Advanced Manufacturing of Non-neridone Intermediates for Cardiovascular Therapeutics
Introduction to Next-Generation Non-neridone Synthesis
The escalating global demand for effective mineralocorticoid receptor antagonists (MRAs) in treating heart failure and chronic kidney disease has placed immense pressure on supply chains to deliver high-purity active pharmaceutical ingredients (APIs) efficiently. Patent CN116082334A introduces a transformative preparation method for Non-neridone and its critical intermediates, addressing the longstanding bottlenecks of low yield and complex purification found in legacy processes. This technical disclosure outlines a robust synthetic route that leverages strategic chiral resolution and optimized cyclization reactions to achieve superior purity profiles. By shifting away from chromatography-dependent purifications toward crystallization-driven isolation, this methodology offers a compelling value proposition for manufacturers seeking to enhance throughput while maintaining rigorous quality standards. The following analysis details how this innovation aligns with the needs of modern pharmaceutical production.
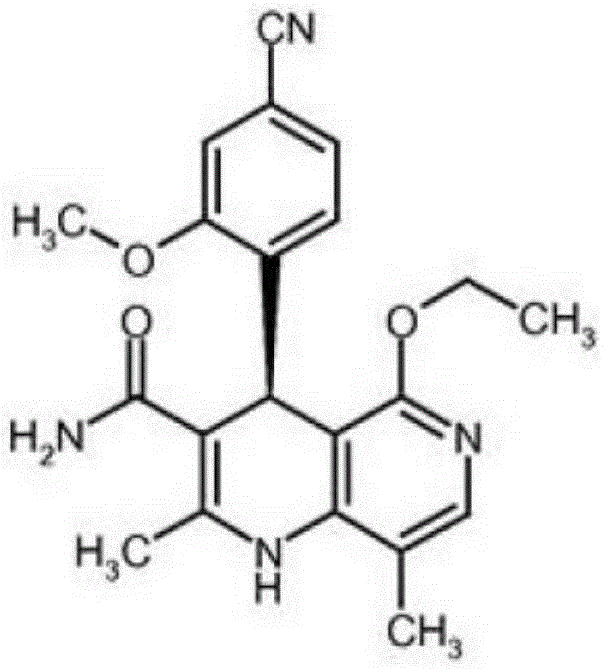
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Non-neridone has been plagued by inefficiencies that render it economically unviable for large-scale commercialization. Prominent prior art, such as WO2008/104306, describes a laborious 10-step sequence starting from vanillin, which culminates in a dismal total yield of merely 3.76%. Such low efficiency not only inflates the cost of goods sold (COGS) due to excessive raw material consumption but also generates significant chemical waste, complicating environmental compliance. Furthermore, earlier methods often relied heavily on chiral chromatography, utilizing specialized phases like Chiralpak AS-V or polymeric selectors, which are prohibitively expensive and difficult to scale beyond kilogram quantities. The reliance on spectral purification and the generation of colored impurities in intermediate stages further necessitated complex downstream processing, creating a fragile supply chain vulnerable to delays and quality deviations.
The Novel Approach
In stark contrast, the methodology disclosed in CN116082334A presents a streamlined, industrially viable pathway that fundamentally re-engineers the synthesis logic. The core innovation lies in the early introduction of a highly efficient chiral resolution step using D-diphenyl tartrate, which replaces costly chromatographic separations with straightforward crystallization. This approach not only simplifies the operational workflow but also ensures that chirality is established early, preventing the propagation of racemic impurities through subsequent steps. The route integrates a concise sequence of condensation, cyclization, and functional group transformations that maximize atom economy. By optimizing reaction conditions—such as utilizing specific alcohol solvents for cyclization and controlling temperature gradients during crystallization—the process achieves high purity without the need for extensive solvent swaps or hazardous reagents. This represents a paradigm shift towards greener, more cost-effective API manufacturing.
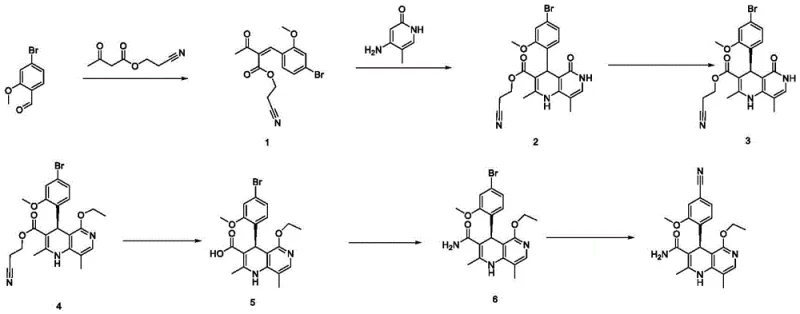
Mechanistic Insights into Chiral Resolution and Pd-Catalyzed Cyanation
The cornerstone of this synthesis is the enantioselective resolution of the racemic intermediate 2 using D-diphenyl tartrate. Mechanistically, this process relies on the formation of diastereomeric salts between the basic amine functionality of the intermediate and the chiral acid. Due to the distinct physical properties, specifically solubility differences in halogenated hydrocarbon solvents like methylene dichloride, one diastereomer preferentially crystallizes out of the solution. This thermodynamic control allows for the isolation of the desired enantiomer with exceptional optical purity, often exceeding 99.5% ee after a single crystallization cycle. The subsequent acid-base liberation step using mild alkalis like sodium phosphate ensures the gentle recovery of the free base intermediate 3 without risking racemization or degradation of the sensitive naphthyridine core. This precise control over stereochemistry is critical, as the biological activity of Non-neridone is strictly dependent on the (4S) configuration.
Furthermore, the final transformation involves a palladium-catalyzed nucleophilic substitution to install the critical cyano group. This step utilizes zinc cyanide as the cyanide source in the presence of ligands such as 1,1'-bis(diphenylphosphino)ferrocene (dppf) or tetrakis(triphenylphosphine)palladium(0). The mechanism proceeds through an oxidative addition of the aryl bromide to the Pd(0) center, followed by transmetallation with the cyanide species and reductive elimination to forge the C-CN bond. The choice of polar aprotic solvents like DMF facilitates the solubility of the inorganic cyanide salt, while the specific catalyst loading (0.05 to 0.2 molar ratio) balances reaction rate with metal residue control. This robust catalytic cycle ensures high conversion rates even on multi-kilogram scales, minimizing the presence of unreacted bromo-impurities that could complicate final purification.
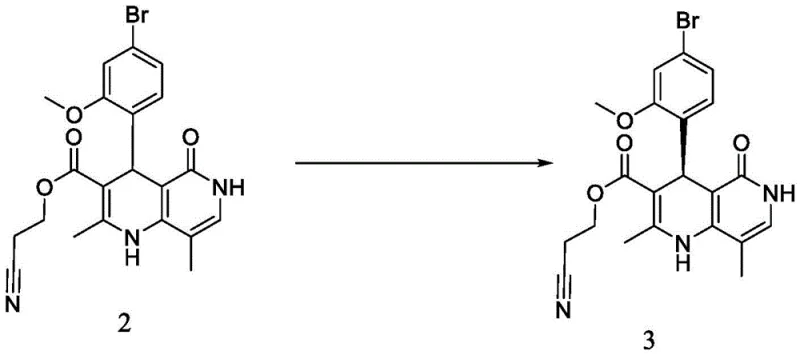
How to Synthesize Non-neridone Intermediates Efficiently
The execution of this synthesis requires precise adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the condensation of 4-bromo-2-methoxybenzaldehyde and ethyl 2-cyanoacetoacetate, followed by a thermal cyclization to build the core heterocyclic scaffold. Critical attention must be paid to the resolution step, where temperature control between 20°C and 40°C is vital for maximizing the yield of the diastereomeric salt. Subsequent functionalization involves nucleophilic substitution with triethyl orthoformate and hydrolysis to generate the carboxylic acid precursor. The detailed standardized operating procedures, including specific solvent volumes, stirring rates, and crystallization cooling profiles, are essential for maintaining the high purity specifications required for pharmaceutical grade materials.
- Perform resolution reaction of racemic intermediate 2 with D-diphenyl tartrate in a halogenated hydrocarbon solvent like methylene dichloride at 20-40°C to form the diastereomeric salt.
- Isolate the resolution salt via filtration and subject it to an acid-base reaction using an alkali such as sodium phosphate to liberate the free base intermediate 3.
- Purify the resulting intermediate 3 through controlled crystallization and washing steps to achieve chiral purity exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the purification train. By eliminating the need for preparative chiral chromatography, manufacturers can significantly reduce capital expenditure on specialized equipment and lower the recurring costs associated with expensive chiral stationary phases. This shift to crystallization-based purification not only accelerates batch turnover times but also reduces the volume of organic solvents required, leading to substantial cost reduction in API manufacturing. Additionally, the use of readily available, commodity-grade reagents such as D-diphenyl tartrate and zinc cyanide enhances supply chain reliability, mitigating the risk of shortages associated with bespoke or highly specialized catalysts.
- Cost Reduction in Manufacturing: The process achieves higher overall yields compared to prior art, which intrinsically lowers the raw material cost per kilogram of finished product. By avoiding low-yielding steps and complex chromatographic separations, the operational expenditure is significantly optimized. The ability to recycle solvents like isopropanol and toluene further contributes to a leaner cost structure, making the final API more competitive in the global market without compromising on quality margins.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials ensures a resilient supply chain. Unlike processes dependent on custom-synthesized chiral selectors or unstable intermediates, this route utilizes robust chemistry that can be sourced from multiple vendors. This diversification of the supplier base reduces lead time for high-purity intermediates and safeguards against single-source disruptions, ensuring continuous availability for downstream drug formulation.
- Scalability and Environmental Compliance: The simplified post-treatment steps, primarily involving filtration and washing rather than complex extractions or column chromatography, make this process highly amenable to commercial scale-up of complex pharmaceutical intermediates. The reduced solvent intensity and the avoidance of heavy metal contaminants (through efficient catalyst removal protocols) align with stringent environmental regulations, facilitating easier permitting and waste management in GMP facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational requirements. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does this new process improve upon the prior art WO2008/104306?
A: The prior art relied on a lengthy 10-step synthesis from vanillin with a total yield of only 3.76% and required complex chromatographic purification. The new method described in CN116082334A significantly reduces step count, utilizes crystallization-based purification instead of chromatography, and achieves substantially higher overall yields suitable for industrial scale.
Q: What is the key advantage of the chiral resolution step using D-diphenyl tartrate?
A: The use of D-diphenyl tartrate allows for the formation of diastereomeric salts that can be separated via simple crystallization. This avoids the need for expensive chiral HPLC columns or simulated moving bed (SMB) chromatography mentioned in other patents, drastically lowering production costs and simplifying equipment requirements.
Q: Is the final cyanation step safe for large-scale manufacturing?
A: Yes, the process utilizes zinc cyanide with a palladium catalyst under controlled conditions (90-140°C). While cyanides require strict safety protocols, the use of solid zinc cyanide is generally manageable in standard pharmaceutical facilities equipped with appropriate containment and waste treatment systems, making it viable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Non-neridone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the supply of life-saving cardiovascular medications. Our team of expert chemists has extensively analyzed the methodology described in CN116082334A and possesses the technical capability to execute this complex sequence with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity and residual solvent limits, guaranteeing that every batch meets the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can be integrated into your supply chain. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to validate the commercial potential of this high-efficiency synthesis. Together, we can accelerate the delivery of high-quality Non-neridone to patients worldwide.
