Advanced Manufacturing of High-Optical-Purity S-Escitalopram Intermediates for Global Pharmaceutical Markets
The pharmaceutical industry's demand for high-purity antidepressants has driven significant innovation in chiral synthesis, specifically for S-escitalopram, the active enantiomer of citalopram. Patent CN102796065A introduces a robust manufacturing methodology that addresses critical bottlenecks in producing this vital CNS agent. By leveraging a sophisticated chiral resolution strategy followed by a mild cyclization step, this technology enables the production of S-escitalopram with exceptional optical purity exceeding 99.8% ee. This technical breakthrough is particularly relevant for generic drug manufacturers seeking to optimize their supply chains for high-volume antidepressant production while maintaining rigorous quality standards.
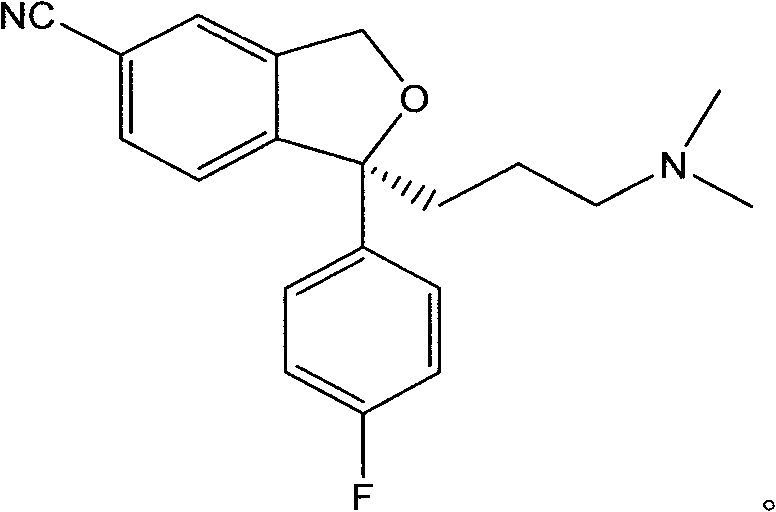
For procurement specialists and R&D directors, understanding the structural integrity of the final product is paramount. The molecule features a characteristic phthalan ring system fused with a fluorophenyl group and a dimethylaminobutyl side chain. Achieving the correct stereochemistry at the C1 position is the defining challenge of this synthesis, as the therapeutic efficacy is entirely dependent on the S-configuration. The patented process ensures that the final API intermediate meets the strict stereochemical specifications required for bioequivalence studies and regulatory filings in major markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of S-escitalopram has been plagued by economic and technical inefficiencies inherent in earlier patent literature. For instance, U.S. Patent 4943590 describes a route utilizing (S)-(+)-α-methoxy-α-trifluoromethylacetyl chloride, commonly known as Mosher's acid chloride. While effective for analytical differentiation, this reagent is prohibitively expensive for industrial scale-up and necessitates complex HPLC separation techniques to isolate the desired isomer. Furthermore, alternative methods such as those disclosed in CN1510024 rely on thionyl chloride for cyclization, a reagent known for its high corrosivity and generation of hazardous acidic byproducts, posing significant safety and environmental compliance challenges for modern manufacturing facilities.
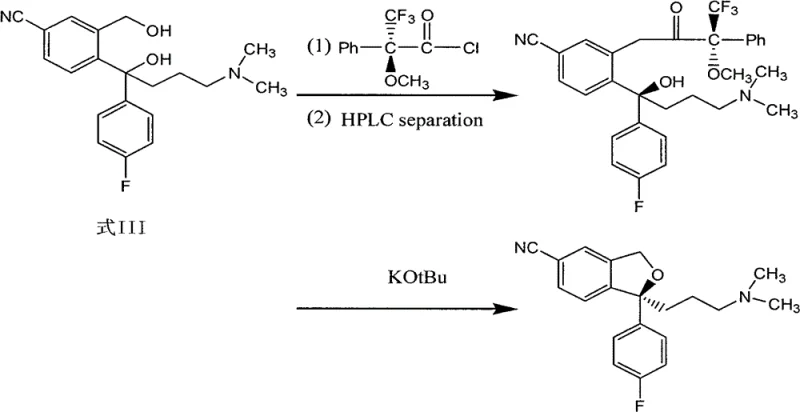
The Novel Approach
In stark contrast to these legacy methods, the process disclosed in CN102796065A offers a streamlined pathway that eliminates the need for costly chiral derivatizing agents and hazardous reagents. The innovation lies in a two-step sequence: first, the resolution of the racemic diol precursor (Formula III) using optically active organic acids like D-(+)-di-p-toluoyl tartaric acid, and second, a gentle cyclization using Tosyl chloride. This approach not only drastically reduces raw material costs but also simplifies the downstream processing by avoiding difficult chromatographic purifications. The result is a chemically elegant solution that transforms a complex chiral synthesis into a manageable crystallization-driven process.
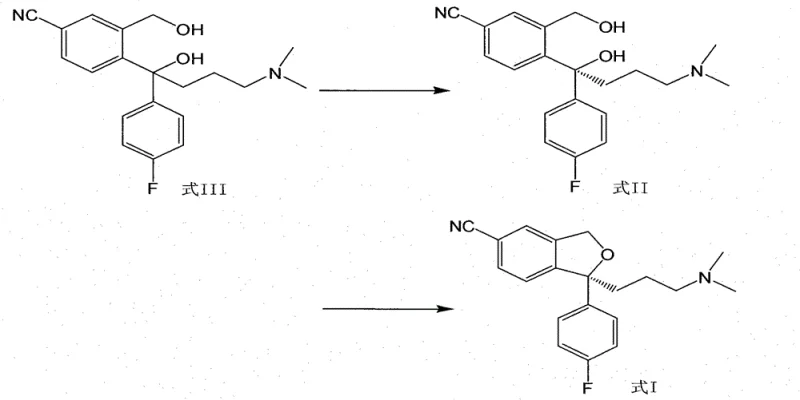
Mechanistic Insights into Chiral Resolution and Cyclization
The core of this technology relies on the principle of diastereomeric salt formation, a classic yet highly effective technique for chiral separation. By reacting the racemic diol (Formula III) with a chiral resolving agent such as D-(+)-di-p-toluoyl tartaric acid in a solvent like isopropanol, diastereomeric salts are formed. Due to the differences in solubility between the (S)-salt and the (R)-salt, the desired (S)-enantiomer preferentially crystallizes out of the solution upon cooling. The patent specifies a critical temperature range of 70 to 85°C for dissolution followed by controlled cooling, which is essential for maximizing the yield and optical purity of the precipitated salt. This thermodynamic control ensures that impurities and the unwanted enantiomer remain in the mother liquor.
Following the isolation of the high-purity (S)-diol (Formula II), the synthesis proceeds via an intramolecular nucleophilic substitution. The primary hydroxyl group of the side chain is activated by Tosyl chloride in the presence of an organic base like triethylamine. This activation converts the hydroxyl into a good leaving group (tosylate), which is then displaced by the phenolic oxygen atom (generated in situ or present as a masked equivalent depending on protection strategy, though here it implies direct cyclization of the phenol-alcohol system). The reaction is conducted in non-protonic solvents like toluene at low temperatures (-5 to 0°C) to suppress side reactions and maintain stereochemical integrity. This mechanistic pathway avoids the harsh conditions of thionyl chloride, preserving the sensitive nitrile and amine functionalities present in the molecule.
How to Synthesize S-Escitalopram Efficiently
The operational protocol for this synthesis is designed for reproducibility and safety in a GMP environment. The process begins with the conversion of the racemic hydrobromide salt to the free base, followed by the critical resolution step in isopropanol. After filtering and washing the chiral salt, it is neutralized to release the pure (S)-diol. The subsequent cyclization requires precise temperature control during the addition of Tosyl chloride to prevent exotherms that could degrade optical purity. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Perform chiral resolution of racemic Formula III diol using D-(+)-di-p-toluoyl tartaric acid in isopropanol, followed by recrystallization to achieve >99.8% ee.
- Neutralize the resolved salt to obtain the free base S-enantiomer diol (Formula II) through standard aqueous workup and extraction.
- Execute intramolecular cyclization in toluene using Tosyl chloride and triethylamine at controlled low temperatures (-5 to 0°C) to form the final phthalan ring structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly impact the bottom line and supply chain resilience. By replacing exotic and expensive reagents like Mosher's acid chloride with commodity chemicals such as di-p-toluoyl tartaric acid and Tosyl chloride, the raw material cost profile is significantly optimized. This shift not only lowers the direct cost of goods sold (COGS) but also mitigates supply risk, as the new reagents are readily available from multiple global vendors, ensuring continuity of supply even during market fluctuations.
- Cost Reduction in Manufacturing: The elimination of preparative HPLC purification represents a massive saving in both operational expenditure and capital equipment. Chromatographic separations are notoriously slow and solvent-intensive; by achieving high purity through crystallization, the process throughput is drastically increased. Furthermore, the use of Tosyl chloride instead of thionyl chloride reduces the need for specialized corrosion-resistant equipment and expensive scrubbing systems for acidic gas treatment, leading to lower maintenance costs and extended reactor lifespan.
- Enhanced Supply Chain Reliability: The reliance on standard solvents like toluene, isopropanol, and ethyl acetate simplifies logistics and inventory management. These solvents are ubiquitous in the fine chemical industry, reducing the likelihood of shortages that can plague specialized reagents. Additionally, the robustness of the crystallization step means that minor variations in raw material quality can often be corrected during the purification phase, providing a buffer against supply chain variability and ensuring consistent output quality for downstream API synthesis.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified workup procedures make this process inherently scalable from pilot plant to multi-ton commercial production. The avoidance of hazardous thionyl chloride aligns with modern green chemistry principles, reducing the generation of sulfur dioxide waste and minimizing the environmental footprint of the manufacturing site. This compliance advantage facilitates easier regulatory approval and reduces the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this S-escitalopram synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential licensees or manufacturing partners.
Q: How does this method improve upon previous S-escitalopram synthesis routes?
A: Unlike prior art requiring expensive Mosher's acid chloride or corrosive thionyl chloride, this patent utilizes cost-effective di-p-toluoyl tartaric acid for resolution and mild Tosyl chloride for cyclization, significantly reducing raw material costs and safety hazards.
Q: What optical purity levels can be achieved with this resolution technique?
A: The described recrystallization process using chiral tartrate salts consistently achieves optical purity (ee values) exceeding 99.8%, meeting stringent regulatory requirements for antidepressant APIs without needing complex chromatographic separation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method employs common solvents like toluene and isopropanol and avoids extreme conditions, making it highly scalable. The simple workup procedures facilitate efficient handling in multi-ton manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Escitalopram Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antidepressants. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of S-escitalopram intermediate meets the high optical purity standards demanded by global regulatory bodies.
We invite pharmaceutical companies and contract manufacturers to collaborate with us on optimizing their supply chains for escitalopram production. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your project's success.
