Advanced Titanium-Catalyzed Route for High-Purity Beta-Aminocarboxylate Manufacturing
Introduction to Next-Generation Beta-Aminocarboxylate Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to access critical building blocks, and the recent disclosure in patent CN113200881A represents a significant leap forward in this domain. This groundbreaking technology introduces a novel catalytic system utilizing pentamethyltrichlorotitanocene (Cp*TiCl3)协同 4-hydroxybenzoic acid to synthesize beta-aminocarboxylate derivatives with exceptional precision. Beta-aminocarboxylates are ubiquitous motifs in medicinal chemistry, serving as pivotal intermediates for a vast array of bioactive molecules, including antibiotics, enzyme inhibitors, and peptidomimetics. The traditional reliance on stoichiometric reagents or harsh acidic conditions has long plagued the supply chain, creating bottlenecks in purity and throughput. By leveraging a synergistic dual-acid catalytic approach, this new method achieves high atom economy under remarkably mild conditions, specifically at room temperature in ethanol. For R&D directors and procurement managers alike, this innovation signals a shift towards greener chemistry that does not compromise on yield or scalability, offering a robust solution for the reliable pharmaceutical intermediate supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-aminocarboxylate derivatives has relied heavily on the classical Mannich reaction or variations employing strong Lewis and Bronsted acids. Conventional protocols often utilize catalysts such as iron(III) chloride, scandium triflate, or strong protic acids like trifluoroacetic acid, which frequently necessitate stringent reaction parameters. These traditional methods suffer from several inherent drawbacks that impact both the economic and environmental viability of large-scale manufacturing. Firstly, many Lewis acid catalysts are moisture-sensitive, requiring anhydrous solvents and inert atmospheres, which drastically increases operational costs and complexity. Secondly, the use of strong acids can lead to poor chemoselectivity, resulting in complex impurity profiles that are difficult and expensive to remove during downstream processing. Furthermore, the substrate scope is often limited; electron-deficient or sterically hindered substrates may react sluggishly or not at all, forcing chemists to explore multiple alternative routes. The reliance on volatile organic solvents like dichloromethane or THF further exacerbates environmental concerns and regulatory compliance burdens, making these conventional pathways increasingly unattractive for modern, sustainability-focused chemical enterprises.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN113200881A employs a sophisticated synergistic catalytic system that overcomes these historical barriers with elegance and efficiency. By combining pentamethyltrichlorotitanocene as a Lewis acid catalyst with 4-hydroxybenzoic acid acting as a ligand or co-catalyst, the reaction proceeds smoothly in ethanol, a green and inexpensive solvent. This dual-activation strategy allows for the condensation of benzaldehyde derivatives, aniline compounds, and silyl ketene acetals to occur at room temperature, eliminating the need for energy-intensive heating or cooling cycles. The mildness of the conditions preserves sensitive functional groups, thereby expanding the applicable substrate scope to include nitro, halo, and alkoxy-substituted aromatics without degradation. Moreover, the reaction exhibits high atom economy, meaning that a greater proportion of the starting materials are incorporated into the final product, reducing waste generation. The simplicity of the workup procedure, which often requires only rotary evaporation followed by standard column chromatography, significantly reduces the time and resources needed for purification, presenting a compelling value proposition for cost reduction in API manufacturing.
Mechanistic Insights into Cp*TiCl3-Catalyzed Mannich-Type Reaction
The success of this synthetic methodology lies in the unique mechanistic interplay between the titanium center and the carboxylic acid ligand, which creates a highly active yet selective catalytic environment. The pentamethylcyclopentadienyl titanium trichloride acts as a potent Lewis acid, coordinating to the carbonyl oxygen of the in situ generated imine or the aldehyde itself, thereby increasing its electrophilicity. Simultaneously, the 4-hydroxybenzoic acid likely functions through hydrogen bonding or proton transfer mechanisms, stabilizing the transition state and facilitating the nucleophilic attack by the silyl ketene acetal. This cooperative activation lowers the activation energy barrier significantly, allowing the reaction to proceed rapidly even at ambient temperatures. The steric bulk of the pentamethylcyclopentadienyl ligand also plays a crucial role in shielding the metal center, preventing unwanted side reactions such as polymerization or over-alkylation, which ensures the formation of a single, well-defined product. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters, as it highlights the importance of maintaining the precise molar ratios of the catalyst and ligand to maximize turnover frequency.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over non-catalytic or harshly catalytic alternatives. The high selectivity of the titanium system minimizes the formation of regioisomers or byproducts derived from the decomposition of the silyl ketene acetal, which is a common issue in acid-catalyzed reactions. The use of ethanol as a solvent further aids in suppressing side reactions, as it is less likely to participate in competing nucleophilic attacks compared to more reactive solvents. Consequently, the crude reaction mixture is typically cleaner, simplifying the purification process and leading to higher overall recovery rates of the target beta-aminocarboxylate. For quality assurance teams, this translates to a more consistent impurity profile across different batches, which is a critical factor for regulatory filings and maintaining supply chain reliability. The ability to tolerate diverse functional groups without protecting group manipulation further streamlines the synthetic route, reducing the total number of steps and associated material losses.
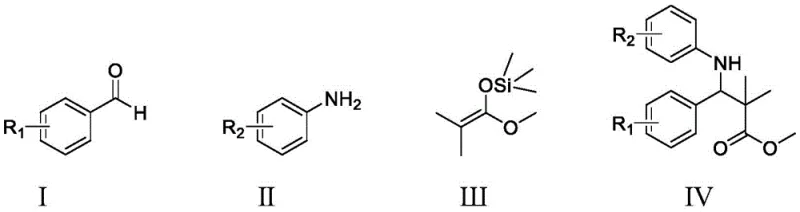
How to Synthesize Beta-Aminocarboxylate Efficiently
Implementing this advanced catalytic protocol in a laboratory or pilot plant setting requires careful attention to reagent quality and mixing dynamics to ensure reproducibility. The process begins with the preparation of the reaction mixture, where the benzaldehyde, aniline, and silyl ketene acetal are combined in a specific stoichiometric ratio, typically favoring a slight excess of the aldehyde to drive the equilibrium forward. The addition of the catalytic system, comprising 1 to 5 percent of the titanium complex and 2 to 10 percent of the hydroxybenzoic acid relative to the aniline, is the critical step that initiates the transformation. Stirring at room temperature for a period of 10 to 12 hours allows the reaction to reach completion, after which the solvent is removed under reduced pressure. The detailed standardized synthesis steps, including specific workup procedures and purification gradients, are outlined in the guide below to assist technical teams in replicating these high-yielding results.
- Charge a reaction flask with o-aminophenol, benzaldehyde derivative, and silylketene acetal in a molar ratio of approximately 1: 1.4:1.
- Add pentamethyltrichlorotitanocene (1-5 mol%) and 4-hydroxybenzoic acid (2-10 mol%) as the catalytic system into ethanol solvent.
- Stir the mixture at room temperature for 10-12 hours, then remove solvent and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this titanium-catalyzed synthesis route offers tangible strategic benefits that extend beyond mere chemical curiosity. The shift from harsh, moisture-sensitive Lewis acids to a robust, air-stable titanium system significantly de-risks the manufacturing process, reducing the likelihood of batch failures due to environmental exposure. This reliability is paramount for maintaining continuous production schedules and meeting tight delivery windows for downstream API manufacturers. Furthermore, the use of ethanol as the primary solvent aligns with global trends towards greener chemistry, potentially lowering waste disposal costs and simplifying regulatory compliance regarding volatile organic compound (VOC) emissions. The simplified workup procedure, which avoids complex aqueous extractions or specialized quenching steps, reduces the consumption of auxiliary chemicals and labor hours, contributing to a leaner and more cost-efficient operation. These factors collectively enhance the overall resilience of the supply chain, ensuring a steady flow of high-quality intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents, coupled with the ability to run reactions at room temperature, leads to significant energy savings and reduced raw material costs. By avoiding the need for cryogenic conditions or high-pressure reactors, capital expenditure on specialized equipment is minimized, while the high atom economy ensures that valuable starting materials are not wasted. The streamlined purification process further reduces the cost of goods sold by shortening cycle times and improving yield retention. Qualitative analysis suggests that the removal of transition metal contaminants is also simpler, avoiding the need for costly scavenging resins often required with other metal catalysts.
- Enhanced Supply Chain Reliability: The broad substrate scope demonstrated in the patent examples indicates that this method can accommodate various substituted benzaldehydes and anilines, providing flexibility in sourcing raw materials. If a specific starting material becomes scarce or expensive, the robustness of the catalytic system allows for the rapid qualification of alternative suppliers or analogues without re-optimizing the entire process. The stability of the catalyst system also implies a longer shelf life for prepared reagents, reducing inventory write-offs. This adaptability is crucial for mitigating supply chain disruptions and ensuring business continuity in a volatile global market.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or tonne scale is facilitated by the exothermic nature of the reaction being manageable at room temperature, removing the need for complex heat exchange systems. The use of ethanol, a renewable and biodegradable solvent, greatly simplifies wastewater treatment and aligns with corporate sustainability goals. The high selectivity of the reaction minimizes the generation of hazardous byproducts, reducing the burden on environmental health and safety teams. This eco-friendly profile not only meets current regulatory standards but also future-proofs the manufacturing process against tightening environmental legislation.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation of this synthesis method, we have compiled a set of questions based on the experimental data and mechanistic understanding of the patent. These answers provide clarity on catalyst loading, solvent selection, and substrate compatibility, helping technical teams make informed decisions about process adoption. Understanding these nuances is essential for successfully translating laboratory results into commercial production, ensuring that the full potential of this catalytic system is realized in an industrial setting.
Q: What are the advantages of using Cp*TiCl3 over traditional Lewis acids?
A: Unlike traditional Lewis acids like FeCl3 or Sc(OTf)3 which may require harsh conditions, the Cp*TiCl3 system operates at room temperature with high atom economy and selectivity.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the use of ethanol as a green solvent and room temperature conditions makes this process highly suitable for commercial scale-up without complex safety measures.
Q: What is the typical yield range for this catalytic system?
A: Experimental data indicates yields ranging from 66% to 93% depending on the electronic nature of the substituents on the benzaldehyde and aniline substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Aminocarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this titanium-catalyzed technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of organometallic catalysis, with rigorous QC labs dedicated to maintaining stringent purity specifications for every batch. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to quality assurance guarantees that the beta-aminocarboxylate derivatives we produce meet the highest industry standards for identity, strength, and purity.
We invite you to collaborate with us to leverage this innovative synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to our deep expertise in process optimization and supply chain management. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline while optimizing your manufacturing costs.
