Advanced Prochlorperazine Manufacturing: Overcoming Genotoxic Impurity Challenges via Sequence Reversal
Advanced Prochlorperazine Manufacturing: Overcoming Genotoxic Impurity Challenges via Sequence Reversal
The pharmaceutical industry constantly seeks robust synthetic routes that ensure patient safety while optimizing manufacturing efficiency. A significant breakthrough in this domain is detailed in patent CN108530384B, which discloses a novel preparation method for Prochlorperazine, a critical piperazine phenothiazine derivative widely used as an antiemetic and antipsychotic agent. This innovative approach fundamentally alters the traditional reaction sequence, addressing long-standing issues regarding incomplete reactions and the presence of genotoxic impurities that plague conventional synthesis pathways. By prioritizing the alkylation of the phenothiazine core before introducing the piperazine moiety, this method achieves a reaction completion rate that was previously difficult to attain, thereby ensuring the final product meets stringent medicinal grade purity specifications. For global procurement teams and R&D directors, understanding this technological shift is vital for securing a reliable supply of high-quality active pharmaceutical ingredients.
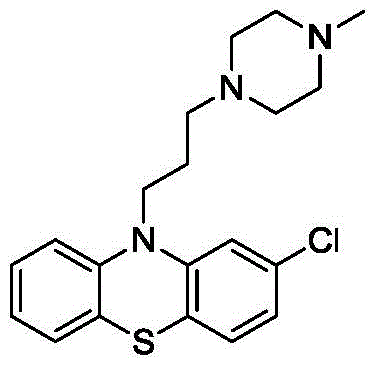
Prochlorperazine serves as a cornerstone therapy for managing acute and chronic psychosis, as well as preventing chemotherapy-induced nausea and vomiting (CINV). The structural integrity and purity of this molecule are paramount, given its potent pharmacological activity. The traditional manufacturing landscape has often been hindered by the formation of hazardous byproducts that require complex and costly purification steps to remove. The methodology outlined in the referenced patent offers a streamlined solution that not only enhances product safety but also simplifies the downstream processing requirements. As a leading entity in fine chemical manufacturing, analyzing such patents allows us to align our production capabilities with the most advanced and compliant synthetic strategies available in the market today.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prochlorperazine has relied on a sequence where the side chain is constructed first. This conventional route involves reacting 1-bromo-3-chloropropane with N-methylpiperazine to form 1-(3-chloropropyl)-4-methylpiperazine, which is subsequently coupled with 2-chlorophenothiazine under alkaline conditions. While conceptually straightforward, this pathway suffers from severe technical defects that impact both yield and safety profiles. The second step of this traditional route frequently fails to proceed to completion, resulting in significant residual amounts of starting materials, specifically 2-chlorophenothiazine and the chloropropyl-piperazine intermediate. More critically, the residual 1-(3-chloropropyl)-4-methylpiperazine is classified as a fatty chloride with known genotoxicity.
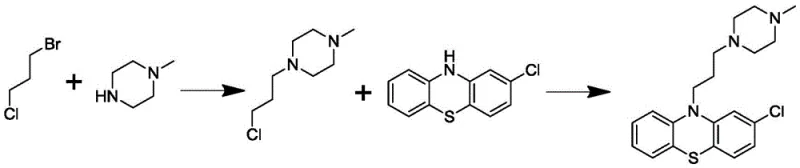
The presence of genotoxic impurities in pharmaceutical products is subject to extremely strict regulatory limits, often in the parts-per-million (ppm) range. The inability of the conventional method to fully consume this genotoxic precursor places an immense burden on the subsequent refining and purification processes. Manufacturers are forced to implement rigorous and expensive cleaning procedures to reduce these impurities to acceptable levels, which inevitably drives up production costs and extends lead times. Furthermore, the incomplete reaction necessitates the use of excess reagents and energy, creating inefficiencies that are unsustainable for large-scale commercial operations. These limitations highlight the urgent need for a process redesign that inherently prevents the formation or persistence of such hazardous species.
The Novel Approach
The inventive method described in patent CN108530384B elegantly resolves these issues by reversing the order of synthetic operations. In this optimized route, 1-bromo-3-chloropropane is first reacted with 2-chlorophenothiazine to generate the intermediate 2-chloro-10-(3-chloropropyl)-phenothiazine. This intermediate is then subjected to a second reaction with N-methylpiperazine to yield the final Prochlorperazine product. This sequence reversal is not merely a procedural change; it is a strategic maneuver that fundamentally alters the impurity profile of the reaction mixture. By introducing the N-methylpiperazine in the final step, any unreacted alkyl halide is scavenged by the excess piperazine, converting potential genotoxic residues into 1,3-bis(4-methylpiperazinyl)propane, a common and non-genotoxic impurity.
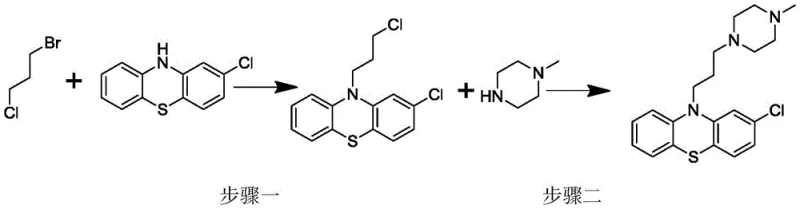
This novel approach ensures that the reaction proceeds to completion with high efficiency, eliminating the persistent residue problems associated with the prior art. The crude product obtained through this method contains only trace amounts of common impurities, such as 2-chlorophenothiazine and the bis-piperazine derivative, both of which are easily controlled to levels below 0.5%. Consequently, the difficulty of the subsequent refining process is drastically reduced, as there is no need for specialized protocols to remove genotoxic contaminants. This results in a cleaner, safer, and more cost-effective manufacturing process that aligns perfectly with modern Good Manufacturing Practice (GMP) standards and regulatory expectations for API intermediates.
Mechanistic Insights into N-Alkylation and Impurity Control
The success of this novel synthesis relies heavily on the precise control of reaction conditions during the initial alkylation of the phenothiazine ring. The process begins with the deprotonation of 2-chlorophenothiazine using a strong base, such as potassium tert-butoxide or sodium hydride, in a suitable solvent like tetrahydrofuran (THF) or dimethylformamide (DMF). This step generates the reactive phenothiazine salt, which must be handled under inert atmosphere protection (nitrogen or argon) to prevent oxidation of the sulfur-nitrogen heterocycle. The subsequent addition of 1-bromo-3-chloropropane at controlled temperatures between -5°C and 40°C facilitates a nucleophilic substitution reaction. Maintaining the temperature within the optimal range of 10°C to 20°C is crucial; excessive heat can trigger elimination reactions, leading to the formation of unwanted byproducts like 2-chloro-10-(2-propenyl)-phenothiazine.
In the second stage, the mechanistic advantage becomes even more apparent regarding impurity management. The intermediate 2-chloro-10-(3-chloropropyl)-phenothiazine reacts with a significant excess of N-methylpiperazine, typically in a molar ratio ranging from 2:1 to 20:1, with 5:1 to 6:1 being preferred. This excess serves a dual purpose: it acts as both the nucleophile for the coupling reaction and as an acid-binding agent to neutralize the hydrogen bromide generated. Crucially, if any 1-bromo-3-chloropropane remains from the first step, the high concentration of N-methylpiperazine ensures it reacts immediately to form the bis-piperazine derivative rather than persisting as a genotoxic hazard. The reaction is driven to completion by heating the mixture to 85-95°C for 4 to 5 hours, a condition that is both energetically favorable and kinetically efficient compared to the harsher conditions required by older methods.
How to Synthesize Prochlorperazine Efficiently
Implementing this advanced synthetic route requires careful attention to reagent quality and thermal management to maximize yield and purity. The process is designed to be telescoped, meaning the intermediate from the first step does not require isolation or purification before proceeding to the second step, which significantly reduces solvent usage and processing time. Operators must ensure strict exclusion of moisture and oxygen during the salt formation phase to maintain the reactivity of the phenothiazine anion. Following the coupling reaction, the recovery of excess N-methylpiperazine under reduced pressure allows for material recycling, further enhancing the economic viability of the process. The detailed standardized synthesis steps, including specific workup procedures and quality control checkpoints, are outlined in the guide below.
- Deprotonate 2-chlorophenothiazine with a strong base like potassium tert-butoxide at low temperature to form the salt, then react with 1-bromo-3-chloropropane to generate the chloropropyl intermediate.
- React the crude intermediate directly with excess N-methylpiperazine at elevated temperatures (85-95°C) to complete the second alkylation step efficiently.
- Recover excess N-methylpiperazine under reduced pressure and perform standard aqueous workup to isolate the high-purity prochlorperazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible operational benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the purification workflow. By eliminating the need to remove genotoxic impurities, manufacturers can bypass complex and resource-intensive cleaning stages, leading to a significant reduction in overall production costs. The ability to use the crude intermediate directly in the second step without refinement also minimizes material loss and solvent consumption, contributing to a leaner and more sustainable manufacturing footprint. These efficiencies allow suppliers to offer more competitive pricing structures while maintaining the highest standards of product quality and regulatory compliance.
- Cost Reduction in Manufacturing: The streamlined process eliminates the expensive and time-consuming steps associated with removing genotoxic impurities, which traditionally require specialized chromatography or extensive recrystallization. Furthermore, the recovery and recycling of excess N-methylpiperazine reduce raw material consumption, directly lowering the variable costs per kilogram of produced API. The higher reaction yield, reported at approximately 97.7% for the crude product compared to lower yields in prior art, ensures better utilization of starting materials, maximizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method enhances supply chain stability by reducing the risk of batch failures due to impurity excursions. The reaction conditions are milder, operating at lower temperatures (85-95°C) compared to the 110-115°C required by conventional methods, which reduces stress on equipment and lowers energy consumption. Additionally, the shorter reaction time of 4 to 5 hours for the second step, versus 10 to 14 hours in the old route, accelerates the overall production cycle, enabling faster turnaround times and improved responsiveness to market demand fluctuations.
- Scalability and Environmental Compliance: This method is highly amenable to commercial scale-up, utilizing common industrial solvents like THF and DMF that are readily available in the global supply chain. The reduction in hazardous waste generation, particularly the avoidance of genotoxic waste streams, simplifies environmental compliance and waste disposal logistics. The process generates fewer byproducts and consumes less energy, aligning with green chemistry principles and helping pharmaceutical companies meet their sustainability goals without compromising on the volume or quality of the final therapeutic agent.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Prochlorperazine synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of why this route represents a superior choice for modern pharmaceutical manufacturing. Understanding these details helps stakeholders make informed decisions about sourcing strategies and process validation requirements.
Q: How does the reversed reaction sequence eliminate genotoxic impurities in Prochlorperazine synthesis?
A: By reacting 2-chlorophenothiazine with 1-bromo-3-chloropropane first, the potentially genotoxic side chain intermediate (1-(3-chloropropyl)-4-methylpiperazine) is never formed. Any residual alkyl halide is scavenged by excess N-methylpiperazine in the second step to form a non-genotoxic bis-piperazine derivative.
Q: What are the optimal reaction conditions for the novel Prochlorperazine preparation method?
A: The first alkylation step is best conducted at 10-20°C using solvents like THF or DMF with strong bases. The second coupling step requires heating to 85-95°C for 4-5 hours, which is significantly milder and faster than conventional methods requiring 110-115°C for over 10 hours.
Q: Can the intermediate be purified before the second reaction step?
A: No, the process is designed for telescoping. The dark brown oily intermediate (2-chloro-10-(3-chloropropyl)-phenothiazine) obtained from the first step can be used directly in the second step without refinement, which streamlines the workflow and reduces material loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prochlorperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to ensure the safety and efficacy of pharmaceutical products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel Prochlorperazine route can be fully realized at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch meets the highest international standards, free from the genotoxic impurities that plague older manufacturing techniques. Our commitment to technological excellence allows us to deliver high-purity intermediates that facilitate smoother regulatory approvals for our clients.
We invite global partners to collaborate with us to leverage these process improvements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how our optimized manufacturing capabilities can support your long-term strategic goals in the competitive pharmaceutical marketplace.
