Scalable Palladium-Free Synthesis of 3'-Nitro-2'-hydroxybiphenyl-3-carboxylic Acid for API Production
Scalable Palladium-Free Synthesis of 3'-Nitro-2'-hydroxybiphenyl-3-carboxylic Acid for API Production
The pharmaceutical industry constantly seeks robust and economically viable pathways for synthesizing critical active pharmaceutical ingredient (API) intermediates. A significant breakthrough in this domain is detailed in patent CN110467531B, which discloses a highly efficient preparation method for 3'-nitro-2'-hydroxybiphenyl-3-carboxylic acid. This compound serves as a pivotal building block for Eltrombopag, a potent thrombopoietin receptor agonist used in treating chronic immune thrombocytopenia. The structural relationship between the final drug and this key intermediate highlights the necessity for high-purity precursors to ensure downstream drug safety and efficacy. As a reliable pharmaceutical intermediate supplier, understanding these advanced synthetic routes is crucial for maintaining supply chain integrity.
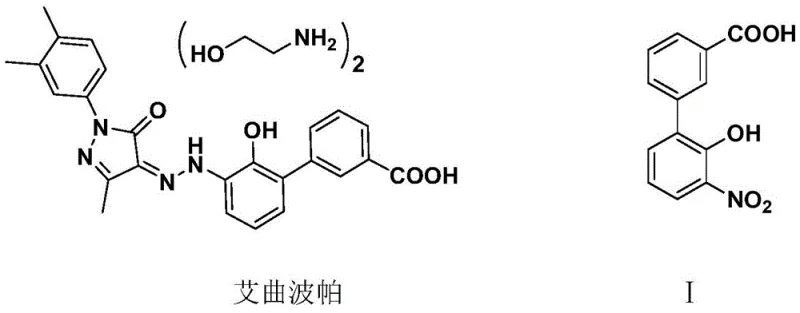
The innovation lies in departing from traditional transition-metal catalyzed couplings, offering a streamlined alternative that addresses both cost and environmental concerns. By leveraging a unique cascade of addition, condensation, and cyclization reactions, this methodology achieves superior yields while minimizing hazardous waste. For procurement managers and R&D directors alike, this represents a strategic opportunity to optimize the manufacturing of complex biphenyl derivatives without compromising on quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3'-nitro-2'-hydroxybiphenyl-3-carboxylic acid has relied heavily on palladium-catalyzed cross-coupling reactions, such as the Suzuki-Miyaura coupling. In prior art routes, such as those described in US2004019190, the construction of the biphenyl skeleton necessitates the use of expensive palladium catalysts and specialized boronic acid reagents. Furthermore, these conventional pathways often involve multiple protection and deprotection steps, such as the methylation of hydroxyl groups using costly and toxic reagents like methyl iodide. The initial nitration steps in these legacy processes frequently suffer from poor selectivity and low yields, sometimes as low as 25%, generating substantial quantities of waste acid that complicate environmental compliance.
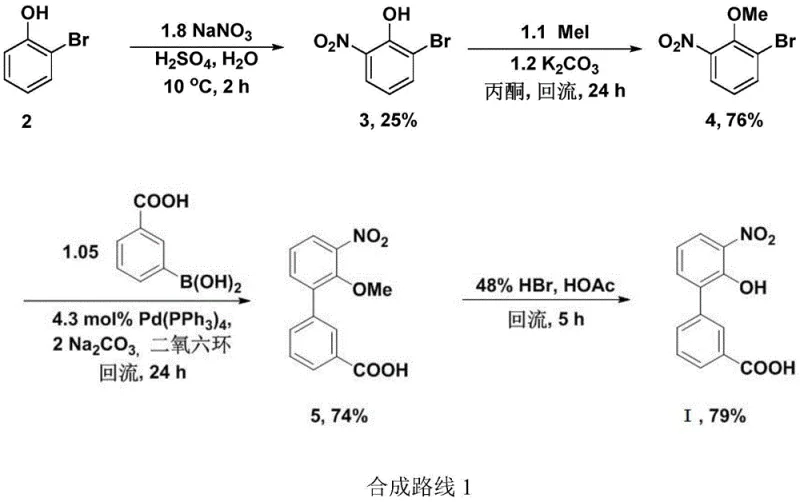
Another existing method involves bromination using N-bromosuccinimide (NBS) followed by palladium coupling, which similarly incurs high raw material costs and requires rigorous purification to remove trace metal residues. These factors collectively inflate the production cost and extend the lead time, making large-scale commercialization challenging. The reliance on precious metals also introduces supply chain vulnerabilities, as fluctuations in palladium availability can disrupt production schedules. Consequently, there is a pressing demand for a metal-free alternative that simplifies the process flow and enhances overall economic efficiency.
The Novel Approach
The novel methodology presented in the patent data revolutionizes this synthesis by utilizing a completely different disconnection strategy that avoids transition metals entirely. Instead of building the biphenyl core through coupling, the new route constructs the molecule through an intramolecular cyclization of a carefully designed precursor. This approach starts with inexpensive and widely available commodities like 2-halogenated acrolein and nitromethane. The process flow is significantly shortened, reducing the number of unit operations and eliminating the need for chromatographic purification typically associated with metal-catalyzed reactions. By removing the dependency on palladium and methylating agents, the method drastically lowers the raw material expenditure and simplifies the waste treatment protocol.
Mechanistic Insights into Base-Catalyzed Addition and Acid-Catalyzed Cyclization
The core of this innovative synthesis involves a two-stage mechanistic sequence that ensures high regioselectivity and yield. The first stage is a base-catalyzed Michael-type addition reaction where nitromethane adds across the double bond of 2-halogenated acrolein. This reaction is facilitated by organic bases such as DBU or piperidine in solvents like tetrahydrofuran or acetonitrile at moderate temperatures ranging from 30°C to 70°C. The result is the formation of 2-halo-4-nitro-n-butyraldehyde, a versatile intermediate that retains the necessary functionality for subsequent ring closure. The choice of base and solvent is critical here to prevent polymerization of the acrolein derivative while ensuring complete conversion of the nitroalkane.
The second stage involves a dehydration condensation followed by a base-mediated cyclization and hydrolysis. The aldehyde intermediate reacts with 3-alkoxycarbonyl phenylacetate in the presence of an acid catalyst like p-toluenesulfonic acid to form an unsaturated ester. Crucially, the subsequent cyclization is performed in a one-pot manner by introducing the reaction mixture into an aqueous alkali solution. This specific operational technique, where the intermediate is added dropwise to the base, maintains a low local concentration of the reactive species. This kinetic control favors the desired intramolecular cyclization over competing intermolecular condensation side reactions, thereby maximizing the formation of the biphenyl core. Finally, acidification precipitates the target 3'-nitro-2'-hydroxybiphenyl-3-carboxylic acid with high purity.
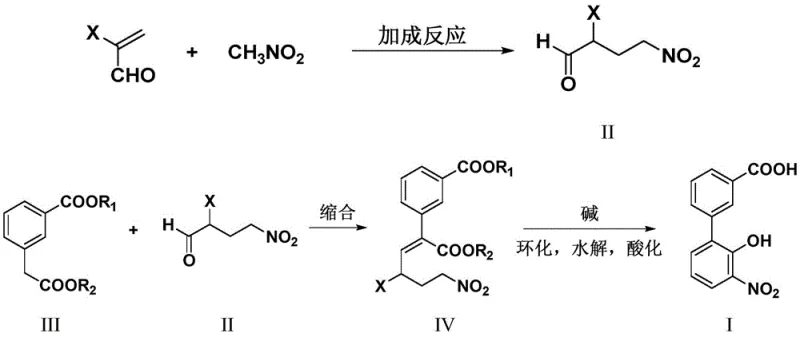
How to Synthesize 3'-Nitro-2'-hydroxybiphenyl-3-carboxylic Acid Efficiently
Implementing this synthesis requires precise control over reaction parameters to replicate the high yields reported in the patent examples. The process begins with the preparation of the nitro-aldehyde backbone, followed by a telescoped condensation and cyclization sequence. Operators must adhere to specific temperature profiles and addition rates to maintain the selectivity advantages inherent in the design. The following guide outlines the standardized operational procedure derived from the patent disclosure, ensuring reproducibility and safety during scale-up. Detailed standard operating procedures for each step are essential for technology transfer.
- Perform an addition reaction between 2-halogenated acrolein and nitromethane using a base catalyst like DBU in a solvent such as THF at 30-70°C to obtain 2-halo-4-nitro-n-butyraldehyde.
- Conduct a dehydration condensation between the resulting aldehyde and 3-alkoxycarbonyl phenylacetate using an acid catalyst like p-toluenesulfonic acid.
- Execute a one-pot cyclization and hydrolysis by adding the condensation product to an aqueous alkali solution, followed by acidification to isolate the final carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this novel synthetic route offers transformative benefits that extend beyond simple chemistry. The elimination of precious metal catalysts removes a significant cost driver and mitigates the risk associated with volatile commodity prices for palladium. Furthermore, the use of commodity chemicals as starting materials ensures a stable and diversified supply base, reducing the likelihood of shortages. The simplified process flow, characterized by fewer isolation steps and a one-pot cyclization, translates directly into reduced processing time and lower energy consumption per kilogram of product. These efficiencies contribute to a more resilient and cost-effective supply chain for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete removal of expensive palladium catalysts and methylating agents like methyl iodide from the bill of materials. In traditional routes, the cost of these reagents combined with the expense of metal scavenging resins to meet residual metal specifications can be prohibitive. By switching to an organocatalytic system using inexpensive amines and sulfonic acids, the variable cost of goods sold is significantly decreased. Additionally, the high yield of the reaction minimizes raw material waste, further enhancing the economic viability of the process for large volume production.
- Enhanced Supply Chain Reliability: Relying on bespoke or highly specialized reagents often creates single points of failure in a supply chain. This new method utilizes 2-halogenated acrolein and nitromethane, which are bulk chemicals produced by multiple global manufacturers. This abundance ensures that procurement teams can source materials competitively and maintain continuous production even if one supplier faces disruptions. The robustness of the chemistry also means that the process is less sensitive to minor variations in reagent quality, providing greater operational flexibility and reducing the need for stringent incoming quality control on exotic starting materials.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers substantial advantages by generating significantly less waste acid and wastewater compared to nitration-heavy prior art. The absence of heavy metals simplifies the effluent treatment process and reduces the regulatory burden associated with disposing of metal-contaminated waste streams. The one-pot nature of the cyclization step reduces solvent usage and energy demand for heating and cooling cycles between steps. These green chemistry attributes facilitate easier permitting for new manufacturing lines and align with corporate sustainability goals, making the facility more attractive to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent documentation. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers provide clarity on yield expectations, purity profiles, and scalability potential.
Q: Why is this new synthesis route more cost-effective than prior art?
A: The novel method eliminates the need for expensive palladium catalysts and methylating agents like methyl iodide, which are required in conventional routes. Additionally, it utilizes cheap and readily available starting materials such as 2-halogenated acrolein and nitromethane.
Q: What represents the key technical advantage regarding purity?
A: The process achieves high selectivity through a controlled one-pot cyclization where the intermediate is added dropwise to an alkali solution. This minimizes intermolecular side reactions and ensures high product purity suitable for pharmaceutical applications.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is designed for scalability with short process flows, mild reaction conditions, and reduced generation of waste acid and water, making it environmentally friendly and operationally simple for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3'-Nitro-2'-hydroxybiphenyl-3-carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications like Eltrombopag. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch supports your downstream synthesis without interruption. Our capability to adapt advanced patent technologies like the one described here allows us to offer superior value to our global partners.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project timelines are met with the highest standards of quality and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
