancing photostability and chemical robustness, which is critical for long-term performance in biological imaging and sensor applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin Boron Difluoride Complex Supplier
As the demand for advanced optoelectronic materials continues to surge, partnering with an experienced CDMO is critical for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of fluorescent dye intermediate meets the highest industry standards. We understand the nuances of handling light-sensitive and moisture-sensitive compounds, implementing specialized packaging and storage protocols to preserve product integrity throughout the logistics chain.
We invite you to collaborate with us to optimize your supply chain for next-generation fluorescent materials. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your time-to-market with reliable, high-quality chemical solutions that drive innovation in the fields of bio-imaging and display technologies.
