Advanced One-Pot Synthesis of Beta-Carotene Intermediates for Commercial Scale Production
The landscape of carotenoid synthesis is undergoing a significant transformation driven by the need for higher purity intermediates and more sustainable manufacturing processes. Patent CN114426508A introduces a groundbreaking preparation method for beta-carotene intermediate polyene thioether, addressing long-standing inefficiencies in the industry. This technology leverages a novel one-pot strategy starting from readily available Vitamin A or its derivatives, such as Vitamin A acetate. By integrating halogenation and coupling reactions into a single streamlined workflow, the process achieves a raw material conversion rate exceeding 98% and a reaction yield surpassing 90%. For R&D directors and procurement specialists, this represents a pivotal shift away from legacy methods that suffer from low atom economy and complex waste streams. The ability to produce high-quality polyene thioethers with minimal byproduct formation positions this technology as a cornerstone for next-generation fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carotenoids like beta-carotene has relied heavily on three primary methodologies, each carrying distinct operational burdens that impact overall production efficiency. The Wittig reaction, while chemically straightforward, generates substantial quantities of triphenylphosphine oxide as a byproduct. This impurity is notoriously difficult to separate from the desired polyene product, necessitating complex and costly refining steps that erode profit margins. Similarly, the Roche method, which utilizes acetylene compounds, often results in the formation of undesirable cis-isomers during the hydrogenation stage. These isomers are less active and complicate the purification process, requiring additional crystallization or chromatography steps to ensure the final product meets stringent stereochemical specifications. Furthermore, the Julia reaction, although capable of producing stable sulfone intermediates, still faces challenges regarding reagent costs and the management of sulfur-containing waste. These conventional pathways collectively contribute to extended lead times and elevated operational expenditures, creating a bottleneck for manufacturers aiming to scale production efficiently.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN114426508A offers a streamlined alternative that fundamentally restructures the synthetic pathway. By utilizing Vitamin A derivatives as the starting scaffold, the process bypasses the need for constructing the carbon chain from smaller fragments, thereby reducing the number of unit operations. The core innovation lies in the direct halogenation of the Vitamin A backbone followed by an immediate coupling reaction using a specialized heterogeneous catalyst. This approach not only simplifies the operational workflow but also ensures a high degree of stereocontrol, favoring the formation of the all-trans configuration which is critical for biological activity. The elimination of phosphine-based reagents and the reduction of cis-isomer formation directly translate to a cleaner crude product profile. For supply chain managers, this means fewer processing stages, reduced solvent consumption, and a significantly lower environmental footprint, aligning perfectly with modern green chemistry mandates.
Mechanistic Insights into Activated Carbon-Mediated Coupling
The heart of this technological advancement is the unique coupling agent system, which employs activated carbon loaded with sulfur salts and specific metal or non-metal nitrides. The reaction initiates with the halogenation of Vitamin A acetate using strong inorganic acids and halides, generating a reactive halogenated intermediate as depicted in the structural formula below. This intermediate serves as the electrophilic partner in the subsequent coupling step. The use of solvents such as dichloromethane, toluene, or heptane allows for precise control over reaction kinetics, ensuring that the halogenation proceeds selectively without degrading the sensitive polyene chain. The careful regulation of temperature between 0°C and 20°C during this phase is crucial for maintaining the integrity of the conjugated system while maximizing the introduction of the halogen leaving group.
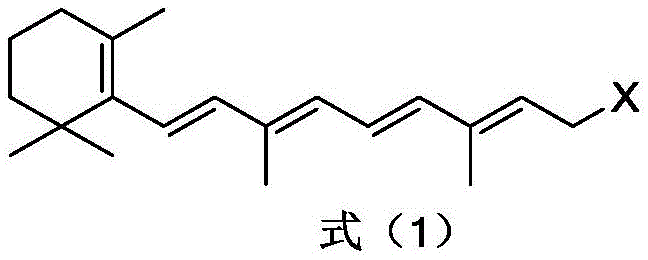
Following the formation of the halogenated species, the system transitions directly into the coupling phase without isolation, a key feature of the one-pot design. The coupling agent, comprising sulfur salts like sodium sulfide supported on coconut shell activated carbon, facilitates the nucleophilic substitution that links two polyene chains via a sulfur bridge. Crucially, the inclusion of auxiliary agents such as aluminum nitride (AlN) or boron nitride (BN) plays a dual role: it enhances the mechanical strength of the catalyst on the carbon support and, more importantly, coordinates with the sulfur species to inhibit the formation of disulfide byproducts. This selectivity mechanism ensures that the reaction yields the desired thioether rather than oxidized sulfur impurities. The resulting polyene thioether structure, shown below, exhibits a symmetrical arrangement ideal for subsequent oxidation and desulfonation to yield beta-carotene. The activated carbon matrix further acts as an adsorbent, trapping inorganic salts and minor organic impurities generated during the reaction, thereby simplifying the final workup.
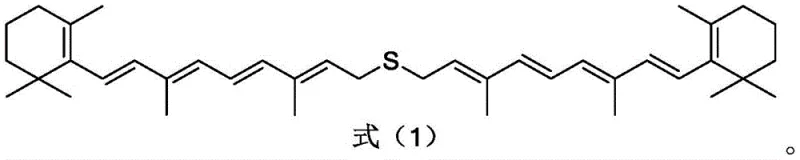
How to Synthesize Polyene Thioether Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing fine chemical manufacturing facilities. The process begins with the preparation of the halogenated precursor under controlled acidic conditions, followed by a straightforward aqueous wash to remove residual acids and salts. This intermediate is then subjected to the coupling reaction in the same vessel, minimizing material transfer losses and exposure to air. The standardized protocol emphasizes the importance of catalyst preparation, specifically the soaking and drying of the activated carbon support to ensure optimal loading of the sulfur and nitride components. Detailed standard operating procedures regarding temperature ramping, stirring rates, and quenching methods are essential to replicate the high yields reported in the patent examples. For technical teams looking to adopt this methodology, the following guide outlines the critical operational parameters required to achieve consistent results.
- Halogenation: React Vitamin A acetate with sodium bromide and sulfuric acid in a chlorinated hydrocarbon solvent at 0-20°C to form the halogenated intermediate.
- Workup: Wash the reaction mixture with water to remove acids and unreacted salts, separating the organic phase containing the halogenated product.
- Coupling: Add a sulfur-loaded activated carbon coupling agent containing metal nitrides to the organic phase and react at 30-50°C to form the polyene thioether.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages that extend beyond mere chemical yield. For procurement managers tasked with optimizing cost structures, the elimination of expensive phosphine reagents and the reduction of purification steps represent a significant opportunity for cost reduction in fine chemical manufacturing. The one-pot nature of the process drastically reduces solvent usage and energy consumption associated with multiple isolation and drying cycles. Furthermore, the use of robust, heterogeneous catalysts that can be potentially regenerated or handled more safely than homogeneous alternatives lowers the total cost of ownership for reagents. These factors combine to create a more economically resilient supply chain, less susceptible to volatility in raw material pricing for exotic catalysts.
- Cost Reduction in Manufacturing: The streamlined workflow eliminates the need for complex chromatographic separations typically required to remove triphenylphosphine oxide in Wittig reactions. By avoiding these resource-intensive purification stages, manufacturers can achieve substantial cost savings in both labor and materials. The high selectivity of the nitride-modified catalyst minimizes waste generation, further reducing disposal costs and improving overall process mass intensity. This efficiency allows for a more competitive pricing structure for the final beta-carotene intermediate, enhancing market positioning.
- Enhanced Supply Chain Reliability: Relying on Vitamin A derivatives as starting materials leverages a well-established global supply chain, ensuring consistent availability of feedstock. Unlike specialized acetylene or sulfone precursors which may have limited suppliers, Vitamin A acetate is a commodity chemical with robust production capacity. This accessibility reduces the risk of supply disruptions and shortens lead times for high-purity polyene thioethers. Additionally, the simplified process flow reduces the dependency on highly specialized equipment, allowing for greater flexibility in production scheduling and facility utilization.
- Scalability and Environmental Compliance: The use of activated carbon as a catalyst support aligns with increasingly stringent environmental regulations regarding heavy metal usage and waste discharge. The solid nature of the coupling agent facilitates easy filtration and separation, minimizing the release of sulfur-containing effluents into wastewater streams. This inherent safety and environmental profile simplifies the permitting process for commercial scale-up of complex carotenoid intermediates. Manufacturers can scale production from pilot batches to multi-ton capacities with confidence, knowing that the process adheres to green chemistry principles and reduces the ecological footprint of the operation.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is critical for stakeholders evaluating its potential for integration into their production portfolios. The following questions address common inquiries regarding catalyst performance, product purity, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. By clarifying these aspects, we aim to facilitate a smoother transition from laboratory validation to industrial implementation.
Q: What is the primary advantage of the activated carbon coupling agent in this synthesis?
A: The activated carbon loaded with sulfur salts and metal nitrides significantly improves reaction selectivity by inhibiting disulfide byproduct formation. Additionally, the porous structure adsorbs impurities, simplifying downstream purification.
Q: How does this method compare to traditional Wittig reactions for carotenoid synthesis?
A: Unlike Wittig reactions which generate difficult-to-remove triphenylphosphine oxide impurities, this thioether method offers a cleaner profile with >90% yield and avoids complex refining steps associated with phosphine oxides.
Q: Can this process be scaled for industrial manufacturing of beta-carotene precursors?
A: Yes, the patent explicitly highlights suitability for industrial production due to the one-pot nature of the reaction, mild temperature conditions (0-60°C), and the use of robust heterogeneous catalysts that facilitate easy separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Carotene Intermediate Supplier
As the demand for high-quality carotenoids continues to grow across the food, feed, and pharmaceutical sectors, securing a dependable source of advanced intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge synthetic technologies like the one described in CN114426508A to deliver superior value. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of polyene thioether meets the exacting standards required for downstream beta-carotene synthesis.
We invite you to explore how our technical expertise can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced capabilities can support your long-term strategic goals. Let us be your partner in driving innovation and efficiency in the global fine chemical market.
