Advanced Manufacturing of Zaleplon Intermediates: Technical Breakthroughs and Commercial Scalability
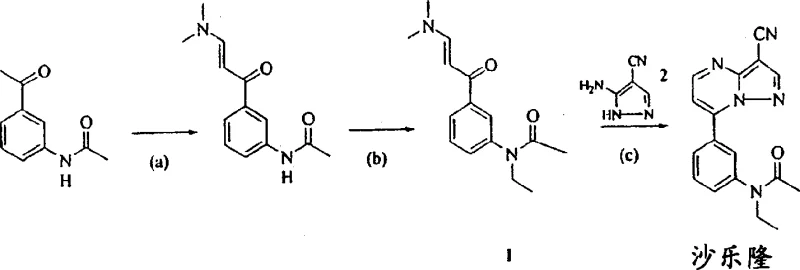
The pharmaceutical landscape for central nervous system therapeutics continues to evolve, driven by the demand for safer, more efficient manufacturing processes for active pharmaceutical ingredients (APIs). Patent CN1537113A represents a significant technological leap in the synthesis of N-[3-(3-cyanopyrazolo[1,5-a]pyrimidin-7-yl)phenyl]-N-ethylacetamide, commonly known as Zaleplon or Saleron, a potent non-benzodiazepine hypnotic agent approved for the short-term treatment of insomnia. This patent discloses a novel condensation methodology that replaces traditional, energy-intensive reflux protocols with a sophisticated aqueous-organic solvent system operating under mild acidic conditions. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic nuances of this invention is critical, as it offers a pathway to substantially higher yields and superior impurity profiles compared to legacy methods described in earlier patents like USP 4,626,538. The core innovation lies in the precise manipulation of solvent polarity to manage the solubility of reactive intermediates, thereby preventing the formation of oily precipitates that historically plagued the scalability of this chemical transformation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Zaleplon intermediates relied heavily on the use of glacial acetic acid as both the solvent and the acid source, requiring prolonged heating under reflux conditions for approximately eight hours to drive the condensation reaction to completion. This conventional approach, while chemically feasible, presents significant drawbacks for modern cost reduction in API manufacturing initiatives, primarily due to the high energy consumption associated with maintaining elevated temperatures over extended periods. Furthermore, the use of large volumes of glacial acetic acid complicates waste stream management and increases the environmental footprint of the production facility, creating regulatory hurdles for sustainability compliance. From a quality control perspective, the harsh thermal conditions can promote the formation of degradation by-products and regioisomers, necessitating extensive downstream purification steps that erode overall process efficiency and final yield. The inability to effectively control the solubility of the imine intermediate in pure acetic acid often leads to heterogeneous reaction mixtures, resulting in inconsistent batch-to-batch reproducibility and potential safety risks associated with handling hot, corrosive acidic media on a large industrial scale.
The Novel Approach
In stark contrast to these legacy limitations, the methodology outlined in CN1537113A introduces a paradigm shift by utilizing a reaction medium composed of water and at least one water-miscible organic compound, such as methanol, ethanol, or acetonitrile. This binary solvent system is ingeniously designed to decouple the solvating power of the medium from the acidity required for catalysis, allowing for independent optimization of reaction kinetics and intermediate stability. By maintaining the reaction at ambient temperatures ranging from 20°C to 25°C, the process eliminates the need for external heating, thereby drastically reducing energy costs and enhancing operational safety for plant personnel. The presence of the organic co-solvent ensures that the critical imine intermediate remains in solution rather than precipitating as an intractable oil, facilitating its smooth conversion into the final pyrazolo-pyrimidine ring system. This refinement not only accelerates the reaction rate, often completing within two to eight hours, but also delivers high-purity pharmaceutical intermediates with HPLC purity levels consistently exceeding 98.5%, directly addressing the stringent quality requirements of global regulatory bodies.
Mechanistic Insights into Acid-Catalyzed Cyclization in Aqueous Media
The chemical elegance of this process is rooted in a detailed understanding of the reaction mechanism, which proceeds through a Michael-type addition followed by an elimination-cyclization sequence. Initially, the 3-amino group of the 3-amino-4-cyanopyrazole nucleophile attacks the beta-carbon of the enamine substrate, N-[3-[3-(dimethylamino)-1-oxo-2-propenyl]phenyl]-N-ethylacetamide, forming a transient charge-separated intermediate. Under the influence of the added mineral acid, such as hydrochloric or phosphoric acid, dimethylamine is alpha-eliminated from this intermediate, generating a reactive imine species that retains the double bond character necessary for subsequent ring closure. Crucially, the patent data reveals that this imine intermediate possesses distinct polarity characteristics; in purely aqueous inorganic acid environments, it tends to separate as an oily precipitate, halting the reaction progress. However, the introduction of the water-miscible organic co-solvent modifies the dielectric constant of the medium, keeping the imine dissolved and available for the final intramolecular cyclization step where the pyrazole nitrogen attacks the ketone carbonyl, eliminating water to form the stable aromatic pyrazolo-pyrimidine core of Zaleplon.
From an impurity control standpoint, this mechanistic clarity allows for the precise suppression of the major regioisomeric impurity, N-[3-(3-cyanopyrazolo[1,5-a]pyrimidin-5-yl)phenyl]-N-ethylacetamide. The formation of this unwanted isomer is highly dependent on the specific acid concentration and solvent composition; the patent demonstrates that by carefully tuning the molar equivalents of acid relative to the limiting reagent, manufacturers can steer the reaction exclusively towards the desired 7-substituted product. The ability to identify and quantify this regioisomer using advanced HPLC-MS techniques provides a robust analytical framework for quality assurance, ensuring that the final API meets the rigorous specifications required for human consumption. This level of mechanistic control is essential for commercial scale-up of complex heterocycles, as it minimizes the risk of batch rejection due to out-of-specification impurity profiles, thereby securing the supply chain continuity for downstream drug product formulation.
How to Synthesize Zaleplon Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized parameters regarding solvent ratios, acid selection, and temperature control to maximize yield and purity. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic cooling, which makes it accessible for a wide range of manufacturing facilities aiming to upgrade their production capabilities. By following the standardized protocol derived from the patent examples, production teams can achieve consistent results with minimal variation, ensuring that every batch meets the stringent quality standards expected by international pharmaceutical clients. The detailed standard operating procedures for this synthesis are critical for technology transfer and validation, providing a clear roadmap from laboratory bench to pilot plant and finally to full-scale commercial production.
- Dissolve N-[3-[3-(dimethylamino)-1-oxo-2-propenyl]phenyl]-N-ethylacetamide and 3-amino-4-cyanopyrazole in a mixture of water and a water-miscible organic co-solvent such as methanol or ethanol.
- Add a mineral acid like hydrochloric acid or phosphoric acid to the reaction mixture to maintain acidic conditions necessary for the condensation.
- Stir the mixture at room temperature (20-25°C) for 2 to 8 hours, then cool to induce precipitation of the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages that extend far beyond simple chemical yield improvements. The shift to a water-based solvent system significantly reduces the reliance on expensive, volatile organic solvents, leading to a substantial decrease in raw material procurement costs and waste disposal fees. Furthermore, the ability to run the reaction at room temperature eliminates the capital expenditure associated with large-scale heating jackets and condensers, while simultaneously reducing the facility's overall energy consumption profile. These factors combine to create a more resilient and cost-effective supply chain, capable of responding rapidly to market demands without the bottlenecks typically associated with thermally intensive chemical processes. The robustness of the method also implies a lower risk of production delays caused by equipment failure or safety incidents, ensuring a steady flow of critical intermediates to API manufacturing sites.
- Cost Reduction in Manufacturing: The elimination of energy-intensive reflux steps and the replacement of glacial acetic acid with cheaper, readily available solvents like methanol and water result in significant operational expenditure savings. By avoiding the need for complex solvent recovery systems required for pure organic streams, the process simplifies the downstream workup, reducing both labor and utility costs associated with distillation and drying. Additionally, the high conversion rates minimize the loss of valuable starting materials, ensuring that the cost per kilogram of the final intermediate is optimized for maximum profitability in a competitive generic drug market.
- Enhanced Supply Chain Reliability: The use of commodity chemicals such as hydrochloric acid, phosphoric acid, and methanol ensures that the supply of raw materials is not subject to the volatility often seen with specialized reagents. This accessibility guarantees that production schedules can be maintained without interruption, even during periods of global supply chain disruption. The simplified process flow also reduces the turnaround time between batches, allowing manufacturers to increase throughput and reducing lead time for high-purity APIs to meet urgent customer orders or unexpected spikes in demand for insomnia treatments.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium aligns perfectly with modern green chemistry principles, significantly lowering the volume of hazardous organic waste generated per unit of product. This environmental benefit simplifies regulatory compliance and reduces the burden on wastewater treatment facilities, making the process highly attractive for manufacturing in regions with strict environmental protections. The inherent safety of operating at ambient temperatures further facilitates seamless scale-up from pilot batches to multi-ton production runs, mitigating the thermal runaway risks that often complicate the expansion of exothermic condensation reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Zaleplon synthesis process, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational feasibility and quality advantages of the method for stakeholders evaluating potential technology partnerships. Understanding these details is crucial for making informed decisions about process adoption and long-term supply agreements.
Q: How does the new aqueous-organic solvent system improve purity compared to glacial acetic acid methods?
A: The use of water mixed with organic co-solvents prevents the premature precipitation of the imine intermediate, ensuring complete conversion to the final pyrazolo-pyrimidine structure and achieving HPLC purity exceeding 98.5%.
Q: What are the primary impurities controlled in this synthesis route?
A: The process specifically manages the formation of the regioisomer N-[3-(3-cyanopyrazolo[1,5-a]pyrimidin-5-yl)phenyl]-N-ethylacetamide, keeping its levels negligible through optimized acid concentration and solvent ratios.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the elimination of high-temperature reflux and the use of common solvents like methanol and water make the process highly scalable, safe, and economically viable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Zaleplon Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of CN1537113A are fully realized in your supply chain. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Zaleplon intermediate we produce meets the highest global standards for safety and efficacy. We understand the critical nature of CNS active agents and the zero-tolerance policy for impurities, which is why our process engineering team focuses relentlessly on optimizing every variable of the synthesis to deliver consistent, high-quality results.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this greener, more efficient process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate our capabilities and secure a reliable supply of this critical pharmaceutical intermediate for your future projects.
