Advanced Halogenation Technology for Scalable Pharmaceutical Intermediate Production
Advanced Halogenation Technology for Scalable Pharmaceutical Intermediate Production
The landscape of fine chemical synthesis is constantly evolving, driven by the relentless demand for higher purity intermediates and more sustainable manufacturing processes. A significant breakthrough in this domain is documented in patent CN110835308A, which details a robust and versatile preparation method for halogenated o-aminobenzoic acid compounds. These compounds serve as critical building blocks in the construction of complex pharmaceutical active ingredients and agrochemical agents, where the precise placement of halogen atoms can drastically alter biological activity and metabolic stability. The disclosed technology addresses long-standing inefficiencies in traditional halogenation routes by introducing a streamlined protocol that utilizes readily available N-halosuccinimides and common organic solvents. For global procurement leaders and R&D directors, this patent represents a pivotal shift towards more reliable supply chains and cost-effective production strategies for high-value heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of poly-halogenated anthranilic acids has been plagued by significant operational challenges that hinder efficient commercial scale-up. Prior art, such as the processes described in WO 2007/090752A1, often suffers from prolonged reaction times and suboptimal yields, which directly inflate the cost of goods sold (COGS) for downstream manufacturers. Traditional methods frequently rely on the use of elemental halogens like chlorine or bromine gas, which pose severe safety hazards regarding storage, handling, and waste gas treatment, necessitating expensive specialized infrastructure. Furthermore, these aggressive reagents often lack the necessary selectivity, leading to complex mixtures of regioisomers that require tedious and yield-eroding purification steps. The inability to consistently control the degree of halogenation results in batch-to-batch variability, a critical risk factor for pharmaceutical supply chains that demand rigorous quality consistency. Consequently, the industry has been in urgent need of a safer, more predictable, and higher-yielding alternative to overcome these bottlenecks.
The Novel Approach
The methodology outlined in the present invention offers a transformative solution by employing solid or liquid N-halosuccinimides as the primary halogen source, fundamentally changing the safety and efficiency profile of the reaction. By reacting general formula (II) anthranilic acids with reagents such as N-chlorosuccinimide (NCS) or N-bromosuccinimide (NBS) in polar solvents, the process achieves exceptional conversion rates within a significantly reduced timeframe of 0.1 to 24 hours. This approach eliminates the need for hazardous gas handling equipment, thereby simplifying the reactor setup and reducing capital expenditure for manufacturing facilities. The versatility of the method is demonstrated by its tolerance to a wide array of substituents (R1), including fluoro, chloro, methoxy, and cyano groups, allowing for the synthesis of a diverse library of intermediates from a single platform technology. As illustrated in the specific embodiment below, the reaction proceeds smoothly to introduce multiple halogen atoms with high precision, ensuring that the final product meets the stringent specifications required for advanced drug synthesis.
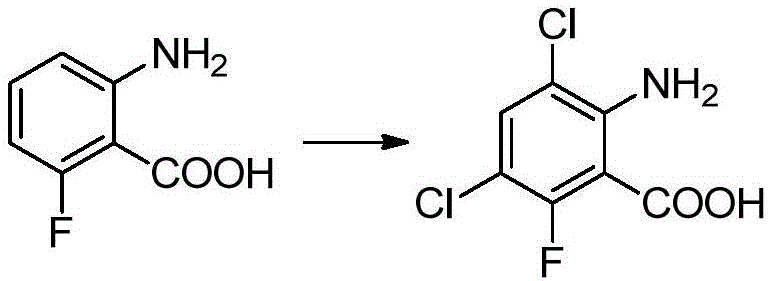
This specific transformation highlights the practical application of the technology, where 2-amino-6-fluorobenzoic acid is efficiently converted into its dichlorinated derivative with an impressive isolated yield of 95%. Such high efficiency not only maximizes raw material utilization but also minimizes waste generation, aligning with modern green chemistry principles. The use of N,N-dimethylformamide (DMF) as a solvent facilitates the dissolution of both the starting material and the halogenating agent, creating a homogeneous reaction environment that promotes uniform heat transfer and reaction kinetics. This level of process control is essential for maintaining product quality during large-scale production runs, ensuring that the supply of these critical intermediates remains uninterrupted and cost-competitive.
Mechanistic Insights into Electrophilic Aromatic Substitution
The core of this synthetic innovation lies in the controlled electrophilic aromatic substitution mechanism facilitated by the succinimide leaving group. When N-chlorosuccinimide or N-bromosuccinimide is introduced into the reaction mixture, the polar solvent environment assists in the polarization of the N-X bond, generating a potent electrophilic halogen species in situ. The electron-rich aromatic ring of the anthranilic acid derivative, activated by the amino group, undergoes attack by this electrophile at the positions ortho and para to the activating group, dictated by the existing substitution pattern. The presence of the carboxylic acid group exerts a meta-directing influence, but the strong activation from the amino group typically dominates, guiding the halogenation to the desired positions on the ring system. This delicate balance of electronic effects allows for the selective introduction of halogen atoms without the need for protecting groups, which simplifies the synthetic route and reduces the number of unit operations.
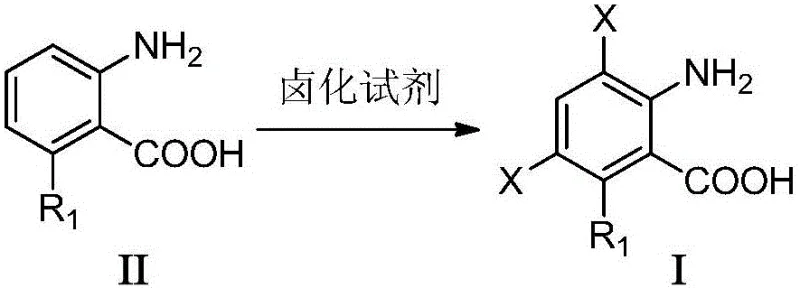
Furthermore, the reaction conditions described in the patent, ranging from -20°C to reflux temperature, provide a tunable parameter for optimizing selectivity versus reaction rate. At lower temperatures, kinetic control may favor mono-halogenation, while elevated temperatures drive the reaction towards thermodynamic products, often resulting in di- or tri-halogenated species as seen in the general formula (I). The choice of solvent plays a crucial role in stabilizing the transition states and intermediates; polar aprotic solvents like DMF and NMP are particularly effective because they solvate the cationic intermediates without interfering with the nucleophilicity of the aromatic ring. Understanding these mechanistic nuances allows process chemists to fine-tune the reaction parameters to minimize the formation of unwanted byproducts, such as over-halogenated species or oxidation products, thereby ensuring a clean impurity profile that simplifies downstream purification.
How to Synthesize Halogenated Anthranilic Acid Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent addition rates and temperature control to manage the exothermic nature of the halogenation. The process begins with the dissolution of the specific anthranilic acid substrate in a selected solvent, followed by the portionwise addition of the N-halosuccinimide reagent to prevent thermal runaway. Detailed standard operating procedures regarding stoichiometry, agitation speeds, and quenching protocols are essential for reproducibility and safety. For a comprehensive guide on the exact step-by-step execution of this synthesis, please refer to the standardized protocol below.
- Dissolve the substituted anthranilic acid starting material in a suitable solvent such as DMF or acetic acid.
- Add the halogenating reagent, preferably N-chlorosuccinimide or N-bromosuccinimide, portionwise while controlling the temperature.
- Maintain the reaction mixture at elevated temperatures (e.g., 60-100°C) until completion, then precipitate the product in water.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this halogenation technology offers substantial benefits that extend beyond mere chemical yield improvements. For procurement managers tasked with reducing the overall cost of goods, the elimination of hazardous elemental gases translates directly into lower operational expenditures related to safety compliance and waste disposal. The use of stable, solid halogenating agents like NCS and NBS simplifies logistics and storage requirements, reducing the risk of supply disruptions caused by the transportation restrictions often placed on compressed gases. Moreover, the high atom economy and simplified workup procedure—often involving simple precipitation in water—drastically reduce solvent consumption and energy usage during the isolation phase. These factors combine to create a more resilient and cost-efficient supply chain for high-purity pharmaceutical intermediates, enabling manufacturers to offer more competitive pricing to their downstream clients.
- Cost Reduction in Manufacturing: The shift from gaseous halogens to solid N-halosuccinimides removes the need for expensive corrosion-resistant reactors and complex scrubbing systems, leading to significant capital and maintenance savings. Additionally, the high yields reported (often exceeding 90%) mean that less raw material is wasted, directly lowering the variable cost per kilogram of the final product. The simplified purification process further reduces the consumption of chromatography media or recrystallization solvents, contributing to a leaner and more economical manufacturing footprint.
- Enhanced Supply Chain Reliability: By utilizing commercially abundant reagents such as N-chlorosuccinimide and common solvents like DMF or acetic acid, the dependency on specialized chemical suppliers is minimized. This broadens the supplier base for key inputs, mitigating the risk of shortages that can plague niche reagent markets. The robustness of the reaction conditions, which tolerate a wide temperature range, ensures that production can continue reliably even if minor fluctuations in utility supplies occur, guaranteeing consistent delivery schedules for critical API intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-kilogram scales without loss of efficiency. The reduction in hazardous waste generation, particularly the avoidance of acidic halogen waste streams, aligns with increasingly strict environmental regulations globally. This eco-friendly profile not only reduces disposal costs but also enhances the corporate sustainability credentials of the manufacturer, a key factor for multinational corporations aiming to meet their carbon neutrality and green chemistry goals.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the process parameters and scope. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of what can be achieved with this methodology. Understanding these details is crucial for integrating this synthesis route into existing production pipelines or for designing new campaigns for halogenated building blocks.
Q: What are the advantages of using N-halosuccinimides over elemental halogens?
A: N-halosuccinimides offer safer handling, better regioselectivity, and eliminate the need for complex gas scrubbing systems required for chlorine or bromine gas.
Q: Can this method be scaled for industrial production?
A: Yes, the process utilizes common solvents and operates within a wide temperature range (-20°C to reflux), making it highly adaptable for large-scale commercial manufacturing.
Q: What is the typical purity profile of the resulting intermediates?
A: The method yields high-purity products often exceeding 90% isolated yield with minimal byproduct formation due to the mild nature of the halogenating agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Anthranilic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require custom synthesis of novel derivatives or reliable supply of catalog compounds, our infrastructure is designed to meet the demanding timelines and quality standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced halogenation technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain security and reduce your overall manufacturing costs.
