Scalable Manufacturing of 2-Aryl Malonic Acid Derivatives via Metal-Free Aromatization
Scalable Manufacturing of 2-Aryl Malonic Acid Derivatives via Metal-Free Aromatization
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical agrochemical intermediates. Patent CN111372913B introduces a groundbreaking methodology for preparing 2-aryl malonic acid derivatives, which serve as pivotal precursors for advanced crop protection agents such as Pinoxaden. This technology represents a significant departure from conventional reliance on noble metal catalysts and hazardous reagents, offering a streamlined pathway that enhances both economic viability and operational safety. By leveraging a sequential isomerization and halogenation-aromatization strategy, manufacturers can achieve superior purity profiles while drastically reducing the environmental footprint associated with heavy metal waste. The versatility of this approach allows for the accommodation of various substituents on the aromatic ring, making it a robust platform for diverse chemical libraries. Furthermore, the ability to execute these transformations under relatively mild thermal conditions underscores its potential for immediate adoption in large-scale industrial settings. 
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-aryl malonic acid framework has been plagued by significant technical and economic hurdles that hinder efficient commercial production. Traditional strategies often rely on the C-C coupling of halogenated aromatics with malonic acid derivatives, a process that necessitates the use of expensive organometallic catalysts which are difficult to recover and recycle. Another prevalent method involves the condensation of phenylacetic acid derivatives using strong bases like sodium hydride, which poses severe safety risks due to the generation of flammable hydrogen gas and the requirement for strictly anhydrous conditions. Additionally, existing dehydrogenation routes typically employ noble metals such as palladium at excessively high temperatures ranging from 180°C to 200°C, leading to energy inefficiencies and potential thermal degradation of sensitive functional groups. These legacy processes also frequently generate substantial amounts of three-waste pollution, particularly when diazo-halogenation is required to prepare specific halogenated starting materials. Consequently, the industry has long sought a alternative that balances cost, safety, and scalability without compromising on yield.
The Novel Approach
The innovative protocol disclosed in the patent data circumvents these historical bottlenecks by utilizing a cyclohexadiene compound as a readily available starting material for a transformative two-step sequence. Instead of relying on precious metals for dehydrogenation, this method employs a base-catalyzed isomerization followed by a halogenation and dehydrohalogenation aromatization sequence. This strategic shift allows the reaction to proceed under much milder thermal conditions, typically between 0°C and 150°C, thereby preserving the integrity of the molecular structure. The elimination of noble metal catalysts not only reduces raw material costs but also simplifies the downstream purification process by removing the need for rigorous heavy metal scavenging steps. Moreover, the avoidance of pyrophoric bases like sodium hydride significantly enhances the safety profile of the manufacturing plant, reducing the risk of fire and explosion. This novel route effectively converts non-aromatic precursors into high-value aromatic systems with exceptional atom economy and minimal environmental impact. 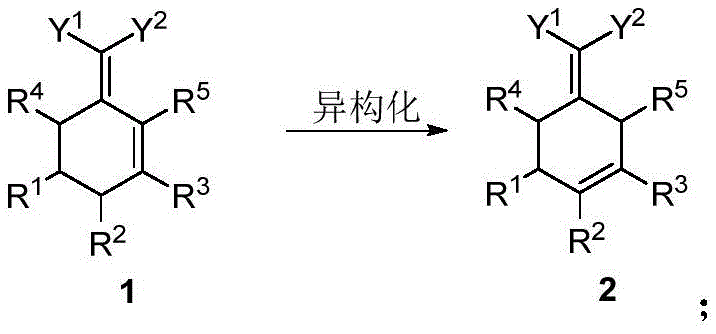
Mechanistic Insights into Base-Catalyzed Isomerization and Halogenation
The first stage of this synthesis involves a precise isomerization reaction where a cyclohexadiene compound is converted into a thermodynamically more stable intermediate through the migration of double bonds. This transformation is facilitated by mild bases such as sodium hydroxide, sodium methylate, or potassium hydroxide, which act as proton shuttles to rearrange the olefinic geometry without inducing unwanted side reactions. The molar ratio of the base to the substrate is carefully optimized, typically ranging from 0.8 to 2.4 equivalents, to ensure complete conversion while minimizing the formation of polymeric byproducts. This step is crucial as it sets the electronic stage for the subsequent aromatization, positioning the double bonds in a configuration that is highly susceptible to electrophilic attack by halogenating agents. The reaction kinetics are favorable at moderate temperatures around 50°C, allowing for rapid equilibration to the desired isomer. By controlling the basicity and temperature, chemists can effectively suppress competing pathways that might lead to ring opening or polymerization, ensuring a clean reaction profile.
Following isomerization, the intermediate undergoes a sophisticated halogenation and dehydrohalogenation sequence to achieve full aromaticity. In this phase, halogenating agents such as chlorine gas, liquid bromine, or sulfuryl chloride introduce halogen atoms onto the cyclic framework, creating a polyhalogenated transient species. Subsequently, a dehydrohalogenation aromatization reaction occurs, often catalyzed by simple alkali metal halides like lithium chloride or sodium chloride. This catalytic system promotes the elimination of hydrogen halide molecules, driving the equilibrium towards the formation of the stable aromatic ring system. The reaction temperature for this aromatization step is typically maintained between 110°C and 150°C, which provides sufficient energy to overcome the activation barrier for elimination without causing thermal decomposition. 
How to Synthesize 2-Aryl Malonic Acid Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and temperature control to maximize the yield of the target 2-aryl malonic acid derivative. The process begins with the dissolution of the cyclohexadiene precursor in a suitable solvent such as methanol or tetrahydrofuran, followed by the controlled addition of the base catalyst to initiate isomerization. Once the isomerization is complete, the reaction mixture is subjected to halogenation, where the flow rate of gaseous halogens or the addition rate of liquid halogenating agents must be meticulously managed to prevent exothermic runaways. The subsequent aromatization step may be performed in a one-pot fashion or as a distinct stage, depending on the specific solvent compatibility and purification requirements of the facility. Detailed standard operating procedures regarding quenching, extraction, and crystallization are essential to isolate the product with the high purity demanded by agrochemical regulations.
- Perform an isomerization reaction on a cyclohexadiene compound using a mild base such as sodium methoxide or potassium hydroxide at moderate temperatures to obtain the isomerized intermediate.
- Subject the intermediate to a halogenation reaction using agents like chlorine or sulfuryl chloride, followed by a dehydrohalogenation aromatization step.
- Complete the aromatization in the presence of an alkali metal halide catalyst at elevated temperatures to yield the final 2-aryl malonic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers compelling strategic advantages that directly impact the bottom line and operational resilience. The most significant benefit lies in the drastic reduction of raw material costs achieved by eliminating the dependency on volatile and expensive noble metal catalysts like palladium or platinum. By substituting these precious metals with abundant and inexpensive alkali metal halides, manufacturers can secure a more stable cost structure that is less susceptible to fluctuations in the global commodities market. Furthermore, the removal of hazardous reagents such as sodium hydride simplifies logistics and storage requirements, as there is no longer a need for specialized inert atmosphere handling or explosive gas monitoring systems. This simplification translates into lower insurance premiums and reduced capital expenditure on safety infrastructure. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a more sustainable and cost-effective manufacturing operation overall.
- Cost Reduction in Manufacturing: The exclusion of expensive transition metal catalysts removes the necessity for complex and costly metal scavenging units, which are traditionally required to meet strict residual metal limits in pharmaceutical and agrochemical products. This streamlining of the purification train reduces both the consumption of auxiliary materials and the time required for batch processing. Additionally, the use of common halogenating agents and simple salt catalysts ensures that the supply chain for reagents is robust and widely available, preventing production delays caused by specialty chemical shortages. The high yields reported in the patent examples further contribute to cost efficiency by maximizing the output per unit of raw material input. Overall, the process economics are significantly improved through the minimization of waste disposal costs associated with heavy metal sludge.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as chlorine, bromine, and sodium methoxide, the manufacturing process becomes decoupled from the supply constraints often associated with specialized organometallic reagents. This accessibility ensures that production schedules can be maintained consistently, even during periods of global supply chain disruption. The inherent safety of the process, characterized by the absence of pyrophoric materials and high-pressure hydrogen generation, reduces the likelihood of unplanned shutdowns due to safety incidents. Moreover, the potential for one-pot processing reduces the number of intermediate isolation steps, thereby shortening the overall cycle time from raw material intake to finished goods. This agility allows suppliers to respond more rapidly to fluctuating market demands for herbicide intermediates.
- Scalability and Environmental Compliance: The mild thermal profile of this synthesis makes it inherently easier to scale from pilot plant to multi-ton commercial production without encountering the heat transfer limitations often seen in high-temperature dehydrogenation reactions. The absence of heavy metals simplifies wastewater treatment protocols, as effluent streams do not require extensive remediation to remove toxic metal ions before discharge. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing entity. The reduction in hazardous waste generation also lowers the long-term liability and disposal costs for the facility. Consequently, this technology supports the long-term viability of the supply chain by adhering to increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology for 2-aryl malonic acid derivatives. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation to assist decision-makers in evaluating the feasibility of adoption. Understanding the nuances of catalyst selection, safety protocols, and yield optimization is critical for successful technology transfer.
Q: What are the primary safety advantages of this new synthesis method compared to traditional routes?
A: This method eliminates the need for hazardous strong bases like sodium hydride, which generates explosive hydrogen gas, and avoids the use of expensive, toxic noble metal catalysts like palladium, significantly improving operational safety.
Q: Can this process be adapted for large-scale industrial production of herbicide intermediates?
A: Yes, the process utilizes mild reaction conditions, common solvents, and inexpensive catalysts like lithium chloride, making it highly suitable for commercial scale-up without the complex waste treatment associated with heavy metals.
Q: What is the typical yield profile for the conversion of cyclohexadiene compounds to the final aromatic product?
A: Experimental data indicates high efficiency, with yields often exceeding 80% to 90% across both the isomerization and the subsequent halogenation-aromatization steps, ensuring minimal raw material waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl Malonic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free aromatization technology in securing a competitive edge for our global partners in the agrochemical sector. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-aryl malonic acid derivative delivered meets the highest standards of quality required for herbicide synthesis. We are committed to leveraging our technical expertise to optimize this specific route, ensuring maximum yield and minimal impurity formation for your specific application needs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. Together, we can drive the next generation of efficient and sustainable agrochemical manufacturing.
