Advanced Biodegradable Chelating Agents for Industrial Pulp and Paper Chemical Manufacturing
The chemical industry is currently witnessing a significant paradigm shift towards sustainable and biodegradable additives, driven by stringent environmental regulations and the need for greener manufacturing processes. Patent CN1158243C introduces a groundbreaking class of N-bis- or N-tris-[(1,2-dicarboxy-ethoxy)-ethyl]amine derivatives that serve as highly effective chelating agents. These novel compounds are specifically engineered to overcome the limitations of traditional chelators like EDTA and DTPA, which are notorious for their persistence in the environment. The invention details a robust synthetic route utilizing lanthanide catalysis to produce these derivatives, which exhibit superior heavy metal sequestration capabilities in alkaline aqueous solutions. This technological advancement is particularly relevant for industries such as pulp and paper, detergents, and photographic chemistry, where metal ion control is critical for process efficiency and product quality. By integrating these biodegradable structures into industrial workflows, manufacturers can achieve substantial improvements in environmental compliance without sacrificing performance.
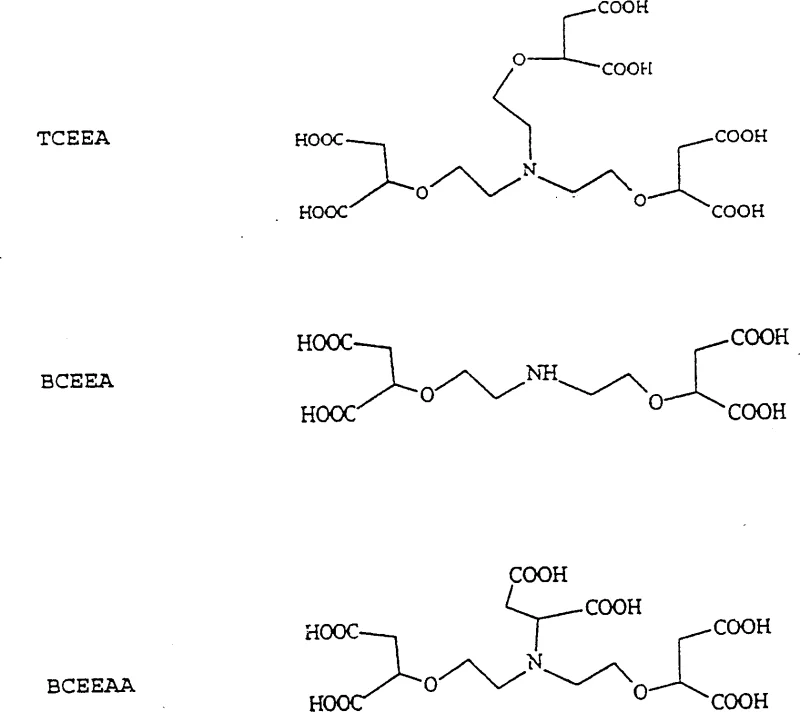
![General chemical formula I of N-bis- or N-tris-[(1,2-dicarboxy-ethoxy)-ethyl]amine derivatives showing variable R groups](/insights/img/n-bis-dicarboxy-ethoxy-ethyl-amine-derivatives-supplier-20260314214819-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the industry has relied heavily on aminopolycarboxylic acids such as ethylenediaminetetraacetic acid (EDTA) and diethylenetriaminepentaacetic acid (DTPA) for sequestering heavy metal ions. While these compounds are effective chelators, their widespread use has led to significant environmental concerns due to their extremely poor biodegradability. In wastewater treatment systems, EDTA and DTPA persist for long periods, facilitating the transport of heavy metals through the environment and potentially contaminating water sources. Furthermore, the regulatory pressure to reduce nitrogen and phosphorus loads in effluent streams is intensifying, making the continued use of these persistent organic pollutants increasingly untenable for forward-thinking manufacturers. The inability of these conventional agents to degrade naturally results in accumulated ecological toxicity, forcing companies to invest heavily in end-of-pipe treatment technologies that drive up operational costs and complicate supply chain logistics.
The Novel Approach
The novel approach presented in patent CN1158243C offers a compelling alternative by introducing N-bis- or N-tris-[(1,2-dicarboxy-ethoxy)-ethyl]amine derivatives that combine high chelation efficiency with biodegradability. These compounds are synthesized through a direct O-alkylation reaction between maleic acid salts and aminoalcohols, catalyzed by lanthanum compounds. This method avoids the complex multi-step syntheses often required for traditional chelators, streamlining the production process. The resulting molecules contain carboxylic acid groups positioned to effectively coordinate with heavy metals while maintaining a molecular structure that is susceptible to biological degradation. This dual functionality ensures that the chelating agent performs its intended role during the industrial process, such as pulp bleaching or detergent formulation, and then breaks down harmlessly afterwards. This represents a significant technological leap towards sustainable industrial chemistry, aligning production capabilities with global environmental goals.
Mechanistic Insights into Lanthanide-Catalyzed O-Alkylation
The core of this innovation lies in the lanthanide-catalyzed O-alkylation of maleate with aminoalcohols, a reaction that proceeds with high selectivity and efficiency under mild conditions. The process begins with the formation of alkali metal or alkaline earth metal salts of maleic acid, which is an exothermic reaction that naturally raises the temperature to a favorable range of 70-90°C. This thermal profile eliminates the need for excessive external heating, thereby reducing energy consumption. The addition of a lanthanum(III) compound, such as lanthanum nitrate or chloride, activates the maleate for nucleophilic attack by the aminoalcohol. The lanthanum ion coordinates with the carboxylate groups, enhancing the electrophilicity of the double bond and facilitating the addition reaction. This catalytic cycle allows for the formation of the ether linkage between the maleate backbone and the aminoalcohol side chains, creating the complex chelating structure in a single synthetic step.
Impurity control is a critical aspect of this synthesis, particularly regarding the removal of the lanthanum catalyst and unreacted starting materials. The patent describes an elegant method for catalyst recovery where the reaction mixture is acidified and treated with oxalic acid. This induces the precipitation of lanthanum oxalate, which can be easily filtered off from the hot solution. This step not only purifies the final product but also allows for the regeneration and reuse of the expensive lanthanum catalyst, significantly improving the economic viability of the process. Furthermore, the reaction conditions are optimized to minimize the formation of by-products such as malic and fumaric acids, ensuring a high-purity profile for the final chelating agent. The ability to control these impurities is essential for applications in sensitive industries like photography or high-grade pulp bleaching, where metal contamination can degrade product quality.
How to Synthesize N-bis-[(1,2-dicarboxy-ethoxy)-ethyl]amine Efficiently
The synthesis of these high-performance chelating agents is designed to be scalable and operationally simple, making it suitable for commercial production facilities. The process utilizes readily available starting materials such as maleic anhydride and diethanolamine or triethanolamine, which are commodity chemicals with stable supply chains. The reaction is conducted in an aqueous medium, which simplifies handling and reduces the need for organic solvents, thereby enhancing safety and environmental performance. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and quality control across different production batches. This streamlined approach allows manufacturers to transition from laboratory scale to industrial production with minimal technical barriers, facilitating rapid adoption of this superior technology.
- Prepare alkali metal or alkaline earth metal salts of maleic acid by reacting maleic anhydride with hydroxides or carbonates in aqueous solution at 70-90°C.
- Add diethanolamine or triethanolamine and a lanthanum(III) catalyst to the basic reaction mixture and stir at 85°C for 10-48 hours.
- Acidify the mixture, precipitate the catalyst with oxalic acid, filter, and isolate the biodegradable chelating agent product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of these biodegradable chelating agents offers significant strategic advantages beyond mere technical performance. The synthesis route eliminates the need for complex protection and deprotection steps common in traditional chelator manufacturing, which drastically simplifies the production workflow. This simplification translates into reduced operational complexity and lower capital expenditure requirements for manufacturing plants. Furthermore, the ability to recycle the lanthanum catalyst mitigates the impact of fluctuating rare earth metal prices, providing greater cost stability over the long term. Companies that integrate this technology can position themselves as leaders in sustainable chemistry, meeting the growing demand from downstream customers for environmentally responsible supply chains. This alignment with sustainability goals can open up new market opportunities and strengthen relationships with key stakeholders who prioritize green procurement policies.
- Cost Reduction in Manufacturing: The streamlined single-step synthesis significantly reduces processing time and energy consumption compared to multi-step conventional methods. By utilizing the exothermic nature of the maleate salt formation, the process minimizes external heating requirements, leading to substantial energy savings. Additionally, the efficient recovery and reuse of the lanthanum catalyst reduce the consumption of expensive raw materials, further driving down the cost of goods sold. The use of aqueous media eliminates the costs associated with solvent recovery and disposal, contributing to a leaner and more cost-effective manufacturing operation that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including maleic anhydride and ethanolamines, are widely produced commodity chemicals with robust global supply networks. This reduces the risk of supply disruptions that are often associated with specialized or niche intermediates. The aqueous nature of the reaction also simplifies logistics and storage requirements, as there is no need for hazardous organic solvents. This reliability ensures consistent production schedules and on-time delivery to customers, which is critical for maintaining trust and competitiveness in the global market. Manufacturers can confidently scale production to meet demand without worrying about the availability of obscure reagents.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory to pilot scales with consistent results. The mild reaction conditions of 75-95°C are easily manageable in large-scale reactors, reducing the engineering challenges associated with high-pressure or high-temperature processes. Moreover, the biodegradability of the final product ensures compliance with increasingly strict environmental regulations regarding wastewater discharge. This reduces the liability and cost associated with environmental remediation and permits, allowing for smoother operations in regions with rigorous ecological standards. The reduced nitrogen content also aids in meeting effluent limitations, facilitating easier regulatory approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chelating agent technology. These answers are derived directly from the patent data and experimental results to provide accurate guidance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating these derivatives into existing industrial processes. The information covers aspects ranging from synthesis mechanics to performance benchmarks against established standards.
Q: Why are these new chelating agents considered more environmentally friendly than EDTA?
A: Unlike EDTA and DTPA, which have extremely poor biodegradability, the N-bis- or N-tris-[(1,2-dicarboxy-ethoxy)-ethyl]amine derivatives described in patent CN1158243C are designed to be biodegradable, significantly reducing environmental pollution and nitrogen load in wastewater.
Q: What is the role of the lanthanum catalyst in this synthesis?
A: The lanthanum(III) compound acts as a highly efficient catalyst for the O-alkylation of maleate with aminoalcohols. It enables a single-step synthesis under mild conditions and can be recycled by precipitation with oxalic acid, enhancing process economics.
Q: Can these chelating agents stabilize alkaline hydrogen peroxide solutions?
A: Yes, experimental data indicates that reaction mixtures containing these derivatives provide significantly greater stability to alkaline hydrogen peroxide compared to conventional agents like DTPA or EDDS, making them ideal for bleaching applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-bis-[(1,2-dicarboxy-ethoxy)-ethyl]amine Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical innovation, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of lanthanide-catalyzed reactions and can ensure that the synthesis of these complex chelating agents meets stringent purity specifications. We operate rigorous QC labs that employ advanced analytical techniques to verify the identity and quality of every batch, ensuring that the biodegradability and chelation performance match the patent specifications. Our commitment to quality and consistency makes us an ideal partner for companies looking to transition to more sustainable chemical solutions without compromising on reliability or performance standards.
We invite you to collaborate with us to optimize your supply chain and reduce your environmental footprint. Contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals. Let us help you engineer a more sustainable and efficient future for your chemical operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
