Scalable Synthesis of 2-Fluoro-5-Alkoxyl Aniline: A Technical Breakthrough for Global Supply Chains
The global demand for fluorinated aromatic amines continues to surge, driven by their critical role as building blocks in modern medicinal chemistry and agrochemical formulations. Patent CN101445460A introduces a robust and industrially viable method for preparing 2-fluoro-5-alkoxyl aniline, a versatile intermediate characterized by its specific substitution pattern that is highly valued in drug discovery. This technical disclosure outlines a three-step synthetic pathway starting from commercially available 2-fluoronitrobenzene, proceeding through selective bromination and reduction, and culminating in a copper-catalyzed etherification. For R&D directors and procurement specialists, this route represents a significant departure from hazardous legacy processes, offering a safer, more cost-effective alternative that aligns with modern green chemistry principles while maintaining rigorous quality standards required for high-purity pharmaceutical intermediates.
The strategic value of this patent lies not only in the chemical transformation itself but in its adaptability for large-scale production. By utilizing common reagents such as iron powder for reduction and avoiding extreme high-pressure conditions, the process mitigates many of the supply chain bottlenecks associated with specialized fluorination reagents. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances allows us to optimize production schedules and ensure consistent batch-to-batch quality. The following analysis dissects the technical merits of this approach, highlighting how it addresses the twin challenges of operational safety and economic efficiency in the manufacture of complex fluorinated scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-fluoro-5-alkoxyl aniline derivatives has been plagued by severe safety hazards and operational complexities. Prior art, such as the method disclosed in U.S. Patent 4,006,185, relies heavily on the use of anhydrous hydrogen fluoride (HF) as both a solvent and a fluorinating agent under extremely high pressures, often reaching 42.5 atmospheres. This approach presents catastrophic risks; HF is a highly corrosive and toxic substance that requires specialized, expensive containment infrastructure and rigorous safety protocols to prevent lethal exposure. Furthermore, the single-step nature of such high-pressure reactions often results in poor selectivity and modest yields, reported around 29.4%, which drastically inflates the cost of goods sold due to extensive purification requirements and low throughput. The reliance on such dangerous reagents also creates significant supply chain vulnerabilities, as fewer manufacturers are willing or equipped to handle bulk quantities of anhydrous HF, leading to potential disruptions in the availability of commercial scale-up of complex polymer additives and pharma precursors.
The Novel Approach
In stark contrast, the methodology described in CN101445460A circumvents these dangers by employing a modular three-step sequence that operates under atmospheric or near-atmospheric pressure with mild thermal conditions. The process initiates with the regioselective bromination of 2-fluoronitrobenzene, followed by a gentle reduction of the nitro group, and concludes with an etherification step that installs the desired alkoxy moiety. This strategic fragmentation of the synthesis allows for precise control over impurities at each stage, ensuring that the final product meets stringent purity specifications without the need for aggressive purification techniques. By replacing the hazardous HF route with standard organic transformations using bromine, iron powder, and copper catalysts, the novel approach significantly lowers the barrier to entry for manufacturing. This shift not only enhances worker safety but also simplifies the engineering requirements for reactors, thereby facilitating cost reduction in pharmaceutical intermediate manufacturing through reduced capital expenditure and lower operational overheads.
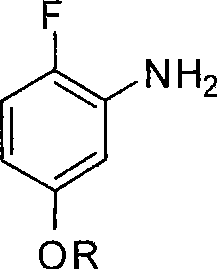
Mechanistic Insights into Copper-Catalyzed Etherification and Regioselective Bromination
The core of this synthetic innovation lies in the final etherification step, which effectively functions as a variation of the Ullmann condensation or a nucleophilic aromatic substitution facilitated by copper catalysis. In this critical transformation, 2-fluoro-5-bromaniline reacts with an alkali metal alkoxide (such as sodium methoxide or potassium ethoxide) in the presence of a copper(I) salt like CuI or CuBr. The mechanism likely involves the oxidative addition of the aryl bromide to the copper center, forming an aryl-copper intermediate, followed by ligand exchange with the alkoxide and subsequent reductive elimination to forge the C-O bond. The patent specifies reaction temperatures between 60°C and 120°C, with optimal results observed around 80°C to 100°C. This thermal window is crucial; temperatures below 60°C result in sluggish reaction kinetics, while excessive heat can promote secondary substitutions or decomposition, leading to high-boiling impurities that are difficult to remove. The choice of solvent, preferably polar aprotic solvents like DMF or alcohols like methanol, further stabilizes the transition state and solubilizes the ionic alkoxide species, driving the equilibrium toward the desired 2-fluoro-5-alkoxyl aniline product.
Prior to this coupling event, the precursor 2-fluoro-5-bromaniline is generated through a highly controlled reduction of the corresponding nitro compound. The patent highlights the use of iron powder in acidic media (Fe/HCl) or catalytic hydrogenation using Raney nickel as effective reduction strategies. The iron-mediated reduction is particularly noteworthy for its chemoselectivity; it efficiently reduces the nitro group to an amine without affecting the sensitive carbon-fluorine bond or the carbon-bromine bond, which are susceptible to hydrodehalogenation under harsher reducing conditions. This selectivity is paramount for maintaining the structural integrity of the fluorinated scaffold. Furthermore, the initial bromination step utilizes the directing effects of the nitro and fluoro groups to ensure substitution occurs exclusively at the 5-position. By conducting this reaction at controlled temperatures ranging from -10°C to 50°C, the process minimizes poly-bromination side products, ensuring that the subsequent reduction and etherification steps proceed with high fidelity and minimal formation of isomeric impurities that could compromise the biological activity of the final API.
How to Synthesize 2-Fluoro-5-Alkoxyl Aniline Efficiently
The synthesis of this valuable intermediate is streamlined into three distinct operational units that can be easily integrated into existing multipurpose chemical plants. The process begins with the preparation of 2-fluoro-5-bromo nitrobenzene, followed by its reduction to the aniline derivative, and finally the copper-catalyzed etherification to install the alkoxy group. Each step has been optimized for yield and purity, with the patent reporting overall yields exceeding 60% and final product purity greater than 99%. The detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined below to guide process engineers in replicating this efficient route.
- Perform regioselective bromination of 2-fluoronitrobenzene using brominating agents like Br2 or NBS at temperatures between -10°C and 50°C to yield 2-fluoro-5-bromo nitrobenzene.
- Reduce the nitro group of the intermediate using iron powder and acid or catalytic hydrogenation at 30°C to 90°C to obtain 2-fluoro-5-bromaniline.
- Execute copper-catalyzed etherification by reacting 2-fluoro-5-bromaniline with alkali metal alkoxides at 60°C to 120°C to finalize the 2-fluoro-5-alkoxyl aniline structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage is the drastic simplification of the raw material portfolio. By eliminating the need for anhydrous hydrogen fluoride and high-pressure autoclaves, the process relies on commodity chemicals such as bromine, iron powder, and simple alcohols, which are readily available from multiple global suppliers. This diversification of the supply base reduces the risk of single-source dependency and price volatility, ensuring a more stable and predictable flow of materials. Additionally, the mild reaction conditions translate directly into lower energy consumption and reduced maintenance costs for reactor vessels, as there is no need for exotic alloys resistant to HF corrosion. These factors collectively contribute to a leaner manufacturing model that is both economically resilient and environmentally sustainable.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like anhydrous HF creates immediate savings in raw material procurement and waste disposal costs. Traditional HF processes require specialized neutralization and treatment facilities to handle toxic effluents, whereas the waste streams from this iron-reduction and copper-catalyzed route are significantly easier and cheaper to manage. Furthermore, the use of iron powder as a reducing agent is vastly more economical than precious metal catalysts or hydride reagents, driving down the variable cost per kilogram of the final product. The high selectivity of the reaction also minimizes the loss of valuable fluorinated intermediates to side reactions, maximizing the atom economy and ensuring that every dollar spent on raw materials contributes effectively to the final yield.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals ensures that production schedules are not held hostage by the scarcity of niche reagents. Iron powder, bromine, and copper salts are produced at massive scales for various industries, guaranteeing a consistent supply even during market fluctuations. This robustness allows for better long-term planning and inventory management, reducing the need for safety stock and freeing up working capital. Moreover, the simplified equipment requirements mean that the process can be transferred to a wider range of contract manufacturing organizations (CMOs) without extensive retrofitting, providing greater flexibility in sourcing and the ability to quickly scale production volumes to meet sudden spikes in market demand.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is far superior to legacy methods. The absence of high-pressure operations and carcinogenic gases significantly lowers the regulatory burden and insurance premiums associated with the manufacturing site. The process generates less hazardous waste, aligning with increasingly strict global environmental regulations and corporate sustainability goals. The ability to run the reaction at atmospheric pressure and moderate temperatures also simplifies the engineering scale-up, allowing for larger batch sizes without the exponential increase in risk associated with high-pressure systems. This scalability ensures that the supply chain can grow seamlessly alongside the commercial success of the downstream pharmaceutical products that utilize this intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-fluoro-5-alkoxyl aniline. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners and stakeholders.
Q: What are the safety advantages of this synthesis route compared to traditional methods?
A: Unlike traditional methods requiring anhydrous hydrogen fluoride (HF) under high pressure (42.5 atm), this patented route utilizes mild conditions and avoids highly toxic and carcinogenic HF, significantly reducing industrial safety hazards and equipment corrosion risks.
Q: What is the expected purity and yield of the final product?
A: According to the patent data, the final 2-fluoro-5-alkoxyl aniline product can achieve a purity of over 99% after purification, with a total reaction yield exceeding 60%, ensuring high quality for downstream pharmaceutical applications.
Q: Which catalysts are preferred for the etherification step?
A: The process preferably employs copper-based catalysts such as CuI, CuCl, CuBr, or CuCN, with CuI being particularly effective in facilitating the substitution reaction between the bromaniline intermediate and the alkoxide source.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-5-Alkoxyl Aniline Supplier
The technical pathway outlined in CN101445460A demonstrates immense potential for the efficient production of high-value fluorinated intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and purity profile of every batch, guaranteeing that the 2-fluoro-5-alkoxyl aniline supplied meets the exacting standards required for global pharmaceutical registration.
We invite you to collaborate with us to optimize your supply chain for this critical building block. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that balance cost, quality, and delivery timelines effectively.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
