Advanced Catalytic Synthesis of Mono-Vinyl Dialkylphosphinic Acids for High-Performance Flame Retardants
Introduction to Patent CN102177169A
The chemical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing high-performance polymer additives, particularly flame retardants. Patent CN102177169A introduces a groundbreaking method for producing mono-vinylfunctionalized dialkylphosphinic acids, their salts, and esters, which serve as critical intermediates in the manufacture of advanced flame-retardant polymers. This technology addresses long-standing challenges in the field by providing a route that achieves high space-time yields without the need for interfering halogen compounds as reactants. The innovation lies in a sophisticated two-step catalytic sequence that allows for precise functionalization of the phosphorus backbone, enabling the creation of reactive flame retardants that can be chemically bonded into polymer matrices. For R&D directors and procurement specialists, this represents a significant opportunity to enhance product performance while simplifying the supply chain for specialized chemical intermediates.
Furthermore, the versatility of this synthesis method allows for the production of a wide range of derivatives, including aluminum, calcium, zinc, and titanium salts, which are essential for meeting diverse regulatory and performance standards in the plastics industry. The patent details specific conditions under which these reactions proceed with high efficiency, utilizing transition metal catalysts that can be recovered and reused, thereby aligning with modern green chemistry principles. By adopting this methodology, manufacturers can produce flame retardants that offer superior thermal stability and mechanical properties compared to traditional additive types. The following analysis delves into the mechanistic details and commercial implications of this proprietary technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vinyl-functionalized phosphorus compounds has been plagued by significant technical hurdles that impact both cost and quality. Traditional methods often rely on halogenated precursors or harsh reaction conditions that generate substantial amounts of hazardous waste and difficult-to-remove by-products. These legacy processes frequently suffer from low atom economy and poor selectivity, leading to complex purification steps that drive up manufacturing costs and extend lead times. Moreover, the presence of residual halogens in the final product can compromise the thermal stability of the resulting polymers and raise environmental compliance issues, particularly in regions with strict regulations on halogenated flame retardants. The inability to easily separate the target compound from the reaction mixture often results in lower overall yields and inconsistent batch-to-batch quality, which is unacceptable for high-performance applications in automotive and electronics sectors.
The Novel Approach
In stark contrast, the method described in CN102177169A utilizes a direct catalytic addition of phosphinic acid sources to olefins, followed by a vinylization step with acetylenic compounds. This approach completely bypasses the need for halogenated intermediates, resulting in a cleaner reaction profile and a much simpler downstream processing workflow. The use of transition metal catalysts, such as rhodium or palladium complexes, enables the reaction to proceed under milder conditions with high regioselectivity, ensuring that the vinyl group is introduced precisely where needed for optimal reactivity. This novel pathway not only improves the purity of the final mono-vinyl functionalized dialkylphosphinic acid but also significantly enhances the space-time yield of the process. By eliminating the interference of halogen compounds, the method facilitates easier product isolation through standard techniques like distillation or crystallization, thereby reducing energy consumption and operational complexity.
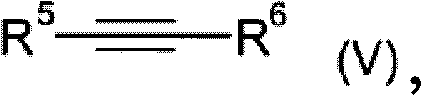
Mechanistic Insights into Transition Metal-Catalyzed Hydrophosphination and Vinylization
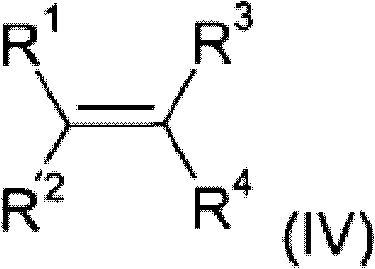
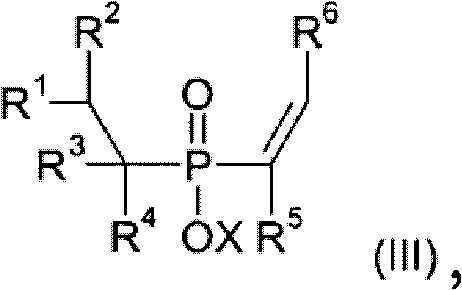
The core of this innovative synthesis lies in the dual-catalyst system employed in steps A and B. In the first step, a phosphinic acid source reacts with an olefin (IV) in the presence of Catalyst A, which is typically a transition metal complex from the 7th or 8th subgroup, such as rhodium, nickel, or palladium. This hydrophosphination reaction involves the activation of the P-H bond by the metal center, followed by insertion of the olefin into the metal-phosphorus bond. The choice of ligand is critical here; the patent highlights the effectiveness of phosphine ligands, including monodentate and bidentate varieties like 1,2-bis(diphenylphosphino)ethane or xanthene-based diphosphines. These ligands modulate the electronic and steric environment around the metal, enhancing catalytic activity and stability. The reaction can be conducted in various solvents, including water, alcohols, or hydrocarbons, offering flexibility for different scale-up scenarios.
Following the formation of the alkylphosphonic acid derivative (II), the second step involves its reaction with an acetylenic compound (V) using Catalyst B to yield the mono-vinyl functionalized product (III). This vinylization step is equally dependent on the precise selection of the transition metal catalyst and ligand system to ensure high conversion and selectivity. The mechanism likely involves the coordination of the alkyne to the metal center, followed by migratory insertion and reductive elimination to form the C-P bond with the vinyl group. Crucially, the patent emphasizes the ability to remove residual transition metals using metal scavengers, ensuring the final product meets stringent purity specifications required for electronic and automotive applications. This rigorous control over the catalytic cycle and impurity profile is what distinguishes this method from prior art, providing a robust platform for the commercial production of high-value flame retardant intermediates.
How to Synthesize Mono-Vinyl Dialkylphosphinic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable intermediates with high efficiency and reproducibility. The process begins with the preparation of the alkylphosphonic acid precursor via hydrophosphination, followed by the vinylization reaction to introduce the reactive double bond. Detailed operational parameters, including temperature ranges from 20°C to 180°C and pressure conditions up to 100 bar, are specified to optimize reaction kinetics and safety. The use of specific metal scavengers, such as those based on silica or functionalized polymers, is recommended to reduce residual metal content to ppm levels, which is critical for downstream polymerization processes. While the patent provides extensive experimental data, the following guide summarizes the key operational phases for implementation.
- React a phosphinic acid source with olefins in the presence of a transition metal catalyst (Catalyst A) to form alkylphosphonic acid derivatives.
- Convert the resulting alkylphosphonic acid derivative with acetylenic compounds using a second transition metal catalyst (Catalyst B) to introduce the vinyl functionality.
- Optionally react the mono-vinyl functionalized product with metallic compounds or alcohols to form specific salts or esters for polymer integration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers substantial strategic benefits beyond mere technical performance. The primary advantage lies in the significant simplification of the raw material supply chain, as the process utilizes commodity chemicals like ethylene, propylene, and acetylene, which are globally available and price-stable. This reduces exposure to volatile specialty chemical markets and ensures a consistent supply of feedstocks for large-scale production. Furthermore, the elimination of halogenated reagents removes the need for expensive corrosion-resistant equipment and hazardous waste disposal services, leading to a drastic reduction in capital expenditure and operating costs. The high selectivity of the catalyst system minimizes the formation of by-products, which translates to higher overall yields and less material waste, directly impacting the bottom line through improved resource efficiency.
- Cost Reduction in Manufacturing: The transition to this halogen-free catalytic route eliminates the costly steps associated with handling and disposing of hazardous halogenated waste streams. By avoiding the use of expensive and regulated halogenated intermediates, manufacturers can achieve substantial cost savings in raw material procurement and waste management. Additionally, the high space-time yield of the process means that existing reactor capacity can produce significantly more product per unit of time, effectively lowering the fixed cost per kilogram of the final flame retardant intermediate. The ability to recover and reuse transition metal catalysts further contributes to long-term cost optimization, making the process economically viable even at fluctuating metal prices.
- Enhanced Supply Chain Reliability: Relying on widely available olefins and acetylenic compounds as starting materials mitigates the risk of supply disruptions often associated with niche specialty chemicals. The robustness of the catalytic system allows for flexible production scheduling and rapid scale-up to meet surging demand in the automotive and electronics industries. Moreover, the simplified purification process reduces the lead time required to bring batches to market, ensuring that customers receive high-purity products without delay. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery schedules of global polymer manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor types such as stirred tanks and trickle bed reactors that are common in the fine chemical industry. This facilitates a smooth transition from pilot plant to commercial production without the need for specialized infrastructure. From an environmental perspective, the halogen-free nature of the synthesis aligns perfectly with increasingly stringent global regulations on flame retardants, such as REACH and RoHS. This future-proofs the supply chain against regulatory bans and enhances the marketability of the final polymer products as eco-friendly and sustainable solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these aspects is essential for stakeholders evaluating the potential integration of these intermediates into their existing product portfolios.
Q: What are the primary advantages of this catalytic method over traditional halogenated routes?
A: This method eliminates the need for interfering halogen compounds as reactants, allowing for the production of high-purity products that are easier to separate and purify. It avoids the environmental and toxicity issues associated with halogenated flame retardants while maintaining high space-time yields.
Q: Which transition metals are suitable for Catalyst A and Catalyst B in this process?
A: The patent specifies that Catalyst A and B can be derived from transition metals of the 7th and 8th subgroups, including rhodium, nickel, palladium, platinum, and ruthenium. These metals can be used as salts, complexes, or supported on various carriers like aluminum oxide or activated carbon.
Q: How does this technology improve the supply chain for flame retardant intermediates?
A: By utilizing readily available olefins and acetylenic compounds as starting materials and employing robust transition metal catalysis, the process ensures high scalability and consistent quality. The ability to easily separate the catalyst and by-products enhances supply continuity and reduces lead times for high-purity polymer additives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mono-Vinyl Dialkylphosphinic Acid Supplier
As the demand for high-performance, halogen-free flame retardants continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM is essential for successful commercialization. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of mono-vinyl dialkylphosphinic acid derivative meets the highest standards of quality and consistency required by the global polymer industry.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis route to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalytic technology. We are ready to provide specific COA data and route feasibility assessments to support your R&D and supply chain planning, ensuring that you stay ahead in the competitive landscape of advanced polymer additives.
