Advanced Synthetic Method for Phenylethynyl Iodide: Commercial Scale-Up and Process Optimization
The landscape of organic synthesis for high-value coupling partners is constantly evolving, driven by the relentless demand for more efficient and cost-effective pathways in the pharmaceutical and agrochemical sectors. Patent CN107501036B introduces a transformative approach to the preparation of phenylethynyl iodine compounds, which serve as critical synthons in the construction of complex molecular architectures. These alkynyl iodides are indispensable precursors for carbon-carbon bond formation, particularly in Sonogashira coupling reactions, yet their traditional synthesis often suffers from prohibitive costs and operational complexities. This new methodology leverages a zinc diiodide and tert-butyl nitrite system to achieve high yields under remarkably mild conditions, addressing the urgent need for a reliable phenylethynyl iodide supplier who can deliver quality without compromising on economic feasibility. The structural versatility of this method is evident in its ability to accommodate a broad spectrum of substituents, as illustrated by the general substrate framework below.
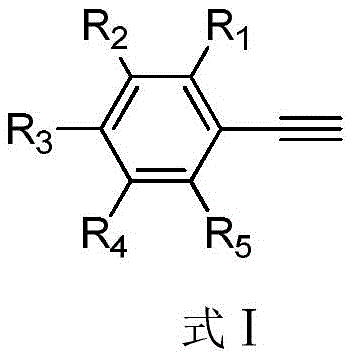
For R&D directors evaluating process viability, the adaptability of the starting material is paramount. The patent specifies that the phenylacetylene reaction substrate can bear various functional groups at the ortho, meta, and para positions, including C1-C5 alkyl, alkoxy, nitro, cyano, and even halogen substituents. This wide substrate scope ensures that the process is not limited to simple model compounds but is robust enough for the synthesis of complex, functionalized intermediates required in modern drug discovery. By enabling the direct iodination of commercially available alkynes, this technology bypasses multi-step sequences that traditionally erode overall yield and increase production timelines. Consequently, this represents a significant strategic advantage for companies aiming to streamline their supply chain for high-purity alkynyl iodides while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkynyl iodides has relied heavily on reagents such as N-Iodosuccinimide (NIS), elemental iodine, or iodide salts combined with strong oxidizing agents. While these methods are documented in literature, they present substantial drawbacks when translated to an industrial setting. The use of NIS, for instance, introduces succinimide byproducts that require extensive purification efforts, thereby increasing solvent consumption and waste disposal costs. Furthermore, many conventional oxidative iodination protocols suffer from poor atom economy, where a significant portion of the reagent mass ends up as waste rather than incorporated into the final product. Operational safety is another concern, as some traditional methods require harsh conditions or generate hazardous intermediates that complicate handling and storage. These inefficiencies collectively contribute to higher manufacturing costs and longer lead times, creating bottlenecks for procurement managers seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in CN107501036B utilizes a combination of zinc diiodide (ZnI2) and tert-butyl nitrite (TBN) in the presence of an organic base. This system operates effectively at temperatures ranging from 0°C to 25°C, eliminating the need for energy-intensive heating or cryogenic cooling. The reagents employed are not only inexpensive and readily available on the global market but also exhibit high stability, simplifying inventory management for supply chain heads. The reaction mechanism avoids the formation of difficult-to-remove byproducts associated with succinimide-based reagents, resulting in a cleaner crude reaction mixture. This simplicity in operation and workup translates directly into enhanced process efficiency, allowing for the commercial scale-up of complex organic synthons with minimal technical risk. The transition from legacy methods to this zinc-mediated protocol represents a paradigm shift towards greener and more economically sustainable chemical manufacturing.
Mechanistic Insights into ZnI2-Mediated Iodination
Understanding the mechanistic underpinnings of this transformation is crucial for assessing its robustness and reproducibility. The reaction likely proceeds through a radical pathway initiated by the decomposition of tert-butyl nitrite, which generates reactive nitrogen oxide species capable of activating the zinc diiodide complex. This activation facilitates the electrophilic iodination of the terminal alkyne triple bond with high regioselectivity. The presence of the organic base, such as triethylamine or DIPEA, plays a critical role in neutralizing acidic byproducts and maintaining the optimal pH environment for the catalytic cycle to proceed uninterrupted. Unlike transition metal-catalyzed couplings that may leave trace metal impurities requiring stringent removal, this method relies on main group elements that are easier to manage in a GMP environment. The mild thermal conditions prevent the polymerization or decomposition of sensitive alkyne substrates, ensuring that the integrity of the molecular scaffold is preserved throughout the synthesis.
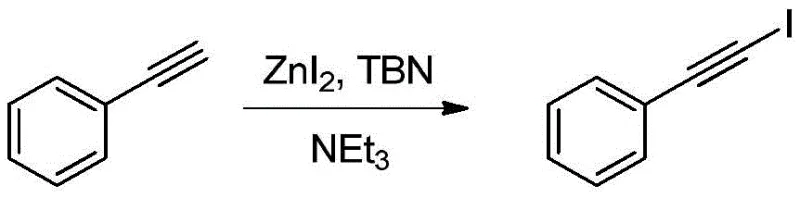
Impurity control is a primary concern for R&D teams, especially when the final product is destined for pharmaceutical applications. The specificity of the ZnI2/TBN system minimizes side reactions such as over-iodination or oxidative degradation of the aromatic ring. The patent data indicates that the reaction can be monitored easily via thin-layer chromatography (TLC), allowing for precise endpoint determination to prevent over-reaction. Furthermore, the workup procedure involves a simple aqueous wash with sodium thiosulfate to quench any remaining oxidants, followed by standard extraction and drying. This streamlined purification process significantly reduces the burden on downstream processing units. By avoiding the use of heavy metal catalysts, the process inherently lowers the risk of metal contamination, aligning with the increasingly strict regulatory guidelines regarding elemental impurities in drug substances. This mechanistic clarity provides confidence in the consistency and reliability of the manufacturing process.
How to Synthesize Phenylethynyl Iodide Efficiently
To implement this synthesis effectively, operators must adhere to strict protocols regarding atmospheric control and reagent stoichiometry. The reaction requires an environment free from oxygen and moisture, typically achieved by purging the reactor with nitrogen or argon gas before introducing the reagents. The precise molar ratios specified in the patent, such as a 1:1.1 ratio of substrate to zinc diiodide, are critical for maximizing yield while minimizing excess reagent waste. Detailed standard operating procedures (SOPs) should be established to govern the addition rates and temperature profiles to ensure safety and reproducibility. For a comprehensive breakdown of the operational parameters and safety precautions, please refer to the standardized synthesis guide provided below.
- Prepare the reaction environment by establishing an inert atmosphere using nitrogen or argon gas to prevent oxidation of sensitive reagents.
- Dissolve the organic base, zinc diiodide, and tert-butyl nitrite in a dry chlorinated solvent such as trichloromethane or dichloromethane.
- Add the phenylacetylene substrate at 0-25°C, monitor via TLC until completion, then quench with sodium thiosulfate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling benefits for procurement managers and supply chain stakeholders focused on efficiency and margin improvement. The substitution of expensive iodinating agents with cost-effective zinc salts and nitrites results in a drastic reduction in raw material expenditure. This cost structure allows for more competitive pricing models without sacrificing product quality, addressing the constant pressure to achieve cost reduction in pharmaceutical intermediates manufacturing. Moreover, the reliance on commodity chemicals ensures that supply disruptions are minimized, as these reagents are sourced from a broad and stable global supplier base. The simplified workflow also reduces the demand for specialized equipment, lowering capital expenditure requirements for production facilities.
- Cost Reduction in Manufacturing: The elimination of high-cost reagents like NIS and the reduction in solvent usage due to simpler workup procedures contribute to substantial cost savings. The high atom economy of the reaction ensures that a greater proportion of input materials are converted into saleable product, reducing waste disposal fees. Additionally, the mild reaction conditions lower energy consumption associated with heating or cooling, further optimizing the operational expenditure profile. These factors combine to create a highly efficient production model that maximizes return on investment for large-scale manufacturing campaigns.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents such as zinc diiodide and tert-butyl nitrite mitigates the risk of supply chain bottlenecks often associated with specialty chemicals. The robustness of the reaction against variations in substrate electronics means that a single process platform can be used to manufacture a diverse portfolio of iodinated intermediates. This flexibility allows manufacturers to respond quickly to changing market demands and custom synthesis requests. Reducing lead time for high-purity alkynyl iodides becomes achievable when the process is not constrained by scarce reagents or complex purification trains, ensuring consistent delivery schedules for downstream clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from milligram to mole scales without loss of performance. The absence of heavy metal catalysts simplifies environmental compliance and waste treatment, as the effluent streams are less toxic and easier to process. This aligns with modern green chemistry principles, enhancing the corporate sustainability profile of the manufacturer. The ability to scale up complex organic synthons safely and responsibly is a key differentiator in the competitive fine chemicals market, appealing to partners who prioritize environmental stewardship alongside commercial performance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthetic technology. They are derived from a detailed analysis of the patent specifications and practical considerations for industrial application. Understanding these aspects helps stakeholders make informed decisions about integrating this method into their supply chains. The answers reflect the consensus on the method's capabilities regarding purity, scalability, and operational safety.
Q: What are the primary advantages of the ZnI2/TBN method over traditional NIS iodination?
A: The ZnI2/TBN system utilizes significantly cheaper and more readily available reagents compared to N-Iodosuccinimide (NIS). Furthermore, it operates under milder conditions (0-25°C) with superior atom economy, reducing hazardous waste generation.
Q: Does this synthetic route support a wide range of substrate functional groups?
A: Yes, the patent data confirms excellent tolerance for various substituents including alkyl, alkoxy, nitro, cyano, and halogen groups on the benzene ring, making it versatile for diverse API intermediate synthesis.
Q: Is this process suitable for large-scale commercial production?
A: Absolutely. The method avoids expensive catalysts and extreme conditions, utilizing common solvents and simple workup procedures (washing and chromatography), which facilitates straightforward scale-up from laboratory to industrial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylethynyl Iodide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand that consistency is key, and our state-of-the-art facilities are designed to handle sensitive chemistries like the ZnI2-mediated iodination with precision and care.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume and purity requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain capable of delivering high-purity phenylethynyl iodide compounds that meet the demanding specifications of the global pharmaceutical industry.
