Advanced Lipase-Catalyzed Synthesis of Chiral 3-Hydroxy Acids for Commercial Scale-up
Advanced Lipase-Catalyzed Synthesis of Chiral 3-Hydroxy Acids for Commercial Scale-up
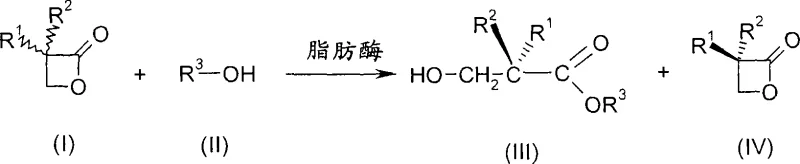
The synthesis of optically active 3-hydroxycarboxylic acids and their esters represents a critical challenge in the development of high-value pharmaceutically active ingredients, where stereochemical purity is paramount for biological efficacy. Patent CN1993472A introduces a groundbreaking methodology for the preparation of essentially enantiomerically pure 3-hydroxypropionic acids or esters of formula (III) through the enantioselective opening of 3-substituted oxetan-2-ones. This innovative approach utilizes lipases derived from Candida antarctica or Burkholderia plantarii to catalyze the reaction between racemic oxetan-2-ketones of formula (I) and compounds of formula (II), effectively resolving the racemic mixture into valuable chiral products. By leveraging biocatalysis, this technology addresses the longstanding limitations of transition metal-catalyzed routes, offering a sustainable and highly selective pathway that aligns with modern green chemistry principles and the rigorous demands of global regulatory bodies for impurity control in drug substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral 3-hydroxy acids has relied heavily on the catalytic hydrogenation of ketones and ketoesters using ruthenium-diphosphine complexes, a method documented extensively in literature such as works by Burk and Noyori. These conventional processes suffer from significant drawbacks, primarily the requirement for very complex and expensive catalysts and ligands that drive up the raw material costs substantially. Furthermore, alternative transfer hydrogenation methods often utilize formic acid or isopropanol as reducing agents, which inevitably lead to the formation of undesirable byproducts like acetone or carbon dioxide that interfere with reaction progress and complicate purification. The use of large amounts of heavy metal catalysts also necessitates stringent and costly removal steps to meet residual metal specifications required for pharmaceutical applications, creating a bottleneck in manufacturing efficiency. Additionally, previous attempts at lipase-catalyzed transesterification of substituted oxetan-2-ones, such as those described by Sakai et al., failed to achieve completely satisfactory results in terms of both chemical yield and optical purity, leaving a gap in reliable synthetic methodology.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent employs specific lipases to achieve high levels of enantioselectivity under mild reaction conditions, effectively bypassing the need for precious metal catalysts. The process involves reacting racemic oxetan-2-ones with water or alcohols in the presence of lipases from Candida antarctica or Burkholderia plantarii, which selectively open the lactone ring of one enantiomer while leaving the other intact. This kinetic resolution strategy allows for the separation of the desired 3-hydroxy acid or ester (Formula III) from the unreacted lactone (Formula IV) based on their distinct chemical properties, such as solubility differences in aqueous versus organic phases. The use of immobilized enzymes, particularly commercially available preparations like Novozym® 435, further enhances the robustness of the process by facilitating catalyst recovery and reuse. As illustrated in the reaction scheme below, this biocatalytic route provides a direct and efficient entry into chiral building blocks with exceptional optical purity, often exceeding 99% ee, thereby eliminating the need for extensive recrystallization or chromatographic purification steps typically associated with chemical synthesis.
Mechanistic Insights into Lipase-Catalyzed Enantioselective Ring-Opening
The core mechanism driving this transformation relies on the precise stereospecificity of the lipase active site, which discriminates between the enantiomers of the racemic 3-substituted oxetan-2-one substrate. When the racemic mixture interacts with the enzyme, the lipase preferentially binds and catalyzes the nucleophilic attack by water or alcohol (R3-OH) on the carbonyl carbon of only one specific enantiomer of the lactone ring. This selective acylation or hydrolysis results in the formation of the open-chain 3-hydroxy carboxylic acid or ester (Formula III) with a defined configuration, while the opposing enantiomer remains largely unreacted as the cyclic lactone (Formula IV). The choice of enzyme source is critical, as lipases from Candida antarctica and Burkholderia plantarii exhibit opposite stereochemical preferences, allowing manufacturers to access either the (R)- or (S)-configured product simply by switching the biocatalyst. This flexibility is invaluable for process chemists who may need to synthesize different diastereomers for structure-activity relationship studies or specific drug candidates without redesigning the entire synthetic route. The reaction proceeds efficiently in solvents like methyl tert-butyl ether or diisopropyl ether, which help suppress competing side reactions such as dimerization, ensuring that the kinetic resolution remains the dominant pathway throughout the conversion.
Controlling impurities in this system is inherently managed by the high specificity of the biocatalyst, which minimizes the formation of regioisomers or over-reaction byproducts common in harsh chemical environments. However, since no lipase operates with absolute 100% stereoselectivity, a small proportion of the undesired enantiomer is inevitably formed over extended reaction times, creating a trade-off between yield and optical purity. To mitigate this, the process parameters are optimized by monitoring reaction kinetics in preliminary laboratory experiments to determine the ideal stopping point that maximizes enantiomeric excess without sacrificing too much conversion. The separation of products is facilitated by their structural differences; for instance, if the product is a free acid, it can be extracted into an aqueous base layer as a salt, leaving the neutral unreacted lactone in the organic phase. This straightforward workup procedure not only simplifies isolation but also ensures that the final product meets the stringent purity specifications required for high-purity pharmaceutical intermediates, reducing the burden on downstream processing units.
How to Synthesize 3-Hydroxy Carboxylic Acids Efficiently
To implement this synthesis effectively, operators must carefully control the reaction environment to balance enzyme activity with substrate stability. The process begins by dissolving the racemic oxetan-2-one precursor in a suitable alkyl ether solvent, followed by the addition of the immobilized lipase and the nucleophile, typically water or a specific alcohol depending on whether the free acid or ester is the target. Maintaining the temperature within the optimal range of 10-50°C is crucial to ensure sufficient enzyme activity while preventing thermal denaturation, with 25°C often cited as a particularly effective condition for balancing reaction rate and selectivity. Detailed standard operating procedures regarding reagent ratios, stirring speeds, and filtration methods are essential for reproducibility, especially when scaling from gram-scale laboratory trials to multi-kilogram production batches. For a comprehensive breakdown of the specific experimental conditions and workup protocols validated in the patent examples, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by dissolving racemic 3-substituted oxetan-2-one in a suitable alkyl ether solvent such as methyl tert-butyl ether.
- Add the specific lipase catalyst, either from Candida antarctica or Burkholderia plantarii, along with the nucleophile (water or alcohol) to initiate the ring-opening.
- Maintain the reaction temperature between 10-50°C and monitor conversion to separate the resulting hydroxy acid ester from the unreacted lactone via extraction.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this enzymatic technology offers profound advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical industry. By replacing expensive transition metal catalysts with robust, immobilized enzymes, the process eliminates the need for costly metal scavenging resins and the associated waste disposal fees, leading to substantial cost savings in raw material consumption. The mild reaction conditions also reduce energy consumption compared to high-pressure hydrogenation processes, contributing to a lower overall carbon footprint and aligning with corporate sustainability goals. Furthermore, the ability to recover and potentially reuse the immobilized lipase catalyst enhances the economic viability of the process, making it a highly attractive option for long-term manufacturing contracts where margin protection is critical. The simplicity of the workup, involving basic extraction and distillation rather than complex chromatography, shortens the production cycle time and increases the throughput capacity of existing manufacturing facilities without requiring significant capital investment in new equipment.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as ruthenium complexes removes a major cost driver from the bill of materials, as these metals are subject to volatile market pricing and supply constraints. Additionally, the avoidance of toxic byproducts like acetone or carbon dioxide simplifies the effluent treatment process, reducing the operational expenditure associated with environmental compliance and waste management. The high selectivity of the enzyme minimizes the formation of difficult-to-remove impurities, which in turn reduces the loss of yield during purification steps and improves the overall mass balance of the synthesis. These factors combined result in a significantly more economical process that allows for competitive pricing strategies in the global market for chiral intermediates.
- Enhanced Supply Chain Reliability: Utilizing commercially available enzymes like Novozym® 435 ensures a stable and reliable supply of the critical catalytic component, mitigating the risk of production delays caused by custom catalyst synthesis bottlenecks. The robustness of the biocatalytic process allows for flexible manufacturing schedules, as the reaction can be run in either batch or continuous modes depending on demand fluctuations. Moreover, the use of common solvents like methyl tert-butyl ether simplifies logistics and inventory management, as these materials are readily available from multiple suppliers worldwide. This resilience in the supply chain is crucial for maintaining continuity of supply for key pharmaceutical customers who require just-in-time delivery of critical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, with patent examples demonstrating successful operation in stirred tank reactors, indicating readiness for commercial scale-up of complex pharmaceutical intermediates. The use of biocatalysts aligns with green chemistry principles by operating at ambient temperatures and pressures, thereby reducing the safety risks associated with high-energy chemical processes. The aqueous workup and lack of heavy metal residues simplify the regulatory filing process for new drug applications, as the impurity profile is cleaner and easier to characterize. This environmental and regulatory friendliness makes the technology future-proof against tightening global regulations on chemical manufacturing emissions and residual contaminants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this lipase-catalyzed technology, based on the specific data and embodiments disclosed in the patent documentation. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their specific pipeline candidates. The answers provided reflect the proven capabilities of the method as demonstrated in the experimental examples, ensuring that expectations are grounded in verified scientific data.
Q: What are the advantages of using lipase over ruthenium catalysts for this transformation?
A: Unlike traditional ruthenium-diphosphine complexes which require complex ligands and generate byproducts like acetone or carbon dioxide, the lipase-catalyzed method operates under mild conditions without heavy metal contamination, significantly simplifying downstream purification.
Q: Can both enantiomers of the 3-hydroxy acid be accessed using this method?
A: Yes, the patent demonstrates that selecting between Candida antarctica lipase and Burkholderia plantarii lipase allows for the production of opposite stereochemical configurations, providing flexibility in synthesizing specific chiral intermediates.
Q: Is this process suitable for large-scale industrial production?
A: The method supports both batch and continuous synthesis modes, particularly when using carrier-bound immobilized enzymes, which enhances catalyst stability and facilitates scalability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this lipase-catalyzed technology for producing high-value chiral intermediates and are fully equipped to bring this innovation to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-hydroxy carboxylic acids or esters meets the highest standards of enantiomeric excess and chemical purity required by top-tier pharmaceutical companies. Our commitment to quality and technical excellence makes us the ideal partner for organizations seeking to leverage advanced biocatalytic routes for their API synthesis.
We invite you to engage with our technical procurement team to discuss how this specific enzymatic route can be tailored to your project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free methodology for your specific molecule. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this reliable pharmaceutical intermediate supplier into your global supply chain network.
