Scalable Synthesis of N-Substituted Phthalimides: A Breakthrough in Process Efficiency and Purity
Scalable Synthesis of N-Substituted Phthalimides: A Breakthrough in Process Efficiency and Purity
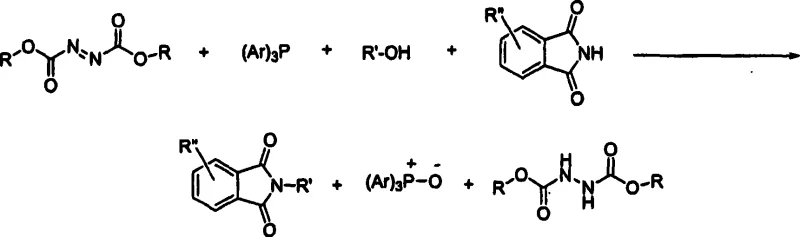
The pharmaceutical and fine chemical industries rely heavily on N-substituted phthalimides as versatile intermediates for synthesizing complex organic molecules, including active pharmaceutical ingredients (APIs). However, the conventional synthetic routes, particularly the classic Mitsunobu reaction, have long been plagued by significant downstream processing challenges that hinder industrial scalability. Patent CN1882539A introduces a transformative methodology that addresses these bottlenecks by optimizing solvent systems and workup procedures. This innovation allows for the direct precipitation of high-purity products from aromatic solvents, effectively bypassing the need for resource-intensive purification techniques like column chromatography or distillation. For R&D directors and process chemists, this represents a critical advancement in designing robust, scalable pathways for drug substance manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted phthalimides via the Mitsunobu reaction has been constrained by the generation of stoichiometric amounts of difficult-to-remove byproducts, specifically triphenylphosphine oxide and dialkyl hydrazinedicarboxylates. In traditional protocols utilizing halogenated solvents such as dichloromethane or chloroform, these byproducts often co-elute with the desired product or form emulsions during aqueous workups, necessitating complex purification strategies. The reliance on chromatography not only escalates manufacturing costs due to high solvent consumption and silica gel usage but also creates substantial logistical bottlenecks in large-scale production environments. Furthermore, the use of chlorinated solvents poses increasing environmental and regulatory compliance burdens, forcing manufacturers to seek greener alternatives that do not compromise yield or purity profiles.
The Novel Approach
The methodology described in CN1882539A fundamentally reengineers the reaction environment by substituting problematic halogenated solvents with aromatic solvents, such as toluene or benzene. This strategic solvent switch alters the solubility profile of the reaction components, enabling a unique isolation strategy where the target N-substituted phthalimide can be selectively precipitated. By introducing a non-solvent like methanol or ethanol directly into the reaction mixture post-conversion, the product crashes out as a solid while the triphenylphosphine oxide and excess reagents remain dissolved in the mother liquor. This elegant simplification transforms a multi-step purification nightmare into a single filtration operation, drastically reducing the operational complexity and enhancing the overall throughput of the manufacturing process.
Mechanistic Insights into Modified Mitsunobu Cyclization
The core of this innovation lies in the precise manipulation of solubility parameters within the Mitsunobu reaction matrix. While the fundamental mechanistic steps involving the activation of the alcohol by the phosphine-azodicarboxylate complex remain consistent with established organic chemistry principles, the choice of toluene as the reaction medium is pivotal. Toluene provides an optimal balance of polarity that supports the formation of the betaine intermediate while ensuring that the resulting phthalimide product exhibits limited solubility upon the addition of polar alkanols. This differential solubility is the key driver for the high purity observed, as the crystal lattice of the precipitating product naturally excludes the soluble phosphine oxide impurities, acting as a self-purifying crystallization event.
Furthermore, the control of reaction temperature plays a critical role in managing the exothermic nature of the azodicarboxylate addition and ensuring high selectivity. The patent specifies maintaining the reaction mixture between -10°C and 30°C during reagent addition, which minimizes side reactions and decomposition of the sensitive azo species. By carefully regulating the addition rate of the diisopropyl azodicarboxylate (DIAD), the process maintains thermal stability, preventing the formation of colored impurities that often complicate downstream processing. This thermal control, combined with the aromatic solvent system, ensures that the final isolated material consistently achieves purity levels exceeding 95% without the need for recrystallization, demonstrating a high degree of process robustness suitable for GMP manufacturing.
How to Synthesize N-Substituted Phthalimides Efficiently
The implementation of this synthesis route requires careful attention to reagent addition sequences and temperature control to maximize the benefits of the precipitation workup. The process begins with the suspension of phthalimide and the alcohol substrate in toluene, followed by the addition of triphenylphosphine. The critical step involves the slow, temperature-controlled addition of the azodicarboxylate to initiate the coupling. Once the reaction is complete, the addition of methanol induces immediate precipitation, allowing for rapid isolation via filtration. For detailed standard operating procedures and specific parameter ranges, please refer to the technical guide below.
- Mix phthalimide, alcohol substrate, and triphenylphosphine in an aromatic solvent like toluene.
- Add diisopropyl azodicarboxylate (DIAD) at controlled low temperatures (-10 to 10°C) to manage exotherm.
- Precipitate the product by adding methanol directly to the reaction mixture, then filter to obtain >95% pure material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented process offers tangible economic and operational benefits that extend beyond simple yield improvements. The most significant advantage is the drastic reduction in processing time and resource allocation associated with purification. By eliminating the need for column chromatography, manufacturers can significantly reduce the consumption of expensive silica gel and vast quantities of elution solvents, leading to substantial cost savings in raw materials and waste disposal. Additionally, the shift away from halogenated solvents like dichloromethane to commodity aromatics like toluene simplifies solvent recovery and aligns with increasingly stringent environmental regulations, reducing the risk of regulatory delays and compliance costs.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps represents a major driver for cost efficiency in pharmaceutical intermediate production. Traditional methods often require multiple passes over silica or extensive distillation to remove triphenylphosphine oxide, which consumes significant energy and labor. This new approach consolidates the workup into a simple filtration and wash step, thereby lowering the overall cost of goods sold (COGS) and improving the margin profile for high-volume intermediates.
- Enhanced Supply Chain Reliability: Utilizing widely available aromatic solvents and standard reagents enhances the resilience of the supply chain against raw material shortages. Unlike specialized ether solvents that may pose peroxide hazards or require specific storage conditions, toluene is a bulk commodity chemical with a stable global supply. This reliability ensures consistent production schedules and reduces the lead time for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to maintain steady inventory levels without disruption.
- Scalability and Environmental Compliance: The simplicity of the precipitation-based isolation makes this process inherently scalable from kilogram to multi-ton production scales without the engineering complexities associated with large-scale chromatography. The reduction in hazardous waste generation, particularly the avoidance of chlorinated solvent waste streams, simplifies effluent treatment and lowers the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only improves corporate sustainability metrics but also facilitates smoother regulatory approvals for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: How does this process improve upon traditional Mitsunobu reactions?
A: Traditional methods often require halogenated solvents and difficult chromatographic purification to remove triphenylphosphine oxide. This patented process uses toluene and simple alcohol precipitation to achieve high purity without chromatography.
Q: What represents the primary cost saving in this manufacturing route?
A: The elimination of column chromatography and distillation steps significantly reduces processing time and solvent consumption, while the use of non-halogenated solvents lowers waste disposal costs.
Q: Is this method suitable for large-scale production?
A: Yes, the protocol is specifically designed for scalability, utilizing common industrial solvents like toluene and simple filtration for isolation, making it ideal for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted Phthalimides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthetic routes in the modern pharmaceutical landscape. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN1882539A are translated into reliable supply chains. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-substituted phthalimides delivered meets the highest quality standards required for drug substance manufacturing.
We invite global partners to leverage our technical expertise to optimize their intermediate sourcing strategies. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the most advanced and cost-effective manufacturing technologies available in the market.
