Advanced One-Pot Synthesis of Ralotinib Intermediate for Commercial Scale-Up
Advanced One-Pot Synthesis of Ralotinib Intermediate for Commercial Scale-Up
The pharmaceutical landscape for oncology treatments is continuously evolving, driven by the need for more efficient and safer manufacturing processes for critical drug intermediates. Patent CN111393347B, published in late 2020, introduces a transformative synthetic methodology for producing 5-(2,5-difluorophenyl)-3,4-dihydro-2H-pyrrole, a pivotal intermediate in the synthesis of the antitumor drug Ralotinib (also known as Larotrectinib). This innovation addresses long-standing challenges in the production of TRK inhibitors by replacing hazardous reagents and energy-intensive conditions with a streamlined, one-pot ammonolysis and cyclization strategy. For global procurement leaders and R&D directors, this patent represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates while mitigating operational risks associated with traditional synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of this key pyrrole intermediate relied on pathways that were either economically prohibitive or operationally hazardous for large-scale manufacturing. One prominent prior art route, disclosed in WO2009140128A2, utilized a Grignard reaction involving N-Boc-2-pyrrolidone. As illustrated in the reaction scheme below, this method necessitates cryogenic conditions at -78°C, which imposes a massive energy burden and requires specialized, expensive reactor infrastructure that limits throughput.
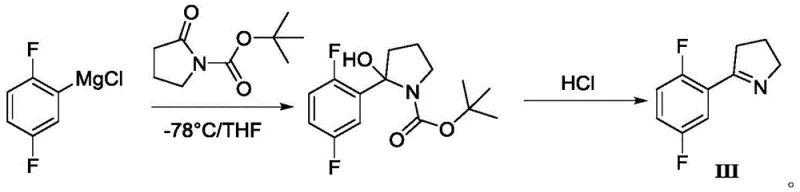
Furthermore, the reliance on N-Boc-2-pyrrolidone introduces high raw material costs, making the overall cost of goods sold (COGS) unfavorable for generic or volume production. Another alternative route reported in Chinese patent CN108794370A attempted to bypass these issues but introduced severe safety liabilities by employing sodium azide. The use of azides carries inherent explosion risks, and the subsequent Staudinger reduction generates triphenylphosphine oxide, a byproduct that is notoriously difficult to separate from the product, often requiring complex chromatographic purification that further erodes yield and increases waste generation.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN111393347B utilizes 4-chloro-1-(2,5-difluorophenyl)butan-1-one as a readily available starting material. This strategy leverages a direct nucleophilic substitution with liquid ammonia followed by an intramolecular cyclization, all achievable within a single reaction vessel. By eliminating the need for cryogenic cooling and avoiding toxic azide reagents, this method fundamentally reshapes the economic and safety profile of the synthesis. The transition to a one-pot process not only simplifies the operational workflow but also minimizes solvent usage and intermediate isolation steps, which are critical factors for achieving cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into One-Pot Ammonolysis and Cyclization
The core of this technological breakthrough lies in the precise control of reaction conditions to facilitate a tandem transformation. The mechanism initiates with the nucleophilic attack of liquid ammonia on the chloro-ketone substrate under moderate pressure (0.3-0.5 MPa) and elevated temperature (50-60°C). This step efficiently converts the chloro group into an amino functionality, generating an unstable amino-ketone intermediate (Compound II) in situ. Unlike traditional methods that might require isolating this sensitive intermediate, the new process maintains the reaction mixture, allowing for immediate progression to the next stage without exposure to air or moisture that could degrade the product.
Following the ammonolysis, the reaction environment is carefully adjusted by introducing an acid, such as formic acid, to lower the pH to a specific range of 4.0-4.5. This acidification triggers the intramolecular condensation of the amino group with the ketone carbonyl, driving the formation of the dihydropyrrole ring through dehydration. The visual representation of this streamlined pathway highlights the elegance of the design, where harsh reagents are replaced by benign ammonia and simple acids.
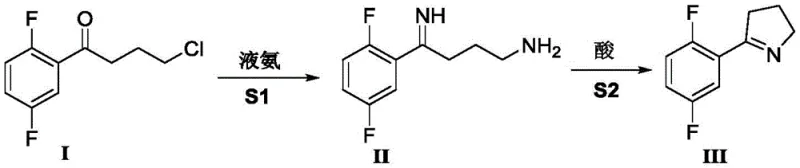
From an impurity control perspective, this mechanism is superior because it avoids the formation of phosphine oxides or complex organometallic byproducts. The primary impurities are likely inorganic salts and unreacted starting materials, which are easily removed via standard aqueous workup and extraction protocols. This clean reaction profile ensures that the final product achieves a gas phase purity of up to 98.5%, meeting the stringent quality standards required for oncology drug substances without the need for resource-intensive recrystallization or column chromatography.
How to Synthesize 5-(2,5-difluorophenyl)-3,4-dihydro-2H-pyrrole Efficiently
Implementing this synthesis requires careful attention to pressure management and pH control to maximize the 93% total yield reported in the patent examples. The process is designed to be robust, tolerating common solvents like tetrahydrofuran or ethanol, which provides flexibility for plant operators to utilize existing solvent recovery systems. The following guide outlines the critical operational parameters derived directly from the patent data to ensure reproducible high-quality output.
- Charge 4-chloro-1-(2,5-difluorophenyl)butan-1-one into an autoclave with solvent (e.g., THF), introduce liquid ammonia, and react at 55°C under 0.4 MPa pressure for 4 hours.
- Cool the reaction mixture, add water, and slowly adjust the pH to 4.0 using formic acid to initiate cyclization.
- Stir at room temperature for 4-5 hours, then concentrate, extract with ethyl acetate, wash, dry, and concentrate to obtain the final product with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic advantages that extend beyond simple chemistry. The elimination of cryogenic requirements and hazardous azides translates directly into reduced operational expenditures and lower insurance premiums, creating a more resilient supply chain for this critical oncology intermediate. By simplifying the process flow, manufacturers can significantly increase batch turnover rates, thereby enhancing the reliability of supply for downstream API producers who depend on consistent volumes of high-purity intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily driven by the removal of expensive reagents like N-Boc-2-pyrrolidone and the elimination of energy-intensive cooling systems. Operating at moderate temperatures of 55°C rather than -78°C drastically reduces utility costs, while the high yield of 93% minimizes raw material waste. Furthermore, the avoidance of difficult-to-remove byproducts like triphenylphosphine oxide reduces the consumption of silica gel and solvents associated with purification, leading to significant overall cost savings in the production budget.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents or the logistical challenges of handling dangerous goods. By utilizing liquid ammonia and commodity acids like formic acid, this method relies on globally available feedstocks that are less susceptible to supply shocks. The simplified one-pot nature of the reaction also reduces the number of unit operations, decreasing the likelihood of mechanical failures or bottlenecks that typically delay production schedules in multi-step syntheses.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden safety and environmental flaws, but this route is inherently designed for industrial expansion. The use of standard autoclaves operating at safe pressures allows for straightforward scale-up from pilot plants to multi-ton commercial production without requiring exotic equipment. Additionally, the absence of heavy metals and explosive azides simplifies wastewater treatment and waste disposal, ensuring easier compliance with increasingly strict environmental regulations in major manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or contract manufacturing opportunities.
Q: How does this new synthesis route improve safety compared to azide-based methods?
A: Unlike prior art methods utilizing virulent and explosive sodium azide, this patented process employs liquid ammonia under controlled pressure, completely eliminating the risk of azide accumulation and the difficult removal of triphenylphosphine oxide byproducts.
Q: What are the yield and purity specifications achievable with this method?
A: The optimized one-pot protocol demonstrates a robust total yield of approximately 93% across two steps, with gas phase purity consistently reaching 98% to 98.5%, ensuring high-quality material for downstream API synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates at moderate temperatures (50-60°C) and pressures (0.3-0.5 MPa) using standard autoclaves, avoiding the extreme cryogenic conditions (-78°C) required by previous Grignard-based routes, thus facilitating easy commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ralotinib Intermediate Supplier
As the demand for targeted cancer therapies continues to grow, securing a stable source of high-quality intermediates is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep technical expertise to master complex synthetic pathways like the one described in CN111393347B. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global clinical and commercial programs. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Ralotinib intermediate meets the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this safer, more efficient method. Contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, and let us help you accelerate the delivery of life-saving medicines to patients worldwide.
