Strategic Manufacturing of High-Purity 13C 15N Double-Labeled Glycine for Global Metabolic Research
Strategic Manufacturing of High-Purity 13C 15N Double-Labeled Glycine for Global Metabolic Research
The disclosed methodology in patent CN101914030B represents a significant paradigm shift in the manufacturing of stable isotope-labeled amino acids, specifically targeting the high-value niche of dual-labeled glycine derivatives essential for advanced metabolic flux analysis. By leveraging a robust two-step synthetic pathway that initiates with the alpha-bromination of 13C-enriched acetic acid, the process effectively circumvents the severe safety hazards and isotopic dilution issues inherent in legacy technologies such as the Schmidt or Gabriel methods. This approach is particularly critical for R&D directors overseeing plant nutrition studies and proteomic research, where the integrity of both the carbon and nitrogen labels is paramount for accurate mass spectrometry data interpretation. The strategic selection of trifluoroacetic anhydride as an activating agent ensures high conversion rates while maintaining the isotopic fidelity required for premium-grade research reagents, thereby establishing a new benchmark for reliability in the supply of complex isotopic standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dual-labeled glycine has been plagued by significant technical and safety barriers that hindered efficient commercial scale-up of complex isotopic compounds. The traditional Schmidt method, for instance, relies on the reaction of fatty acid halides with hydrazoic acid, a process notorious for its low yields and the generation of hazardous nitrogen gas byproducts that complicate reactor safety protocols. Furthermore, this legacy route often suffers from the unintended loss of the valuable 13C label during the rearrangement steps, leading to suboptimal isotopic abundance that fails to meet the rigorous specifications of modern analytical instrumentation. Alternatively, the improved Gabriel method necessitates the use of 13C-labeled sodium cyanide, a precursor that is not only prohibitively expensive due to strict regulatory controls on cyanide salts but also presents extreme toxicity risks that escalate operational costs for waste treatment and personnel safety monitoring.
The Novel Approach
In stark contrast to these cumbersome predecessors, the novel approach detailed in the patent utilizes a streamlined sequence starting from readily available 13C-labeled acetic acid, transforming it into a reactive bromo-intermediate before coupling with U-15N labeled hexamethylenetetramine. This strategic pivot eliminates the need for explosive hydrazoic acid or highly toxic cyanide salts, thereby drastically simplifying the safety infrastructure required for production facilities. The reaction conditions are remarkably mild, operating within a temperature range of 40 to 80°C and utilizing ethanol as a benign solvent system, which facilitates easier solvent recovery and reduces the environmental footprint of the manufacturing process. By achieving yields greater than or equal to 85 percent with isotopic abundances exceeding 99 percent for both 13C and 15N, this method offers a commercially viable pathway that aligns perfectly with the demand for cost reduction in metabolic tracer manufacturing without compromising on the stringent purity profiles demanded by top-tier pharmaceutical research.
Mechanistic Insights into Bromination and Nucleophilic Substitution
The core chemical transformation driving this synthesis involves a carefully controlled alpha-bromination of the carboxylic acid followed by a nucleophilic substitution that installs the nitrogen label with high precision. In the first stage, trifluoroacetic anhydride acts as a potent dehydrating and activating agent, facilitating the formation of an mixed anhydride intermediate that enhances the acidity of the alpha-protons on the 13C-acetic acid. This activation allows for the selective introduction of the bromine atom using molecular bromine, a reaction that proceeds efficiently at temperatures between 40 and 80°C to yield 13C-labeled monobromoacetic acid. The subsequent step leverages the nucleophilic character of the nitrogen atoms within the hexamethylenetetramine cage, which attack the electrophilic carbon of the bromoacetic acid derivative. This substitution reaction, conducted in an ethanol solution maintained at a pH of 7 to 10, ensures that the 15N label is incorporated directly into the amino group of the final glycine structure while minimizing side reactions such as hydrolysis or polymerization that could degrade the isotopic purity.
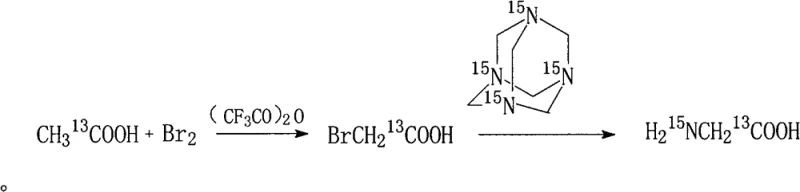
Furthermore, the mechanism inherently supports superior impurity control, a critical factor for R&D directors concerned with the interference of unlabeled species in sensitive NMR or MS assays. The use of hexamethylenetetramine as the nitrogen source provides a protected amine equivalent that prevents over-alkylation, a common issue when using free ammonia or primary amines in similar substitutions. The final purification via ethanol recrystallization capitalizes on the differential solubility of the glycine product compared to unreacted starting materials and inorganic salts, effectively stripping away residual bromide ions and organic byproducts. This physical purification step, combined with the high selectivity of the chemical transformation, ensures that the final product meets the specification of greater than 99 percent chemical purity, thereby reducing the burden on downstream analytical validation and ensuring consistent performance in biological tracing applications where signal-to-noise ratios are critical.
How to Synthesize 13C 15N Double-Labeled Glycine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing operational simplicity and reproducibility across different batches of high-purity stable isotope labeled amino acids. The process begins with the preparation of the bromo-intermediate under cooled conditions to manage the exotherm of bromination, followed by a heating phase to drive the reaction to completion over a period of 2 to 24 hours depending on the specific substrate loading. Following the isolation of the bromo-acid, the second stage involves mixing with the 15N-labeled hexamine in ethanol, where pH control using sodium hydroxide is vital to maintain the nucleophilicity of the amine source without degrading the sensitive ester or acid functionalities. Detailed standardized synthetic steps see the guide below for precise molar ratios and workup procedures that guarantee the high yields reported in the examples.
- Prepare 13C-labeled monobromoacetic acid by reacting 13C-acetic acid with bromine and trifluoroacetic anhydride at 40-80°C.
- React the resulting 13C-bromoacetic acid with U-15N-hexamethylenetetramine in ethanol at pH 7-10 and 40-80°C.
- Purify the crude product via hot filtration, ethanol washing, and recrystallization to obtain high-purity dual-labeled glycine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits that extend beyond mere technical feasibility, addressing critical pain points related to raw material sourcing and regulatory compliance. By shifting the feedstock base from restricted and hazardous cyanides to commodity-grade acetic acid derivatives, the supply chain becomes significantly more resilient against market volatility and regulatory shutdowns that frequently affect facilities handling Schedule 1 or highly toxic chemicals. The elimination of heavy metal catalysts or complex transition metal systems further simplifies the waste stream profile, allowing for more straightforward disposal protocols and reducing the overall environmental compliance costs associated with hazardous waste management. This streamlined material flow ensures that production timelines are not delayed by the lengthy procurement cycles often associated with specialized isotopic precursors, thereby enhancing the overall reliability of the supply chain for time-sensitive research projects.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the substitution of expensive 13C-sodium cyanide with more affordable 13C-acetic acid, which is available from a broader range of global suppliers. The avoidance of complex protection-deprotection sequences and the reliance on simple recrystallization for purification significantly lower the operational expenditure related to solvent consumption and energy usage. Additionally, the high yield of greater than or equal to 85 percent maximizes the utilization of the costly isotopic starting materials, ensuring that minimal value is lost to side products or inefficient conversion steps. These factors combine to create a manufacturing process that delivers substantial cost savings while maintaining the high quality standards required for premium isotopic reagents.
- Enhanced Supply Chain Reliability: The reliance on robust, well-understood chemical transformations such as bromination and nucleophilic substitution means that the process can be easily transferred between different manufacturing sites without extensive re-validation. The raw materials, including trifluoroacetic anhydride and ethanol, are bulk commodities with stable supply chains, reducing the risk of production stoppages due to raw material shortages. This stability is crucial for maintaining continuous supply to research institutions and pharmaceutical partners who depend on consistent availability of labeled standards for long-term longitudinal studies. The simplified process flow also reduces the dependency on specialized equipment, allowing for greater flexibility in production scheduling and capacity allocation.
- Scalability and Environmental Compliance: The process is explicitly designed for batch production, utilizing standard reactor configurations that can be scaled from kilogram to multi-ton quantities without fundamental changes to the chemistry. The absence of gaseous byproducts like nitrogen gas (from the Schmidt method) simplifies reactor pressure management and venting requirements, making scale-up safer and more predictable. Furthermore, the use of ethanol as the primary solvent aligns with green chemistry principles, facilitating solvent recycling and reducing the environmental impact of the manufacturing operation. This alignment with sustainability goals is increasingly important for corporate procurement policies that prioritize environmentally responsible suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these dual-labeled compounds, derived directly from the specific advantages and data points presented in the patent documentation. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this material into their existing research pipelines or supply networks. The answers reflect the proven capabilities of the described method to overcome historical limitations in isotopic synthesis.
Q: What are the primary advantages of this synthesis method over the Schmidt method?
A: Unlike the Schmidt method which utilizes hazardous hydrazoic acid and often results in the loss of the 13C label through decarboxylation, this patented route preserves isotopic integrity with abundances ≥99% while eliminating severe safety risks associated with explosive intermediates.
Q: How does this process impact the cost of goods for stable isotope-labeled amino acids?
A: By utilizing 13C-acetic acid instead of expensive and toxic 13C-sodium cyanide required in the Gabriel method, the process significantly lowers raw material procurement costs and simplifies regulatory compliance for waste disposal.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent explicitly states the process is suitable for mass production, achieving yields ≥85% with simple unit operations like bromination, filtration, and recrystallization that are easily scalable in standard chemical reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 13C 15N Double-Labeled Glycine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-fidelity isotopic tracers play in advancing drug discovery and agricultural science, which is why we have invested heavily in mastering complex labeling chemistries like the one described in CN101914030B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the fluctuating demands of global research initiatives with unwavering consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art mass spectrometry and NMR instrumentation to verify that every batch of 13C 15N double-labeled glycine meets the ≥99 percent abundance and purity thresholds required for sensitive analytical applications. Our commitment to quality assurance ensures that your research data remains uncompromised by isotopic impurities or chemical contaminants.
We invite you to collaborate with us to optimize your supply chain for stable isotope-labeled intermediates, leveraging our technical expertise to secure a competitive advantage in your field. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in metabolic research and pharmaceutical development.
