Advanced Synthesis of Polysubstituted Naphthoquinone Derivatives for Oncology Applications
Introduction to Novel Anti-Tumor Naphthoquinone Intermediates
The landscape of oncology drug discovery is constantly evolving, driven by the urgent need for more effective therapeutic agents against resistant malignancies. In this context, patent CN113292532B introduces a groundbreaking class of polysubstituted naphthoquinone derivatives that exhibit profound anti-tumor potential. These compounds, characterized by a unique 1,4-naphthoquinone scaffold substituted with diverse functional groups at key positions, represent a significant advancement in medicinal chemistry. The general structure, depicted below, allows for extensive structural modulation to optimize pharmacokinetic properties and biological efficacy. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such novel scaffolds is critical for accelerating the development pipeline from bench to bedside.
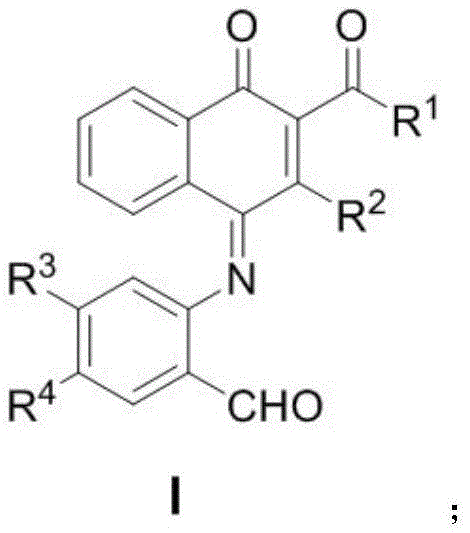
The significance of this technology lies not only in the biological potency of the final molecules but also in the elegance of their construction. Traditional methods for synthesizing complex quinone systems often involve harsh oxidative conditions, multiple protection-deprotection sequences, and low overall yields, which pose significant bottlenecks for process chemists. In contrast, the methodology described in this patent offers a streamlined pathway that aligns perfectly with the principles of green chemistry. By leveraging a sophisticated gold-catalyzed transformation, researchers can access these high-value intermediates with unprecedented efficiency. This report delves deep into the mechanistic underpinnings, comparative advantages, and commercial implications of this technology for stakeholders in the global pharmaceutical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted naphthoquinones has been a formidable challenge in organic synthesis, often plagued by inefficiencies that hinder large-scale production. Conventional routes typically rely on the oxidation of naphthalene or anthracene precursors using stoichiometric amounts of toxic heavy metal oxidants such as chromium(VI) reagents or manganese dioxide. These processes are not only environmentally hazardous due to the generation of substantial heavy metal waste but also suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate. Furthermore, introducing specific substituents at the 2- or 3-positions of the quinone ring often requires multi-step sequences involving Friedel-Crafts acylations or Diels-Alder reactions followed by aromatization, each step adding to the cumulative loss of yield and increasing the overall cost of goods sold (COGS). The harsh reaction conditions, frequently requiring high temperatures or strong acids, can also degrade sensitive functional groups, limiting the scope of chemical diversity that can be explored during lead optimization.
The Novel Approach
The innovative strategy outlined in patent CN113292532B fundamentally disrupts these traditional paradigms by employing a transition metal-catalyzed annulation reaction. This approach utilizes readily available diazo compounds and benzisoxazole derivatives as building blocks, which react in a single pot to construct the naphthoquinone core. This one-step convergence eliminates the need for pre-functionalized quinone starting materials and avoids the use of stoichiometric oxidants entirely. The reaction proceeds under remarkably mild conditions, typically around 30°C, which preserves the integrity of sensitive moieties such as esters, ethers, and halides present on the substrate. This mildness translates directly into higher purity profiles and simplified downstream processing. For procurement managers and supply chain heads, this shift represents a move towards cost reduction in API manufacturing by reducing the number of unit operations, minimizing solvent consumption, and eliminating the expensive disposal costs associated with heavy metal waste streams.
Mechanistic Insights into Gold-Catalyzed Cyclization
The core of this technological breakthrough is the gold(I)-catalyzed reaction between a diazo compound (SI-1) and a benzisoxazole derivative (SI-2). The mechanism likely initiates with the activation of the diazo species by the cationic gold catalyst, generating a reactive gold-carbenoid intermediate. This electrophilic species then undergoes a nucleophilic attack by the nitrogen or oxygen atom of the benzisoxazole ring, triggering a cascade of rearrangements. Specifically, the N-O bond of the benzisoxazole is cleaved, facilitating the incorporation of the oxygen atom into the quinone carbonyl system while the nitrogen forms the imine linkage observed in the final product structure. This tandem process effectively stitches together the two fragments with high atom economy, as nearly all atoms from the starting materials are incorporated into the final product or benign byproducts like nitrogen gas. The use of a specialized ligand system, such as [(2-biphenyl)di-tert-butylphosphine], stabilizes the gold center and fine-tunes its electronic properties to ensure high turnover numbers and exceptional selectivity for the desired 1,4-naphthoquinone scaffold over potential side products.
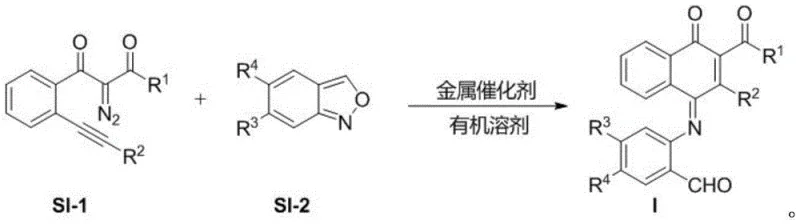
From an impurity control perspective, this mechanism offers distinct advantages for R&D directors focused on quality. The high selectivity of the gold catalyst minimizes the formation of regioisomers and oligomeric byproducts that are common in radical-based oxidations. The reaction is performed in anhydrous 1,2-dichloroethane, a solvent that provides excellent solubility for both organic substrates and the metal catalyst while maintaining a stable reaction environment. The mild temperature of 30°C further suppresses thermal decomposition pathways. Consequently, the crude reaction mixture is significantly cleaner than those obtained from traditional methods, often requiring only filtration to remove the catalyst followed by standard column chromatography to achieve high-purity pharmaceutical intermediates. This level of control is essential for meeting the stringent purity specifications required for clinical trial materials and ensures a robust, reproducible process that can be validated for GMP manufacturing.
How to Synthesize Polysubstituted Naphthoquinone Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward due to the operational simplicity of the protocol. The process begins by accurately weighing the diazo compound SI-1 and the benzisoxazole SI-2 in a molar ratio of approximately 1:1.2, ensuring a slight excess of the benzisoxazole to drive the reaction to completion. These solids are dissolved in anhydrous 1,2-dichloroethane to achieve a concentration of roughly 0.1 mol/L. A precise amount of the gold(I) catalyst, typically 5 mol% relative to the diazo compound, is added to the mixture. The detailed standardized synthesis steps for scaling this reaction are provided in the guide below.
- Mix diazo compound SI-1 and benzisoxazole SI-2 in anhydrous 1,2-dichloroethane with a gold(I) catalyst.
- Stir the reaction mixture at 30°C for 24 hours until the diazo compound is fully consumed.
- Filter the reaction solution and purify the crude product via column chromatography to obtain the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling strategic benefits that extend beyond mere technical novelty. The shift from multi-step, waste-intensive processes to a convergent, catalytic one-step synthesis fundamentally alters the cost structure of producing these valuable oncology intermediates. By reducing the number of isolation and purification steps, manufacturers can significantly lower labor costs, energy consumption, and solvent usage. The elimination of stoichiometric heavy metal oxidants removes a major environmental liability and reduces the regulatory burden associated with waste disposal. Furthermore, the use of commercially available starting materials ensures a stable supply chain, mitigating the risk of raw material shortages that often plague custom synthesis projects. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream drug developers.
- Cost Reduction in Manufacturing: The streamlined nature of this gold-catalyzed process drives substantial cost savings by collapsing multiple synthetic transformations into a single operation. Traditional routes often require protecting group strategies and separate oxidation steps, each incurring material and processing costs. In contrast, this method achieves the target complexity directly, reducing the overall material throughput required per kilogram of product. The high atom economy means less raw material is wasted as byproducts, directly improving the yield and lowering the effective cost per gram. Additionally, the mild reaction conditions reduce energy expenditures for heating or cooling, contributing to a leaner and more cost-effective manufacturing profile that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry, and this synthesis method bolsters reliability through the use of robust and accessible feedstocks. The starting materials, diazo compounds and benzisoxazoles, are widely available from bulk chemical suppliers, reducing dependency on niche vendors. The simplicity of the reaction setup—requiring only standard glassware or reactors without specialized high-pressure equipment—allows for flexible manufacturing across different facilities. This flexibility ensures that production can be easily scaled or shifted if necessary, providing a buffer against logistical disruptions. The consistent quality of the output further reduces the risk of batch failures, ensuring that downstream customers receive materials that meet specifications on time, every time.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often reveals hidden challenges, but this methodology is inherently scalable due to its homogeneous nature and lack of exothermic hazards. The reaction proceeds smoothly at 30°C, eliminating the need for complex temperature control systems required for highly exothermic oxidations. From an environmental standpoint, the process aligns with modern sustainability goals by minimizing waste generation and avoiding toxic reagents. The high selectivity reduces the load on wastewater treatment systems, and the potential for catalyst recovery further enhances the green credentials of the process. This compliance with environmental standards facilitates smoother regulatory approvals and supports the growing demand for sustainably sourced pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these polysubstituted naphthoquinone derivatives. These insights are derived directly from the patent data and practical experience in process development, aiming to clarify the value proposition for potential partners. Understanding these details helps stakeholders make informed decisions about integrating this technology into their own R&D pipelines or supply networks.
Q: What is the primary advantage of the gold-catalyzed synthesis method for naphthoquinones?
A: The primary advantage is the significant reduction in reaction steps, achieving the complex naphthoquinone scaffold in a single step with high atom economy and mild conditions (30°C), which drastically simplifies purification and reduces waste compared to traditional multi-step oxidations.
Q: Which cancer cell lines have shown sensitivity to these derivatives?
A: The derivatives have demonstrated potent inhibitory activity against HCT-116 human colorectal adenocarcinoma cells, SJSA-1 human osteosarcoma cells, and MCF-7 breast cancer cells, with IC50 values ranging from approximately 4.92 μM to 15.07 μM for the most active compounds.
Q: Is this synthesis method scalable for commercial production?
A: Yes, the method utilizes commercially available starting materials and operates under mild temperatures with simple workup procedures (filtration and chromatography), making it highly suitable for commercial scale-up from kilogram to metric ton quantities without requiring extreme pressure or cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Naphthoquinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in patent CN113292532B for the development of next-generation anti-tumor therapies. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into viable commercial realities. We are committed to delivering high-purity polysubstituted naphthoquinones that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch supplied adheres to the highest industry standards, providing our clients with the confidence needed to advance their clinical programs.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced chemistry for their oncology portfolios. Whether you require custom synthesis of specific analogs or bulk supply of the core intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our capabilities can accelerate your project timelines and optimize your manufacturing costs.
