Advanced Dehydration Strategy for Commercial Scale-up of p-Hydroxyphenylacetonitrile Intermediates
Advanced Dehydration Strategy for Commercial Scale-up of p-Hydroxyphenylacetonitrile Intermediates
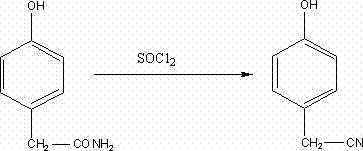
The pharmaceutical industry constantly seeks robust synthetic routes that balance high efficiency with environmental sustainability, particularly for key intermediates like p-hydroxyphenylacetonitrile. Patent CN102180810A introduces a transformative preparation method that shifts the paradigm from complex multi-step nitration sequences to a direct, high-yield dehydration strategy. This technical breakthrough utilizes p-hydroxyphenylacetamide as the starting material, reacting it with thionyl chloride under mild conditions to achieve yields surpassing 95%. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for critical drugs such as Valdecoxib (Bextra) and Atenolol. By adopting this novel pathway, manufacturers can bypass the hazardous and wasteful steps associated with traditional diazotization, ensuring a more reliable and cost-effective source of high-purity pharma intermediates for global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of p-hydroxyphenylacetonitrile has relied heavily on the diazotization of p-aminophenylacetonitrile, a route fraught with significant operational and environmental challenges. This legacy process necessitates a cumbersome sequence involving the nitration of benzyl cyanide, followed by reduction to the amine, and finally diazotization and hydrolysis to introduce the hydroxyl group. Such a multi-step approach not only consumes vast quantities of corrosive reagents like concentrated sulfuric acid and nitric acid but also generates substantial volumes of acidic wastewater, known as "three wastes," which incur high disposal costs. Furthermore, the cumulative yield of this traditional pathway is notoriously poor, often stagnating around 30%, rendering the final product economically vulnerable to fluctuations in raw material prices. The complexity of handling hazardous nitration reactions also poses severe safety risks, complicating regulatory compliance and limiting the scalability required for modern API manufacturing demands.
The Novel Approach
In stark contrast, the innovative method detailed in the patent leverages the direct dehydration of p-hydroxyphenylacetamide using thionyl chloride, offering a streamlined and atom-economical alternative. This approach capitalizes on the improved availability and reduced cost of p-hydroxyphenylacetamide, transforming a simple amide-to-nitrile conversion into a highly efficient industrial process. By operating at mild temperatures between 35-38°C, the reaction minimizes thermal degradation and side reactions, ensuring a clean transformation that is easy to control on a large scale. The simplicity of the workup procedure, involving straightforward neutralization and crystallization, further enhances the operational feasibility, allowing for rapid turnover and reduced cycle times. This strategic shift not only eliminates the need for dangerous nitration steps but also dramatically improves the overall mass balance of the production line.
Mechanistic Insights into Thionyl Chloride-Mediated Amide Dehydration
The core of this synthetic advancement lies in the precise mechanistic interaction between the amide functionality and thionyl chloride, facilitated by a catalytic amount of DMF in a dichloromethane solvent system. The reaction initiates with the nucleophilic attack of the amide oxygen on the sulfur atom of thionyl chloride, forming a reactive imidoyl chloride intermediate while releasing sulfur dioxide and hydrogen chloride gases. The presence of DMF acts as a crucial catalyst, likely forming a Vilsmeier-Haack type active species that accelerates the dehydration rate without requiring excessive heat. This mechanism ensures that the conversion proceeds rapidly within 4 hours, avoiding the prolonged exposure to harsh conditions that typically lead to polymerization or tar formation in nitrile synthesis. Understanding this catalytic cycle is vital for process chemists aiming to replicate the high selectivity and minimal byproduct formation observed in the patent examples.
Impurity control is meticulously managed through a sophisticated workup protocol that targets the removal of acidic residues and unreacted starting materials. Following the reaction, the mixture is quenched with water and carefully neutralized to a specific pH range of 3 to 4 using sodium bicarbonate, a critical step that prevents the hydrolysis of the newly formed nitrile group while precipitating the product. The subsequent recrystallization process, utilizing activated carbon treatment at 85-90°C followed by ethyl acetate extraction, effectively removes colored impurities and trace organic contaminants. This rigorous purification strategy guarantees a final product with an HPLC purity exceeding 96.5% and a sharp melting point of 67-69°C, meeting the stringent quality standards required for downstream pharmaceutical applications where trace impurities can affect drug safety and efficacy.
How to Synthesize p-Hydroxyphenylacetonitrile Efficiently
To implement this high-yield synthesis in a laboratory or pilot plant setting, operators must adhere to strict procedural controls regarding temperature and stoichiometry to maximize the 95.5% yield reported in the patent. The process begins with the preparation of a homogeneous solution of p-hydroxyphenylacetamide in dichloromethane and DMF, followed by the controlled addition of thionyl chloride to maintain the reaction temperature within the narrow 35-38°C window. Detailed standard operating procedures for the quenching, neutralization, and multi-stage recrystallization steps are essential to ensure batch-to-batch consistency and safety. For a comprehensive guide on the exact reagent ratios and equipment setup required for this transformation, please refer to the standardized synthesis protocol outlined below.
- React p-hydroxyphenylacetamide with thionyl chloride in a dichloromethane and DMF solvent system at 35-38°C for 4 hours.
- Quench the reaction with water, neutralize to pH 3-4 using sodium bicarbonate, and isolate the crude product via ice bath crystallization.
- Purify the crude material by decolorizing with activated carbon at 85-90°C, extracting with ethyl acetate, and recrystallizing from water.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this dehydration technology offers profound strategic advantages that extend far beyond simple yield improvements. By transitioning away from the legacy diazotization route, manufacturers can significantly reduce the dependency on volatile and hazardous raw materials like strong mineral acids, thereby stabilizing the supply chain against regulatory crackdowns on hazardous waste disposal. The simplified process flow, which consolidates multiple reaction steps into a single dehydration event, inherently lowers the operational overhead and energy consumption associated with heating, cooling, and separating multiple intermediate stages. This streamlining translates directly into a more resilient supply network capable of responding quickly to market demand surges for key cardiovascular and anti-inflammatory drug precursors without the bottlenecks typical of complex multi-step syntheses.
- Cost Reduction in Manufacturing: The economic impact of this new route is driven primarily by the drastic reduction in processing steps and the elimination of expensive waste treatment protocols associated with nitration chemistry. By achieving a yield that is more than three times higher than the traditional method, the effective cost per kilogram of the active intermediate is substantially lowered, providing a competitive edge in pricing negotiations. Furthermore, the use of readily available solvents like dichloromethane and ethyl acetate, combined with the avoidance of precious metal catalysts or exotic reagents, ensures that the variable costs of production remain predictable and low. This structural cost advantage allows suppliers to offer more attractive pricing models to downstream API manufacturers while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The robustness of the thionyl chloride method significantly mitigates the risk of production stoppages caused by the strict transportation and storage regulations governing nitric and sulfuric acids. Since the reaction operates under mild thermal conditions and utilizes common industrial chemicals, the barrier to entry for qualified contract manufacturing organizations is lower, expanding the pool of potential suppliers and reducing single-source dependency. This diversification capability is crucial for pharmaceutical companies seeking to de-risk their supply chains against geopolitical disruptions or local environmental enforcement actions that frequently target facilities handling large volumes of corrosive acids.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a major leap forward in green chemistry principles by minimizing the generation of acidic effluents and toxic byproducts. The ability to recycle solvents and the reduced volume of waste sludge simplify the permitting process for facility expansion, enabling faster scale-up from pilot batches to multi-ton commercial production. This alignment with increasingly stringent global environmental regulations ensures long-term operational continuity and protects the brand reputation of partners who prioritize sustainable manufacturing practices in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, derived directly from the comparative data and experimental results presented in the patent literature. These insights are designed to clarify the operational benefits and quality assurances associated with the thionyl chloride dehydration method for stakeholders evaluating this technology for integration into their supply networks. Understanding these nuances is essential for making informed decisions about vendor qualification and process validation.
Q: Why is the thionyl chloride method superior to the traditional diazotization route?
A: The traditional diazotization route involves multiple hazardous steps including nitration and reduction, resulting in significant acidic waste and a low overall yield of approximately 30%. In contrast, the thionyl chloride dehydration method described in patent CN102180810A simplifies the process to a single reaction step with a yield exceeding 95%, drastically reducing environmental burden and production costs.
Q: What are the critical quality parameters for this intermediate?
A: High-purity p-hydroxyphenylacetonitrile requires strict control over residual solvents and related impurities. The patented process ensures a content of greater than 96.5% by HPLC and a melting point range of 67-69°C, which are critical specifications for downstream synthesis of APIs like Valdecoxib and Atenolol.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process operates at mild temperatures (35-38°C) and utilizes common industrial solvents like dichloromethane and ethyl acetate. The elimination of harsh nitration conditions and the high yield make it highly scalable and safer for commercial production compared to legacy methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Hydroxyphenylacetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key pharmaceutical intermediates like p-hydroxyphenylacetonitrile to support the global development of life-saving medications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch meets the exacting standards required for API synthesis. We are committed to leveraging advanced synthetic technologies, such as the efficient dehydration route described above, to deliver superior value and reliability to our international partners.
We invite you to collaborate with our technical team to explore how this optimized manufacturing process can enhance your project economics and timeline. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate our capability to serve as your long-term strategic partner in the fine chemical sector.
