Advanced Synthesis of Pyrene Derivative Fluorescent Molecules for Commercial Scale-Up
Advanced Synthesis of Pyrene Derivative Fluorescent Molecules for Commercial Scale-Up
The landscape of fluorescent probe development is undergoing a significant transformation driven by the need for simpler, more scalable synthetic routes that do not compromise on photophysical performance. Patent CN113121566A introduces a groundbreaking methodology for the preparation of pyrene derivative fluorescent molecules, specifically targeting applications in biological imaging and optoelectronic materials. This technology leverages the inherent stability and high fluorescence quantum yield of the pyrene core, addressing the critical industry pain point of complex multi-step syntheses often associated with polycyclic aromatic hydrocarbons (PAHs). By utilizing a direct Michael addition reaction between pyrene-1,6-dione and specific ethylenediamine derivatives, this process achieves a streamlined production workflow that is highly attractive for industrial adoption. The resulting compounds exhibit strong green fluorescence with emission maxima around 530nm and quantum efficiencies reaching up to 29.6%, making them superior candidates for next-generation sensing applications compared to traditional carbon dot methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of force-induced fluorescence color-adjustable materials and similar pyrene-based probes has been plagued by inefficient and labor-intensive protocols. Prior art, such as the method disclosed in patent CN201910950412.4, relies on a cumbersome sequence involving solvent thermal reactions followed by extensive purification steps like dialysis and centrifugation. These traditional approaches not only require specialized equipment like autoclaves and dialysis bags but also suffer from low throughput and difficulty in introducing specific ligands with precise spatial functions. The necessity for subsequent grinding treatments to induce fluorescence color changes further complicates the manufacturing process, introducing variability and potential contamination risks. Furthermore, the reliance on high-temperature solvothermal conditions often leads to broader particle size distributions and inconsistent optical properties, which are unacceptable for high-precision biological imaging or electronic display applications where batch-to-batch consistency is paramount.
The Novel Approach
In stark contrast, the novel approach detailed in CN113121566A simplifies the entire value chain into a single, elegant chemical transformation. The core innovation lies in the exploitation of the electrophilic nature of the ortho-diketone moiety in pyrene-1,6-dione, which undergoes a cascade Michael addition with ethylenediamine derivatives under mild conditions. This one-pot synthesis eliminates the need for harsh thermal treatments and complex post-reaction processing, allowing for the direct isolation of the target fluorescent molecule as a yellow powder. The reaction proceeds efficiently in acetonitrile at moderate temperatures between 35°C and 45°C, significantly lowering energy consumption and operational hazards. By bypassing the dialysis and centrifugation steps entirely, this method drastically reduces the production cycle time and minimizes solvent waste, aligning perfectly with green chemistry principles. The structural integrity of the extended pi-conjugated system is preserved, ensuring the high fluorescence quantum yields necessary for sensitive detection tasks.
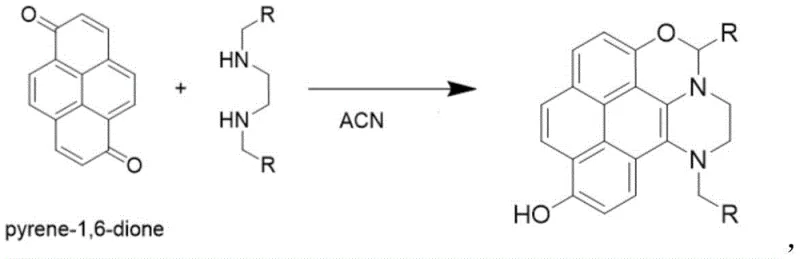
Mechanistic Insights into Michael Addition Cyclization
The chemical elegance of this process stems from the unique reactivity of the pyrene-1,6-dione scaffold. The presence of the ortho-diketone functionality renders the adjacent carbons highly electrophilic, facilitating a nucleophilic attack by the amine groups of the ethylenediamine derivative. This initial attack triggers a cascade cyclization that effectively extends the conjugated system of the pyrene core, a critical factor in red-shifting the absorption and emission wavelengths into the visible spectrum. The resulting quinone-hydroquinone structural motif within the final molecule promotes rapid proton-electron transfer, which enhances electron conductivity and stabilizes the excited state. This mechanistic pathway avoids the formation of random polymeric structures often seen in dopamine-like condensations, instead yielding discrete, well-defined molecular species with predictable photophysical properties. The specific choice of substituents on the ethylenediamine, such as benzyl or pyridyl groups, allows for fine-tuning of the steric environment, which can influence the packing of the molecules in the solid state and their interaction with biological targets.
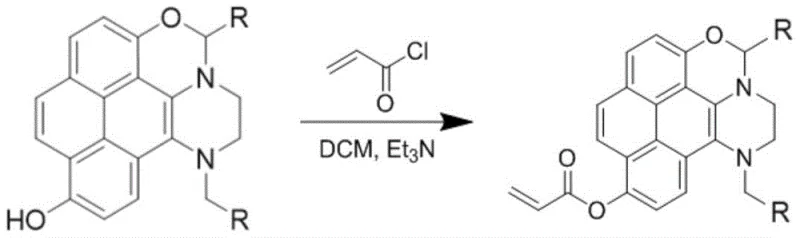
Furthermore, the versatility of this core structure allows for downstream functionalization to create advanced molecular probes. As illustrated in the patent, the phenolic hydroxyl group generated during the cyclization serves as a handle for further chemical modification. Reacting this intermediate with acryloyl chloride in the presence of a base like triethylamine introduces an acrylate moiety, transforming the passive fluorophore into a reactive probe capable of specific analyte binding. This two-stage strategy—first establishing the robust fluorescent core via Michael addition, then tailoring the periphery for specific applications—provides a modular platform for material design. The ability to selectively detect biomolecules like cysteine with a 20-fold fluorescence enhancement demonstrates the efficacy of this mechanistic design, where the binding event likely restricts intramolecular rotation or alters the electron density of the conjugated system, thereby boosting radiative decay rates.
How to Synthesize Pyrene Derivative Fluorescent Molecules Efficiently
The implementation of this synthesis protocol requires careful attention to stoichiometry and purification to maximize the yield of the high-value fluorescent intermediates. The process begins with the dissolution of pyrene-1,6-dione in acetonitrile, followed by the controlled addition of the ethylenediamine derivative. Maintaining the mass ratio between the diketone and the diamine within the range of 1:1.5 to 1:3 is critical; deviations can lead to incomplete conversion or the formation of oligomeric byproducts. The reaction mixture is then stirred at a controlled temperature of 35-45°C for a duration of 12 to 18 hours, allowing the thermodynamic equilibrium to favor the cyclized product. Upon completion, the solvent is removed via rotary evaporation, and the resulting crude solid is subjected to silica gel column chromatography. Using a mobile phase of ethyl acetate and n-hexane (1:1 v/v), the target compound is isolated as a pure yellow powder, ready for characterization or further derivatization into probes like PY-1 or PY-2.
- Dissolve pyrene-1,6-dione in acetonitrile and add ethylenediamine derivative at a mass ratio of 1: 1.5 to 1:3.
- Stir the mixture at 35-45°C for 12-18 hours to complete the Michael addition cyclization.
- Purify the crude yellow powder via silica gel column chromatography using ethyl acetate and n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from multi-step solvothermal processes to this streamlined Michael addition route represents a substantial opportunity for cost optimization and risk mitigation. The elimination of unit operations such as dialysis, centrifugation, and high-pressure thermal treatment directly translates to reduced capital expenditure (CapEx) and lower operational expenditure (OpEx). By removing the need for specialized dialysis membranes and high-speed centrifuges, manufacturers can utilize standard reactor vessels and filtration equipment, thereby simplifying the facility requirements and reducing maintenance overheads. Additionally, the use of common organic solvents like acetonitrile and dichloromethane, which are readily available in bulk quantities from established chemical suppliers, ensures a stable and resilient supply chain free from the bottlenecks often associated with specialty reagents or consumables.
- Cost Reduction in Manufacturing: The simplified workflow inherently drives down manufacturing costs by minimizing labor hours and energy consumption. Since the reaction proceeds at near-ambient temperatures (35-45°C) rather than the high temperatures required for solvothermal synthesis, the energy load on heating systems is drastically reduced. Furthermore, the high selectivity of the Michael addition minimizes the generation of complex impurity profiles, which reduces the burden on quality control laboratories and lowers the cost of goods sold (COGS) associated with extensive analytical testing and reprocessing. The ability to achieve high purity through standard column chromatography rather than complex fractionation techniques further enhances the economic viability of large-scale production.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as pyrene-1,6-dione and simple ethylenediamine derivatives ensures a robust supply chain with multiple sourcing options. Unlike proprietary precursors that may be subject to single-source restrictions or long lead times, these feedstocks are commodity chemicals with established global production networks. This diversity in sourcing mitigates the risk of supply disruptions and allows for flexible inventory management strategies. The short reaction time of 12 to 18 hours also enables a faster turnaround from raw material intake to finished goods, allowing manufacturers to respond more agilely to fluctuating market demands and urgent customer orders without maintaining excessive safety stock.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process offers significant advantages over traditional methods. The absence of heavy metal catalysts or toxic reagents simplifies waste stream management and reduces the cost of effluent treatment. The atom economy of the Michael addition is favorable, meaning less raw material is wasted as byproduct, aligning with increasingly stringent environmental regulations. Scaling this process from laboratory to commercial production is straightforward, as the reaction kinetics are not dependent on difficult-to-control parameters like pressure or supercritical conditions. This ease of scale-up ensures that supply continuity can be maintained as demand grows, supporting the long-term commercialization of devices and assays utilizing these advanced fluorescent materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pyrene derivatives. The answers are derived directly from the experimental data and process parameters outlined in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is crucial for R&D teams evaluating the integration of these fluorophores into new diagnostic kits or electronic components. The robustness of the synthesis and the clarity of the structure-activity relationships provide confidence in the reproducibility of the material properties across different production batches.
Q: What are the key advantages of this pyrene derivative synthesis over traditional carbon dot methods?
A: Unlike traditional methods requiring solvothermal reactions, dialysis, and grinding, this patent utilizes a direct one-step Michael addition. This eliminates complex purification steps like centrifugation and dialysis, significantly reducing processing time and equipment requirements while maintaining high fluorescence quantum yields.
Q: What are the optimal reaction conditions for maximizing yield in this process?
A: The patent specifies an optimal mass ratio of pyrene-1,6-dione to ethylenediamine derivative of 1:2.0. The reaction should be conducted in acetonitrile at a temperature range of 35-45°C for approximately 12 to 18 hours to ensure complete conversion without degrading the sensitive fluorophore structure.
Q: Can these fluorescent molecules be further functionalized for specific biological targets?
A: Yes, the synthesized compounds contain reactive hydroxyl groups that allow for further derivatization. As demonstrated in the patent, reacting the core molecule with acryloyl chloride creates fluorescence-enhanced molecular probes (PY-1/PY-2) capable of selective cysteine detection with a 20-fold increase in fluorescence intensity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrene Derivative Fluorescent Molecule Supplier
As the global demand for high-performance fluorescent probes and optoelectronic materials continues to surge, partnering with an experienced CDMO is essential for navigating the complexities of commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market deployment is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of pyrene chemistry, including rigorous QC labs that enforce stringent purity specifications to guarantee the photophysical consistency of every batch. We understand that in applications like biological imaging and OLED manufacturing, even trace impurities can degrade performance, which is why our quality assurance protocols are designed to exceed industry standards.
We invite you to collaborate with our technical team to explore how this patented synthesis route can be optimized for your specific application needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to validate the feasibility of scaling this Michael addition process, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the rapidly evolving market for advanced fluorescent materials.
