Advanced Separation of Cephalosporin Z/E Isomers for Commercial API Manufacturing
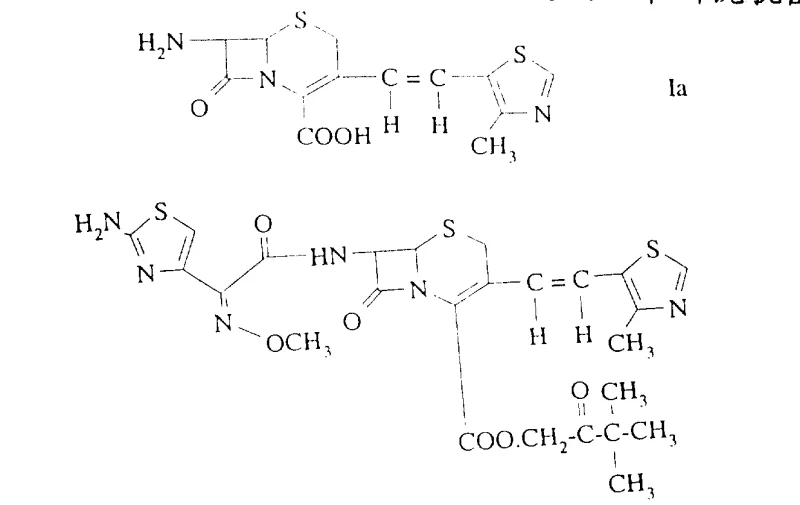
The pharmaceutical industry continuously seeks robust methodologies to ensure the highest purity of active pharmaceutical ingredients (APIs), particularly for complex beta-lactam antibiotics where stereochemistry dictates biological activity. Patent CN1107850A introduces a groundbreaking process for reducing the content of the undesired E-isomer in the Z/E mixture of 7-amino-3-[2-(4-methyl-5-thiazolyl)vinyl]-3-cephem-4-carboxylic acid. This specific intermediate is critical for the synthesis of Cefditoren pivoxil, a potent oral cephalosporin antibiotic. The invention addresses a persistent challenge in cephalosporin synthesis where conventional coupling reactions inevitably produce a mixture of Z (cis) and E (trans) isomers at the 3-position vinyl side chain. Since the Z-configuration is essential for the optimal antibacterial efficacy of the final drug product, the ability to efficiently separate or enrich the Z-isomer is of paramount importance to process chemists and manufacturing teams alike.
Historically, the separation of these isomers has been fraught with difficulties, often requiring cumbersome protection and deprotection strategies or the use of hazardous solvents. The disclosed technology offers two distinct yet complementary pathways: selective crystallization of novel amine salts and improved chromatographic separation. By leveraging the differential solubility and adsorption properties of the Z and E isomers, this process enables manufacturers to achieve high-purity intermediates with significantly reduced E-isomer content, potentially lowering it to undetectable levels. This technical advancement not only streamlines the synthetic route but also aligns with modern green chemistry principles by eliminating the need for carcinogenic solvents like benzene, which were previously standard in purification protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for isolating the pure Z-isomer of 7-amino-3-[2-(4-methyl-5-thiazolyl)vinyl]-3-cephem-4-carboxylic acid derivatives have relied heavily on chromatographic techniques that pose significant operational and environmental hazards. For instance, earlier literature describes the use of silica gel chromatography with benzene and ethyl acetate mixtures to separate isomers. Benzene is a known human carcinogen, and its use in large-scale pharmaceutical manufacturing is increasingly restricted due to stringent regulatory guidelines regarding residual solvents. Furthermore, these traditional chromatographic methods often suffer from low loading capacities and require vast amounts of solvent, leading to high waste generation and increased disposal costs. Another conventional approach involves synthesizing protected intermediates, separating the isomers at an earlier stage, and then performing multiple deprotection and re-protection steps. This multi-step sequence drastically reduces the overall yield and increases the production timeline, making it economically unviable for commercial scale-up.
The Novel Approach
The methodology presented in CN1107850A represents a paradigm shift by introducing a direct purification strategy that operates on the free acid or its simple salts without the need for complex protecting groups. The core innovation lies in the discovery that specific amine salts of the cephalosporin acid exhibit vastly different crystallization behaviors depending on the stereochemistry of the vinyl side chain. By reacting the Z/E mixture with selected amines such as dicyclohexylamine or t-octylamine in solvents like acetone or alcohols, the Z-isomer preferentially crystallizes out of the solution. This allows for a simple filtration step to physically separate the desired isomer from the E-isomer which remains in the mother liquor. Alternatively, the patent describes an improved chromatographic method using macroporous adsorption resins or activated carbon with aqueous alcohol eluents. This avoids toxic organic solvents entirely and leverages the differential adsorption affinity of the isomers, where the E-isomer is retained longer on the column, allowing the Z-isomer to elute first with high purity.
Mechanistic Insights into Amine Salt Crystallization and Chromatography
The success of the amine salt crystallization method hinges on the subtle stereoelectronic differences between the Z and E isomers of the cephalosporin nucleus. When the carboxylic acid group at the 4-position reacts with a bulky amine, such as dicyclohexylamine, it forms an ammonium carboxylate salt. The spatial arrangement of the 3-vinyl side chain in the Z-configuration allows for a more favorable crystal lattice packing energy compared to the E-configuration. Consequently, the solubility product of the Z-amine salt is significantly lower in specific solvent systems like acetone/water mixtures. As the solution cools or as a counter-solvent is added, the Z-salt reaches supersaturation first and nucleates, effectively pulling the Z-isomer out of the equilibrium mixture. This dynamic equilibrium can be driven further towards the Z-product by repeating the crystallization cycle, progressively enriching the solid phase with the desired stereoisomer while the E-isomer accumulates in the liquid phase.
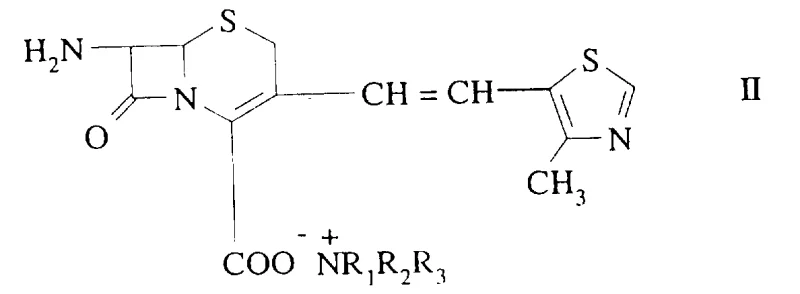
In parallel, the chromatographic mechanism described utilizes the principle of differential partitioning between the mobile phase and the stationary phase. Unlike traditional silica gel which relies on polarity, the use of non-ionic macroporous resins or activated carbon separates the isomers based on hydrophobic interactions and steric fit within the resin pores. The E-isomer, having a more extended conformation due to the trans-double bond, interacts differently with the hydrophobic surface of the resin compared to the more compact Z-isomer. By carefully controlling the pH of the eluent to be near the isoelectric point of the compound (around pH 3.0 to 3.7), the ionization of the carboxylic acid and amino groups is minimized, enhancing the hydrophobic interaction. This results in the E-isomer being retained more strongly on the column, allowing the Z-isomer to pass through with higher velocity. This mechanism ensures that the initial fractions collected are highly enriched in the Z-isomer, providing a continuous and scalable purification process that is far superior to batch crystallization for certain feedstock qualities.
How to Synthesize 7-Amino-3-[2-(4-methyl-5-thiazolyl)vinyl]-3-cephem-4-carboxylic Acid Efficiently
To implement this technology in a laboratory or pilot plant setting, the process begins with dissolving the crude Z/E mixture in a mixture of water and a water-miscible organic solvent such as acetone or ethanol. The choice of solvent is critical; acetone is particularly preferred for its ability to modulate the solubility of the amine salts effectively. Once a homogeneous solution or suspension is achieved at mild temperatures (around 30°C), a stoichiometric amount of the selected amine, such as dicyclohexylamine, is added. The mixture is stirred to ensure complete salt formation, often resulting in a clear solution initially. Crystallization is then induced either by cooling the mixture in an ice bath or by slowly adding a counter-solvent like additional acetone or ether to reduce solubility further. The resulting precipitate is filtered, washed, and dried to yield the purified amine salt. Finally, the salt is dissolved in water and acidified with a mineral acid like sulfuric acid to pH 3.5 to regenerate the free acid form of the cephalosporin, which precipitates as a high-purity solid.
- Dissolve the Z/E mixture of 7-amino-3-[2-(4-methyl-5-thiazolyl)vinyl]-3-cephem-4-carboxylic acid in a suitable solvent system such as acetone/water or alcohol/water.
- Add a stoichiometric or excess amount of a specific amine (e.g., dicyclohexylamine, t-octylamine, or t-butylamine) to form the corresponding amine salt.
- Induce crystallization by cooling or adding a counter-solvent, filter the precipitated Z-enriched amine salt, and regenerate the free acid via acidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this isomer separation technology translates directly into enhanced operational efficiency and risk mitigation. The elimination of benzene from the purification process removes a significant regulatory burden and safety hazard, thereby reducing the costs associated with specialized solvent handling, storage, and hazardous waste disposal. Furthermore, the simplicity of the crystallization process means that it can be performed in standard stainless steel reactors without the need for complex chromatography columns or gradient elution systems, significantly lowering capital expenditure requirements for new production lines. The ability to upgrade lower-quality feedstock containing high levels of E-isomer into pharmaceutical-grade intermediate ensures a more flexible supply chain, allowing manufacturers to utilize a broader range of raw materials without compromising final product quality.
- Cost Reduction in Manufacturing: The process drastically simplifies the downstream processing of cephalosporin intermediates by removing the need for multiple protection and deprotection steps that were previously required to facilitate separation. By avoiding the use of expensive and toxic solvents like benzene, the facility saves substantially on solvent procurement and recycling costs. Additionally, the high recovery yields associated with the crystallization method mean that less raw material is wasted, directly improving the cost-of-goods-sold (COGS) profile for the final API. The regeneration of the amine base from the mother liquor further contributes to cost efficiency by allowing for the reuse of reagents in subsequent batches.
- Enhanced Supply Chain Reliability: Reliance on hazardous solvents often creates bottlenecks in the supply chain due to strict transportation regulations and limited supplier availability. By shifting to a solvent system based on acetone, water, and alcohols, the manufacturing process becomes much more resilient to supply disruptions. The robustness of the crystallization technique also ensures consistent batch-to-batch quality, reducing the likelihood of failed quality control tests that could delay shipments. This reliability is crucial for maintaining uninterrupted production schedules for life-saving antibiotics, ensuring that downstream formulation partners receive their materials on time.
- Scalability and Environmental Compliance: The technologies described are inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production. The use of aqueous-based chromatographic systems or simple crystallization vessels fits well within existing infrastructure, facilitating rapid technology transfer. From an environmental perspective, the reduction in toxic solvent usage and the potential for solvent recycling align with global sustainability goals and ISO 14001 standards. This eco-friendly profile not only minimizes the environmental footprint of the manufacturing site but also enhances the corporate social responsibility (CSR) standing of the organization in the eyes of stakeholders and regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this isomer separation technology. These answers are derived directly from the experimental data and claims found in the patent documentation, providing a reliable basis for process evaluation. Understanding these details is essential for R&D teams planning to integrate this method into their existing workflows and for quality assurance personnel establishing specification limits.
Q: How does the amine salt crystallization method improve isomer purity compared to traditional chromatography?
A: The amine salt method selectively precipitates the Z-isomer salt while leaving the E-isomer in the mother liquor, avoiding the need for toxic solvents like benzene often used in traditional silica gel chromatography.
Q: What specific amines are effective for separating cephalosporin isomers?
A: The patent identifies dicyclohexylamine, t-octylamine, and t-butylamine as particularly effective amines for forming crystalline salts that significantly reduce E-isomer content.
Q: Can this process be scaled for industrial production of Cefditoren intermediates?
A: Yes, the process utilizes standard crystallization and filtration equipment with common solvents like acetone and water, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Amino-3-[2-(4-methyl-5-thiazolyl)vinyl]-3-cephem-4-carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the purity of intermediates like 7-amino-3-[2-(4-methyl-5-thiazolyl)vinyl]-3-cephem-4-carboxylic acid is the foundation of a successful antibiotic campaign. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. We employ stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the Z/E ratio and impurity profile of every batch. Our commitment to quality ensures that the intermediates we supply meet the exacting standards required for the synthesis of high-performance cephalosporins like Cefditoren pivoxil.
We invite pharmaceutical partners to collaborate with us to leverage these advanced purification technologies for their specific supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates how implementing these isomer separation methods can optimize your specific production economics. We encourage potential clients to contact us to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us be your strategic partner in delivering high-quality cephalosporin intermediates that drive the next generation of antimicrobial therapies.
