Scalable Synthesis of Substituted Pyrazole Amides for Next-Generation Insecticide Manufacturing
Scalable Synthesis of Substituted Pyrazole Amides for Next-Generation Insecticide Manufacturing
The global demand for high-efficacy, low-toxicity insecticides has driven significant innovation in the field of diamide chemistry, particularly for compounds targeting ryanodine receptors. A pivotal advancement in this sector is detailed in patent CN110835330A, which discloses a robust preparation method for substituted pyrazole amide compounds exhibiting potent insecticidal activity. This technology represents a strategic leap forward for manufacturers aiming to optimize the production of benzamide-class agrochemicals, offering a streamlined alternative to legacy processes that often struggle with yield consistency and impurity profiles. By leveraging a distinct three-step sequence involving acyl halogenation, controlled cyclization, and selective ring-opening, this methodology addresses critical bottlenecks in the supply chain for key agricultural intermediates.
For R&D directors and process chemists, the significance of this patent lies in its ability to decouple the formation of the amide bond from the cyclization event, thereby allowing for tighter control over reaction kinetics and thermal management. The process begins with the activation of a pyrazole carboxylic acid derivative, transforming it into a highly reactive acyl halide species. This intermediate is then subjected to a condensation reaction with a substituted anthranilic acid in the presence of a tertiary amine base, followed by an intramolecular cyclization mediated by a sulfonyl chloride to form a benzoxazinone core. Finally, the ring is opened by a specific amine nucleophile to deliver the target molecule. This modular approach not only enhances the structural diversity accessible to chemists but also ensures that the final product meets the stringent purity specifications required for modern crop protection formulations.
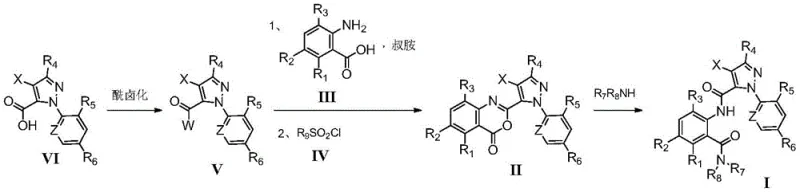
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing benzamide insecticides, such as those disclosed in earlier literature like WO 03/015519A1, typically rely on a direct coupling strategy where the carboxylic acid and anthranilic acid react in the presence of activating agents like methanesulfonyl chloride and pyridine. While conceptually straightforward, these one-pot or telescoped procedures often suffer from significant drawbacks when translated to large-scale manufacturing. The primary limitation is the formation of stubborn dehydration by-products and oligomeric impurities that are difficult to remove without expensive chromatographic purification, leading to overall isolated yields that frequently hover between 58% and 65%. Furthermore, the exothermic nature of direct activation can lead to thermal runaways if not meticulously managed, posing safety risks and compromising batch-to-batch reproducibility. The reliance on stoichiometric amounts of coupling reagents in a single vessel also complicates the workup procedure, generating substantial volumes of aqueous waste that increase the environmental footprint of the production facility.
The Novel Approach
In stark contrast, the methodology outlined in CN110835330A introduces a segmented strategy that isolates the reactive acyl halide intermediate prior to the coupling event. By pre-forming the acid chloride (or bromide) under controlled conditions, the subsequent reaction with the anthranilic acid derivative proceeds with much higher fidelity and selectivity. This separation of steps allows for the optimization of solvent systems and temperatures for each specific transformation, minimizing side reactions such as N-acylation of the wrong nitrogen or polymerization. The introduction of the benzoxazinone intermediate serves as a thermodynamic sink that drives the reaction forward, effectively locking in the desired connectivity before the final ring-opening step. This approach not only simplifies the purification protocol—often allowing for simple crystallization or filtration—but also dramatically improves the atom economy of the process by reducing the load of auxiliary reagents needed to force the reaction to completion.
Mechanistic Insights into Acyl Halogenation and Benzoxazinone Cyclization
The success of this synthetic route hinges on the precise execution of the acyl halogenation step, where the carboxylic acid moiety is activated for nucleophilic attack. As illustrated in the specific example of converting 3-bromo-1-(3-chloropyridin-2-yl)-1H-pyrazole-5-carboxylic acid, the choice of halogenating agent—whether it be thionyl chloride, oxalyl chloride, or solid phosgene—dictates the reaction profile and byproduct spectrum. The mechanism involves the initial formation of a mixed anhydride or acyl chlorosulfite intermediate, which subsequently collapses to release sulfur dioxide or carbon monoxide gases, driving the equilibrium toward the formation of the acyl chloride. This gaseous byproduct evolution is a critical process parameter; efficient venting is required to prevent pressure buildup in closed reactors, and the use of catalytic amounts of DMF can further accelerate the formation of the Vilsmeier-Haack type active species, ensuring complete conversion even at lower temperatures.
Following the generation of the acyl halide, the subsequent amidation and cyclization represent the most chemically intricate phase of the synthesis. The anthranilic acid acts as a bifunctional nucleophile, where the amino group attacks the acyl halide to form an amide bond, while the carboxylic acid group remains poised for the next transformation. The addition of a sulfonyl chloride, such as methanesulfonyl chloride, activates the carboxylic acid of the anthranilic moiety, facilitating an intramolecular nucleophilic attack by the adjacent amide oxygen or nitrogen to close the six-membered benzoxazinone ring. This cyclization is highly sensitive to the basicity of the medium; hence, the use of tertiary amines like triethylamine or pyridine is essential to scavenge the generated hydrogen chloride and maintain the nucleophilicity of the reacting species. The resulting benzoxazinone is a stable, isolable solid that serves as a protected form of the final amide, preventing premature hydrolysis and allowing for storage or transport if necessary before the final step.
How to Synthesize Substituted Pyrazole Amide Efficiently
The operational protocol for this synthesis is designed to be robust and scalable, accommodating a wide range of substrates without requiring specialized equipment beyond standard glass-lined or stainless steel reactors. The process begins with the suspension of the starting carboxylic acid in a non-polar or moderately polar solvent such as toluene or dichloromethane, followed by the slow addition of the halogenating agent to manage gas evolution. Once the acyl halide is confirmed via TLC or HPLC, the reaction mixture is cooled, and the anthranilic acid component is introduced along with the base. The subsequent addition of the sulfonyl chloride must be controlled to manage the exotherm, ensuring the temperature remains within the optimal window of -10°C to 30°C to prevent decomposition. The final ring-opening step is remarkably mild, often proceeding at room temperature or with slight heating in polar aprotic solvents like DMF or acetonitrile, yielding the final product after a simple aqueous quench and filtration.
- Perform acyl halogenation of the pyrazole carboxylic acid using reagents like thionyl chloride or solid phosgene at temperatures between -20°C and reflux to generate the reactive acyl halide intermediate.
- Execute amidation and cyclization by reacting the acyl halide with substituted anthranilic acid and a tertiary amine, followed by the addition of sulfonyl chloride to form the benzoxazinone core.
- Complete the synthesis via a ring-opening reaction where the benzoxazinone intermediate reacts with a primary or secondary amine to yield the final substituted pyrazole amide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this three-step synthetic route offers compelling advantages that directly impact the bottom line and operational resilience. The primary benefit stems from the significant improvement in process reliability and yield consistency compared to older direct-coupling methods. By breaking the synthesis into discrete, controllable stages, manufacturers can reduce the incidence of off-spec batches, thereby minimizing waste and maximizing the throughput of existing production assets. This reliability translates into a more predictable supply of critical agrochemical intermediates, reducing the risk of stockouts that can disrupt the formulation schedules of downstream pesticide producers. Furthermore, the ability to isolate stable intermediates like the benzoxazinone provides a strategic buffer, allowing for inventory management strategies that decouple the production of the core scaffold from the final customization steps.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the reduction of byproduct formation lead to substantial cost savings in raw material consumption and waste disposal. By avoiding the use of excessive coupling reagents and minimizing the generation of hard-to-remove impurities, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the use of commodity chemicals like thionyl chloride and triethylamine ensures that the process remains insulated from the price volatility associated with exotic catalysts or proprietary reagents, providing long-term cost stability for procurement managers.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway enhances supply chain continuity by reducing dependency on single-source specialty reagents. The flexibility to use various solvents (toluene, DCM, acetonitrile) and halogenating agents allows production facilities to adapt quickly to regional availability issues or logistical disruptions. This adaptability ensures that production schedules can be maintained even when specific raw materials face temporary shortages, securing the supply of high-purity agrochemical intermediates for global customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, utilizing standard unit operations that are easily transferred from pilot plant to multi-ton manufacturing. The simplified workup procedures, which often involve filtration and washing rather than energy-intensive distillations or chromatography, reduce the energy footprint of the manufacturing process. Moreover, the containment of gaseous byproducts and the potential for solvent recovery align with increasingly stringent environmental regulations, mitigating compliance risks and supporting sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the practical aspects of adopting this technology for commercial production.
Q: How does this novel synthesis route improve upon traditional benzamide production methods?
A: Traditional methods often suffer from moderate yields (58-65%) and complex purification due to direct coupling side reactions. This patented three-step approach isolates the acyl halide and utilizes a controlled benzoxazinone intermediate, significantly enhancing purity and overall process efficiency.
Q: What are the critical safety considerations for the acyl halogenation step?
A: The acyl halogenation step employs reagents such as thionyl chloride or solid phosgene, which require strict temperature control between -20°C and reflux. Proper ventilation and scrubbing systems are essential to manage acidic gas byproducts safely during commercial scale-up.
Q: Can this process be adapted for different substituents on the pyrazole ring?
A: Yes, the methodology is highly versatile. The patent explicitly covers a broad range of substituents (R1-R8) including halogens, alkyl groups, and cyano groups, allowing for the customization of the insecticidal profile without altering the core synthetic workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Pyrazole Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation crop protection solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our expertise in handling reactive intermediates and managing complex multi-step synpositions positions us as a strategic partner for companies seeking to secure their supply of advanced agrochemical building blocks.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements and quality targets. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this optimized process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
