Revolutionizing Bactericide Intermediate Production via One-Pot Synthesis and Commercial Scale-Up
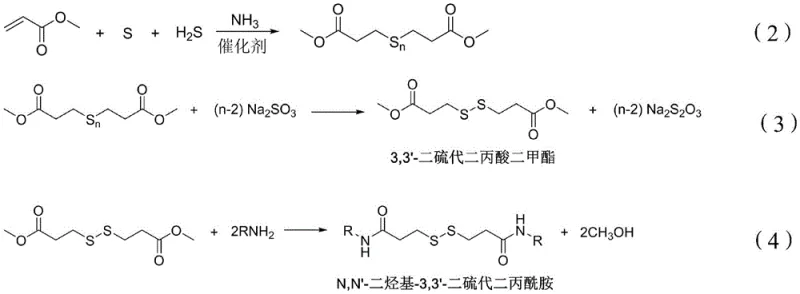
The chemical industry is witnessing a transformative shift in the production of critical bactericide precursors, driven by the innovative methodology disclosed in patent CN115838346A. This patent introduces a highly efficient one-pot synthesis route for N,N'-dialkyl-3,3'-dithiodipropionamide, a pivotal intermediate in the manufacture of broad-spectrum 3-isothiazolinone biocides such as MIT, CMIT, and OIT. Traditional manufacturing pathways have long been plagued by multi-step isolation procedures and significant environmental burdens, creating bottlenecks for reliable agrochemical intermediate supplier networks globally. The disclosed technology leverages a seamless integration of vulcanization, reduction, and aminolysis reactions within a unified operational framework, drastically simplifying the process flow. By utilizing readily available feedstocks like methyl acrylate, sulfur, and hydrogen sulfide, this method not only enhances reaction efficiency but also ensures the generation of high-purity intermediates essential for downstream pharmaceutical and industrial applications. For R&D directors and procurement strategists, understanding this technological leap is crucial for optimizing supply chains and achieving substantial cost reduction in biocide manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N'-dialkyl-3,3'-dithiodipropionamide has relied on fragmented processes that involve the separate preparation and isolation of dimethyl 3,3'-dithiodipropionate. Conventional techniques typically require the reaction of dimethyl 3,3'-dithiodipropionate with hydrocarbyl amines in the presence of solvents like toluene or methanol, often necessitating rigorous temperature controls and extended reaction times spanning several days. A significant drawback of these legacy methods is the requirement for solid-liquid separation and drying operations to isolate the intermediate, which introduces complexity and increases the risk of product degradation or impurity incorporation. Furthermore, existing patents describe yields that fluctuate significantly, often hovering around 60% to 76%, indicating substantial material loss and inefficiency in raw material utilization. The generation of large volumes of wastewater and the difficulty in recycling solvents further exacerbate the environmental footprint, making compliance with modern green chemistry standards increasingly challenging for manufacturers relying on these outdated protocols.
The Novel Approach
In stark contrast, the novel one-pot method described in the patent data revolutionizes the production landscape by integrating the synthesis into a continuous, streamlined workflow that eliminates the need for intermediate isolation. This approach utilizes a strategic sequence where methyl acrylate undergoes vulcanization with sulfur and hydrogen sulfide, followed immediately by a reduction step using sodium sulfite to generate the disulfide ester in situ. The resulting oil layer is directly subjected to aminolysis with alkylamines, bypassing the energy-intensive drying and solid-handling steps characteristic of traditional routes. This integration not only accelerates the overall production cycle but also facilitates the recycling of byproducts, such as sodium thiosulfate from the aqueous layer, thereby closing the loop on waste generation. The operational simplicity allows for easier scale-up and reduces the dependency on complex equipment, offering a robust solution for the commercial scale-up of complex polymer additives and biocide intermediates alike.
Mechanistic Insights into One-Pot Vulcanization and Aminolysis
The core of this technological advancement lies in the precise control of the vulcanization and reduction mechanisms that dictate the purity of the final disulfide structure. The process initiates with the reaction of methyl acrylate, sulfur, and a catalytic amount of water at mild temperatures between 10°C and 20°C, where ammonia and hydrogen sulfide gases are introduced to form a polysulfide intermediate known as dimethyl polythiodipropionate. This step is critical as it establishes the carbon-sulfur backbone, and the reaction is monitored via gas phase detection to ensure methyl acrylate consumption drops below 0.5%, guaranteeing high conversion rates before proceeding. Subsequently, the addition of deionized water and sodium sulfite at 40°C to 60°C triggers a selective reduction reaction that cleaves the polysulfide chains into the desired 3,3'-dithiodipropionate structure while generating sodium thiosulfate as a recoverable byproduct. This mechanistic precision ensures that the oil layer obtained is rich in the target ester, minimizing the formation of higher molecular weight sulfur impurities that could complicate downstream purification.
Following the formation of the disulfide ester, the mechanism shifts to a nucleophilic acyl substitution during the aminolysis phase, where the ester groups are displaced by alkylamines to form the final amide bonds. The reaction is conducted in a methanol solution at controlled temperatures of 10°C to 30°C, allowing for the gradual conversion of the ester to N,N'-dialkyl-3,3'-dithiodipropionamide over a period of 15 to 25 hours. The use of methanol serves a dual purpose as both a solvent and a reactant medium, facilitating the dissolution of the amine and ensuring homogeneous reaction conditions throughout the mixture. Upon completion, indicated by the residual ester content falling below 0.5%, the mixture undergoes a sophisticated purification sequence involving vacuum distillation and solvent exchange to remove residual amines and methanol to levels below 0.2%. This rigorous control over the reaction endpoint and purification parameters is what enables the production of high-purity bactericide intermediates suitable for sensitive applications in water treatment and coatings.
How to Synthesize N,N'-Dialkyl-3,3'-dithiodipropionamide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for executing this one-pot transformation with high fidelity and reproducibility in an industrial setting. The process begins with the careful mixing of methyl acrylate, sulfur, and water, followed by the sequential introduction of gaseous reagents under strict temperature monitoring to manage the exothermic nature of the vulcanization. Once the polysulfide intermediate is formed and verified, the reduction step is initiated by adding sodium sulfite, which requires precise pH and temperature management to ensure complete conversion to the disulfide oil layer without degrading the sensitive sulfur-sulfur bond. The final aminolysis stage demands the slow addition of the alkylamine methanol solution to control the reaction rate and prevent localized overheating, which could lead to side reactions or impurity formation. Detailed standardized synthetic steps see the guide below.
- Perform vulcanization of methyl acrylate with sulfur, ammonia, and hydrogen sulfide at 10-20°C to form polysulfide intermediates.
- Execute reduction using sodium sulfite at 40-60°C to convert polysulfides into dimethyl 3,3'-dithiodipropionate oil layer.
- Conduct aminolysis with alkylamine methanol solution at 10-30°C followed by solvent exchange and vacuum distillation for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis method presents a compelling value proposition centered around operational efficiency and waste minimization. By eliminating the need for intermediate isolation, filtration, and drying, the process significantly reduces the man-hours and energy consumption associated with batch processing, leading to a more streamlined production schedule. The ability to recycle the aqueous layer components, specifically sodium thiosulfate, transforms a potential waste liability into a recoverable asset, thereby lowering the overall cost of goods sold through improved material utilization. Furthermore, the compatibility of the solvent residue in the final product with downstream synthesis solvents means that the intermediate can be fed directly into the next manufacturing stage without additional purification or solvent swapping. This seamless integration reduces the inventory holding time and minimizes the risk of contamination during transfer, enhancing the overall reliability of the supply chain for critical biocide ingredients.
- Cost Reduction in Manufacturing: The elimination of solid-liquid separation and drying steps removes the need for expensive filtration equipment and the energy costs associated with thermal drying processes. By conducting the reaction in a single vessel or a tightly coupled series of vessels, the capital expenditure for plant infrastructure is optimized, and the operational expenditure is lowered due to reduced utility consumption. The recovery of valuable solvents and the recycling of byproducts like sodium thiosulfate further contribute to a leaner cost structure, allowing manufacturers to offer competitive pricing without compromising on quality margins. Additionally, the high conversion rates and minimized side reactions ensure that raw material costs are maximized, reducing the frequency of replenishment orders and stabilizing the cost base against market volatility.
- Enhanced Supply Chain Reliability: The simplified operational workflow reduces the number of potential failure points in the manufacturing process, leading to more consistent batch-to-batch quality and predictable delivery timelines. Since the process relies on commodity chemicals like methyl acrylate and sulfur, the risk of raw material shortages is mitigated compared to routes requiring specialized or scarce reagents. The robustness of the one-pot method allows for flexible production scheduling, enabling manufacturers to respond quickly to fluctuations in market demand for bactericides used in water treatment and personal care products. This agility ensures that downstream customers experience fewer disruptions and can maintain their own production schedules with greater confidence, fostering stronger long-term partnerships between suppliers and end-users.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as the reaction conditions are mild and do not require extreme pressures or temperatures that would pose engineering challenges at larger volumes. The reduction in three-waste discharge, particularly the minimization of organic solvent waste and the conversion of inorganic byproducts into reusable forms, aligns perfectly with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden and the costs associated with waste disposal permits and treatment facilities, making the facility more sustainable in the long term. Moreover, the green nature of the synthesis enhances the brand reputation of the manufacturer, appealing to eco-conscious clients in the coatings and agrochemical sectors who prioritize sustainable sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of adopting this technology for large-scale production of bactericide intermediates.
Q: What are the primary advantages of the one-pot synthesis method for N,N'-dialkyl-3,3'-dithiodipropionamide?
A: The one-pot method eliminates complex solid-liquid separation and drying steps required in traditional processes, significantly simplifying operations and reducing waste discharge while allowing for the recycling of byproducts like sodium thiosulfate.
Q: How does this process improve environmental compliance in biocide manufacturing?
A: By enabling the recycling of reaction solvents and converting waste streams into usable sodium thiosulfate, the process minimizes three-waste discharge and aligns with stringent green chemistry standards for industrial production.
Q: Can this intermediate be directly used for synthesizing 3-isothiazolinone bactericides?
A: Yes, the solvent residue in the final product is compatible with solvents used for synthesizing 3-isothiazolinones, allowing for direct feeding into the subsequent cyclization reaction without additional solvent exchange steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-Dialkyl-3,3'-dithiodipropionamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of effective and safe bactericides for global markets. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of N,N'-dialkyl-3,3'-dithiodipropionamide meets the exacting standards required for downstream synthesis of 3-isothiazolinones. Our commitment to technical excellence means that we can support your R&D efforts with custom synthesis solutions that leverage the latest advancements in one-pot methodology.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your manufacturing costs and supply chain stability. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for reliable agrochemical intermediate supplier partnerships.
