Advanced Synthesis of Cyclic 1,3-Diketodiimines for High-Purity Solvent Applications
Advanced Synthesis of Cyclic 1,3-Diketodiimines for High-Purity Solvent Applications
The pharmaceutical and fine chemical industries constantly face the challenge of maintaining ultra-high purity in solvents and raw materials, particularly when dealing with sensitive organometallic reagents like Grignard or organolithium compounds. Trace amounts of water or alcohols can deactivate these reagents, leading to significant batch failures and increased costs. Patent CN116410108A introduces a groundbreaking solution through the development of a novel class of cyclic 1,3-diketodiimine compounds. These compounds exhibit exceptional binding capacity for alcohol and water molecules via hydrogen bonding, enabling the reduction of impurity levels to below 50ppm. Unlike previous methods that struggled with sterically hindered substrates, this invention provides a robust synthetic route applicable to a wide range of substituted anilines. As a reliable cyclic 1,3-diketodiimine supplier, understanding the technical nuances of this patent is crucial for integrating these high-performance purification agents into your supply chain.
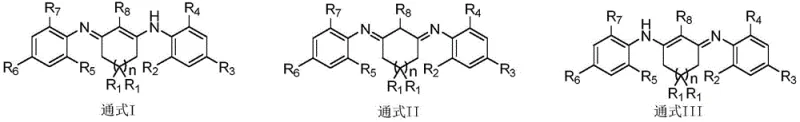
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclic beta-diketodiimine compounds has been fraught with significant technical barriers, particularly when attempting to introduce bulky substituents onto the nitrogen atoms. Conventional literature describes methods using open-chain beta-diketones or simple aliphatic amines, but these approaches often fail when applied to aromatic amines with substantial steric hindrance, such as 2,6-disubstituted anilines. Previous attempts to synthesize these hindered structures using direct condensation or activation via iodoethane, enaminothione intermediates, or boron fluoride ethoxylates have proven unsuccessful, often yielding no reaction even after extending reaction times to excessive durations like 240 hours. Furthermore, traditional solvent purification methods rely heavily on molecular sieves requiring energy-intensive high-temperature calcination or dangerous active metals like sodium metal and lithium aluminum hydride. These legacy processes pose safety risks and operational inefficiencies, highlighting the urgent need for a safer, more chemically selective alternative for cost reduction in specialty chemical manufacturing.
The Novel Approach
The patented methodology overcomes these historical limitations by employing a sophisticated two-step acid-catalyzed condensation strategy that tolerates significant steric bulk. By utilizing specific organic acid catalysts, such as p-toluenesulfonic acid, in high-boiling benzene solvents, the process drives the equilibrium towards the formation of the desired enaminone intermediate and subsequently the final diimine product. This approach successfully enables the incorporation of bulky groups like isopropyl and tert-butyl at the ortho positions of the aniline rings, which was previously unachievable. The resulting cyclic 1,3-diketodiimines possess a unique structural rigidity and electronic environment that facilitates strong hydrogen bonding with small molecules like methanol and water. This innovation not only expands the library of available ligands but also provides a practical, scalable tool for the commercial scale-up of complex organic ligands used in high-purity applications.
Mechanistic Insights into Acid-Catalyzed Condensation and Tautomerism
The core of this synthesis lies in a sequential condensation mechanism that carefully manages reaction thermodynamics and kinetics to prevent side reactions. The first step involves the reaction of a cyclic 1,3-diketone with a substituted aniline to form an enaminone intermediate, a process facilitated by the protonation of the carbonyl oxygen by the organic acid catalyst. This activation increases the electrophilicity of the carbonyl carbon, allowing the nucleophilic attack by the amine nitrogen even in the presence of steric hindrance. The second step is the critical transformation where the enaminone reacts with a second equivalent of substituted aniline under more vigorous conditions, typically involving higher temperatures between 160-230°C in solvents like mesitylene. This elevated thermal energy is essential to overcome the activation barrier imposed by the bulky substituents, ensuring the complete conversion to the cyclic diimine salt before final neutralization.
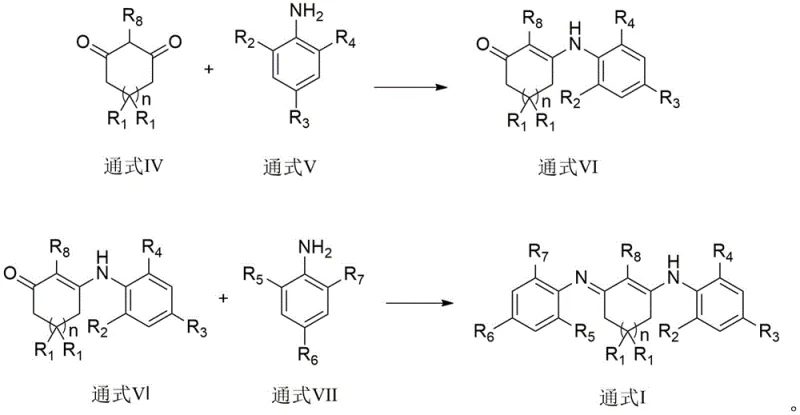
Furthermore, the structural integrity and functionality of the final product are governed by its tautomeric behavior, which is vital for its application as a purification agent. The cyclic 1,3-diketodiimine exists in equilibrium between three tautomeric forms, as illustrated in the patent documentation, allowing the molecule to adapt its hydrogen bonding donors and acceptors to bind impurities effectively. This dynamic structural feature ensures that the compound can trap water and alcohol molecules in a 1:1 stoichiometric ratio within its crystal lattice or solution phase. The stability of these hydrogen-bonded adducts is confirmed by the requirement of vacuum heating above 95°C to release the trapped alcohols, demonstrating the strength of the interaction. For R&D teams, understanding this mechanism is key to optimizing the loading ratios and contact times required to achieve the target purity specifications of less than 50ppm in various solvent systems.
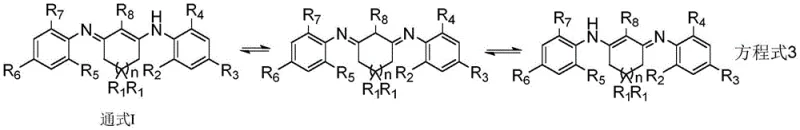
How to Synthesize Cyclic 1,3-Diketodiimine Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for producing these high-value compounds with consistent quality and yield. The process begins with the preparation of the enaminone intermediate by refluxing the cyclic diketone and aniline in toluene with a catalytic amount of acid, followed by a basic workup to isolate the neutral intermediate. The subsequent step involves heating this intermediate with a second aniline derivative in a high-boiling solvent like mesitylene for an extended period, typically ranging from 30 to 120 hours, to ensure full conversion despite steric constraints. Detailed standardized synthesis steps see the guide below.
- Reflux cyclic 1,3-diketone with substituted aniline and organic acid catalyst in benzene solvent to form enaminone intermediate.
- React the enaminone intermediate with a second substituted aniline under high-temperature reflux conditions with acid catalysis.
- Treat the resulting organic acid salt with aqueous alkaline solution to isolate the final cyclic 1,3-diketodiimine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this technology offers substantial strategic benefits beyond mere technical performance. The ability to source high-purity cyclic 1,3-diketodiimines produced via this robust method translates directly into enhanced process reliability and reduced operational risks associated with solvent contamination. By replacing hazardous metal-based drying agents with this stable organic compound, facilities can significantly lower their safety compliance burdens and waste disposal costs. Moreover, the scalability of the synthesis, which relies on commodity chemicals and standard reactor equipment, ensures a stable supply chain capable of meeting large-volume demands without the bottlenecks often associated with specialized reagents.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available organic acids and solvents drastically simplify the production cost structure. Traditional methods often require precious metal complexes or hazardous reagents like lithium aluminum hydride, which incur high procurement and handling costs. By utilizing a metal-free organic synthesis route, the overall manufacturing expense is significantly optimized, allowing for more competitive pricing models for downstream users. Additionally, the high separation yields reported in the examples indicate efficient material utilization, minimizing waste generation and further contributing to substantial cost savings in the production lifecycle.
- Enhanced Supply Chain Reliability: The reliance on common chemical feedstocks such as cyclic diketones and substituted anilines ensures that raw material availability is not a bottleneck for production. Unlike specialized reagents that may have single-source suppliers or long lead times, the precursors for this synthesis are widely manufactured globally. This diversity in the supply base reduces the risk of disruption and allows for flexible sourcing strategies. Furthermore, the robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without extensive requalification, ensuring continuous availability of high-purity intermediates for critical pharmaceutical and chemical processes.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing standard reflux and distillation unit operations that are common in fine chemical plants. The absence of heavy metals simplifies the environmental footprint, as there is no need for complex heavy metal removal steps or specialized waste treatment for toxic residues. The final product's ability to remove alcohols to very low levels (<50ppm) also enhances the quality of the end-user's product, potentially reducing their own rejection rates and rework costs. This alignment with green chemistry principles and operational efficiency makes the technology highly attractive for long-term partnerships focused on sustainable and scalable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this cyclic 1,3-diketodiimine technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: This method successfully synthesizes cyclic 1,3-diketodiimines with bulky steric hindrance (e.g., 2,6-disubstituted anilines), which conventional methods fail to produce even after extended reaction times.
Q: How effective is the compound in removing impurities?
A: The compound forms strong hydrogen bonds with water and alcohols, reducing their content in solvents or raw materials to below 50ppm after simple filtration.
Q: Is the process scalable for industrial production?
A: Yes, the process uses common organic acid catalysts and standard reflux conditions in benzene solvents, making it highly suitable for commercial scale-up without requiring exotic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclic 1,3-Diketodiimine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity additives play in the success of modern chemical synthesis. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Whether you require custom derivatives of the cyclic 1,3-diketodiimine scaffold or standard grades for solvent purification, our infrastructure is designed to support your specific technical requirements with precision and consistency.
We invite you to collaborate with us to explore how this innovative technology can optimize your operations. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current solvent purification challenges. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our high-purity cyclic 1,3-diketodiimines can enhance your product quality and operational efficiency.
