Advanced Asymmetric Synthesis of Chiral Tricyclic Benzofuran Intermediates for Pharmaceutical Applications
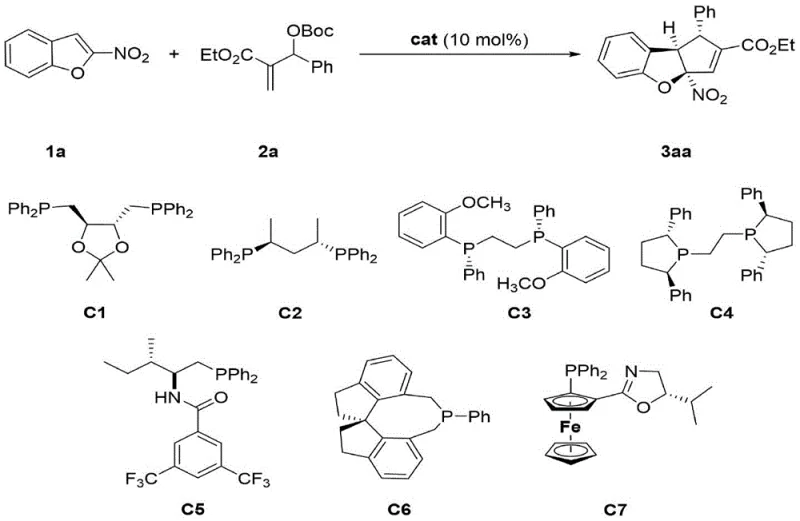
The development of efficient synthetic routes for complex heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry, particularly for accessing bioactive cores found in natural products and pharmaceutical agents. Patent CN110156730B discloses a groundbreaking methodology for the synthesis of chiral tricyclic benzofuran compounds through an asymmetric dearomatization [3+2] cycloaddition reaction. This technology addresses the critical industry need for constructing polycyclic heterocyclic units with benzofuran skeletons, which are prevalent in molecules exhibiting anti-cancer, anti-coagulation, and anti-inflammatory properties. Unlike traditional approaches that often struggle with stereocontrol, this invention leverages an organic phosphine catalyst to drive the reaction between nitro-substituted benzofurans and MBH carbonates. The result is a highly selective transformation that generates products possessing three contiguous chiral centers in a single operational step. This represents a significant leap forward in the field of asymmetric synthesis, offering a robust platform for generating high-value pharmaceutical intermediates.
![General reaction scheme for asymmetric dearomatization [3+2] cycloaddition of nitro-substituted benzofuran and MBH carbonate](/insights/img/chiral-benzofuran-synthesis-pharma-supplier-20260304134251-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral tricyclic benzofuran frameworks has been fraught with synthetic challenges that hinder both research efficiency and commercial viability. Conventional methodologies often rely on transition metal catalysis, which introduces significant downstream processing burdens due to the necessity of removing toxic heavy metal residues to meet stringent regulatory standards for active pharmaceutical ingredients (APIs). Furthermore, many existing protocols suffer from poor stereochemical control, yielding racemic mixtures or products with low diastereomeric ratios that require costly and material-intensive chiral separation techniques. The harsh reaction conditions frequently employed, such as extreme temperatures or strong bases, can also lead to substrate decomposition and limited functional group tolerance. These factors collectively contribute to extended development timelines and inflated manufacturing costs, creating a bottleneck for the rapid deployment of new benzofuran-based therapeutics into the clinical pipeline.
The Novel Approach
The methodology outlined in the patent data presents a transformative solution by utilizing an organocatalytic system that circumvents the drawbacks of metal-mediated processes. By employing specifically designed chiral organic phosphine catalysts, such as catalyst C6, the reaction achieves exceptional levels of regioselectivity, diastereoselectivity, and enantioselectivity under remarkably mild conditions. The optimization data reveals that conducting the reaction in chloroform at 0°C yields superior results compared to other solvents and temperatures, demonstrating the precision of this catalytic system. For instance, the model reaction between 2-nitrobenzofuran and MBH carbonate proceeds with a yield of up to 93% and an enantiomeric excess (ee) of 97%, with a diastereomeric ratio (dr) exceeding 20:1. This high degree of control eliminates the need for difficult separations and ensures that the desired chiral architecture is established directly during the bond-forming event.
Mechanistic Insights into Organophosphine-Catalyzed Asymmetric Dearomatization
The core of this technological advancement lies in the mechanism of the asymmetric dearomatization [3+2] cycloaddition. The reaction initiates with the nucleophilic attack of the chiral phosphine catalyst on the MBH carbonate, generating a reactive zwitterionic intermediate. This species then engages with the electron-deficient nitro-substituted benzofuran, which acts as a 1,3-dipole equivalent. The chiral environment provided by the bulky substituents on the phosphine catalyst dictates the facial selectivity of the addition, ensuring that the new carbon-carbon bonds are formed with precise spatial orientation. This stereocontrolled cyclization disrupts the aromaticity of the benzofuran ring temporarily, leading to the formation of the complex tricyclic framework. The presence of the nitro group is crucial not only for activating the benzofuran towards nucleophilic attack but also for stabilizing the transition state, thereby facilitating the formation of the three contiguous stereocenters observed in the final product structure.
From an impurity control perspective, the high selectivity of this catalytic cycle is paramount for maintaining product purity. The strict dr value of >20:1 indicates that the formation of unwanted diastereomers is effectively suppressed, minimizing the generation of structurally similar impurities that are notoriously difficult to separate. Additionally, the use of an inert atmosphere (nitrogen protection) prevents oxidative degradation of the sensitive phosphine catalyst and the reactive intermediates. The subsequent workup involves standard purification techniques such as preparative thin-layer chromatography or column chromatography using ethyl acetate and petroleum ether, which are scalable and cost-effective. The ability to derivatize the nitro group post-reaction, either through denitration to form compound 4 or dihydroxylation to form compound 5, further underscores the versatility of this scaffold, allowing medicinal chemists to access a diverse library of analogues from a common intermediate.
How to Synthesize Chiral Tricyclic Benzofuran Efficiently
To implement this synthesis in a laboratory or pilot plant setting, strict adherence to the optimized protocol is essential to replicate the high yields and selectivity reported in the patent. The process begins with the careful preparation of dry glassware and the establishment of an inert nitrogen atmosphere to protect the catalyst. The stoichiometry of the reactants is critical, with a molar ratio of nitro-substituted benzofuran to MBH carbonate typically ranging from 1:1 to 1:2, and a catalyst loading of 5 to 10 mol%. The detailed standardized synthesis steps below outline the precise procedure for achieving optimal results, including specific quenching and purification methods that ensure the isolation of high-purity material suitable for downstream applications.
- Prepare the reaction vessel by adding nitro-substituted benzofuran and MBH carbonate under inert atmosphere.
- Introduce the optimized organic phosphine catalyst (C6) and anhydrous chloroform solvent at 0°C.
- Stir the mixture for 12 hours, monitor by TLC, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organophosphine-catalyzed route offers distinct strategic advantages over traditional metal-dependent syntheses. The elimination of expensive and toxic transition metals significantly simplifies the purification workflow, reducing the reliance on specialized scavenging resins or complex extraction protocols required to meet heavy metal specifications. This streamlining of the downstream process translates directly into reduced operational expenditures and shorter batch cycle times. Furthermore, the starting materials, including various substituted nitrobenzofurans and MBH carbonates, are readily accessible from commercial suppliers or can be synthesized via established pathways, ensuring a stable and resilient supply chain. The robustness of the reaction conditions, which do not require cryogenic cooling or high-pressure equipment, lowers the barrier for technology transfer from laboratory to commercial scale.
- Cost Reduction in Manufacturing: The use of organocatalysts instead of precious metal complexes drastically lowers the raw material costs associated with the catalytic system. Since the catalyst is metal-free, there is no need for costly analytical testing for residual metals like palladium or platinum, nor is there a need for expensive disposal procedures for metal-contaminated waste streams. The high atom economy and excellent yields minimize raw material waste, contributing to a more sustainable and economically efficient manufacturing process that aligns with green chemistry principles.
- Enhanced Supply Chain Reliability: The reaction operates effectively at 0°C, a temperature that is easily maintained using standard industrial cooling systems without the energy intensity of deep freeze operations. This thermal accessibility reduces the risk of process deviations due to temperature fluctuations. Additionally, the broad substrate scope demonstrated in the patent, accommodating various electron-withdrawing and electron-donating groups on the aromatic rings, allows for the flexible production of a wide range of derivatives using a unified platform, thereby simplifying inventory management and production scheduling.
- Scalability and Environmental Compliance: The solvent system primarily utilizes chloroform or dichloromethane, which are well-understood solvents in the pharmaceutical industry with established recovery and recycling protocols. The absence of heavy metals simplifies environmental compliance and waste treatment, reducing the regulatory burden on the manufacturing facility. The high diastereoselectivity ensures that the process generates minimal isomeric byproducts, maximizing the throughput of the desired active ingredient and reducing the volume of chemical waste generated per kilogram of product.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the method. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific drug discovery programs.
Q: What is the primary advantage of this organophosphine catalytic method?
A: The method utilizes metal-free organic catalysis to construct three contiguous chiral centers with high enantioselectivity (up to 99% ee) and diastereoselectivity (dr > 20:1) under mild conditions.
Q: Can the resulting tricyclic benzofuran products be further derivatized?
A: Yes, the nitro group can be removed to form denitrated products, or the double bond can undergo dihydroxylation, providing diverse scaffolds for drug discovery.
Q: Is this process suitable for large-scale manufacturing?
A: The reaction operates at accessible temperatures (0°C) using common solvents like chloroform, avoiding cryogenic conditions and expensive transition metals, which facilitates scalable production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Tricyclic Benzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and potency. Our capability to handle complex asymmetric syntheses, such as the organophosphine-catalyzed dearomatization described here, positions us as a strategic partner for companies seeking to secure their supply of challenging chiral building blocks.
We invite you to contact our technical procurement team to discuss your specific requirements for chiral tricyclic benzofuran derivatives. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new targets, our experts are ready to provide tailored solutions. Let us help you accelerate your drug development timeline with reliable, high-purity intermediates manufactured to the highest international standards.
