Advanced Synthesis of 2-(2-Amino-5-Bromo-Benzoyl) Pyridine for Commercial Anesthetic Production
Advanced Synthesis of 2-(2-Amino-5-Bromo-Benzoyl) Pyridine for Commercial Anesthetic Production
The pharmaceutical landscape for intravenous anesthetics is undergoing a significant transformation with the emergence of Remimazolam, a short-acting GABAa receptor agonist that combines the safety profile of midazolam with the efficacy of propofol. At the heart of this therapeutic innovation lies the critical intermediate, 2-(2-amino-5-bromo-benzoyl) pyridine, whose efficient and safe production is paramount for meeting global demand. Patent CN110746345B discloses a groundbreaking synthetic methodology that addresses the severe safety and scalability limitations of prior art, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. This novel approach replaces hazardous cryogenic chemistry with a streamlined sequence involving MEM protection, Suzuki-Miyaura coupling, and oxidative rearrangement, thereby establishing a new benchmark for high-purity pharmaceutical intermediates manufacturing.
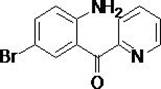
The strategic importance of this molecule cannot be overstated, as it serves as the foundational scaffold for next-generation anesthesia protocols used in surgical procedures, preoperative sedation, and ICU care. By leveraging the technical breakthroughs outlined in CN110746345B, manufacturers can now access a supply chain that is not only more reliable but also significantly safer, mitigating the risks associated with handling pyrophoric reagents and extreme temperatures. This report provides a deep technical analysis of this superior synthetic route, demonstrating how it aligns with the rigorous quality and safety standards required by top-tier multinational pharmaceutical companies seeking a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN110746345B, the industry standard for synthesizing 2-(2-amino-5-bromo-benzoyl) pyridine relied heavily on the methodology disclosed in patent EP 1183243, which presented substantial operational hazards and logistical bottlenecks. This legacy process necessitates the use of n-butyllithium, a highly pyrophoric organolithium reagent that poses severe fire and explosion risks upon exposure to air or moisture, requiring specialized inert atmosphere handling and extensive safety protocols. Furthermore, the reaction must be conducted at cryogenic temperatures of approximately -40°C, demanding energy-intensive refrigeration equipment and imposing strict limitations on reactor size and batch throughput. The workup procedure involves concentrating the reaction mixture to a black oil followed by flash chromatography, a purification technique that is notoriously difficult to scale industrially due to high solvent consumption and low throughput efficiency. These factors collectively result in a fragile supply chain vulnerable to safety incidents and production delays, making the conventional route economically and operationally unsustainable for modern cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to the perilous legacy methods, the novel synthetic route introduced in CN110746345B employs a sophisticated six-step sequence that operates under mild, industrially friendly conditions, effectively eliminating the need for cryogenic cooling and pyrophoric reagents. The process initiates with the protection of the phenolic hydroxyl group using MEMCl (2-methoxyethoxymethyl chloride), a strategy that masks reactive sites and enables subsequent selective functionalization without compromising the integrity of the aromatic system. The core carbon-carbon bond formation is achieved via a palladium-catalyzed Suzuki-Miyaura coupling, a reaction renowned for its tolerance to functional groups and ability to proceed at moderate temperatures between 30°C and 100°C. This shift from lithiation to cross-coupling chemistry not only enhances the safety profile by removing explosive hazards but also simplifies the purification workflow, allowing for straightforward extraction and crystallization techniques that are easily adaptable to multi-ton production scales. By utilizing common industrial solvents such as THF, DMF, and dichloromethane, the new method ensures that raw material sourcing remains stable and cost-effective, directly addressing the procurement challenges faced by supply chain managers.
Mechanistic Insights into Suzuki-Miyaura Coupling and Oxidative Rearrangement
The chemical elegance of this synthesis lies in its strategic use of the Suzuki-Miyaura cross-coupling reaction to construct the biaryl ketone skeleton, a pivotal step that dictates the overall purity and yield of the final intermediate. In this mechanism, a boronic acid derivative, generated in situ from the bromomethyl precursor via magnesium-mediated borylation, reacts with 2-bromopyridine in the presence of a palladium catalyst, specifically [1,1'-bis(diphenylphosphino)ferrocene]palladium(II) dichloride. The catalytic cycle involves the oxidative addition of the aryl halide to the Pd(0) species, followed by transmetallation with the organoboron compound and subsequent reductive elimination to forge the C-C bond, releasing the coupled product and regenerating the active catalyst. This mechanism is particularly advantageous because it avoids the harsh basic conditions often required for other coupling methods, thereby minimizing side reactions such as dehalogenation or polymerization that could compromise the impurity profile. The use of MEM protection during this stage is critical, as it prevents the interference of the phenolic oxygen with the catalyst or the boron species, ensuring high chemoselectivity and consistent batch-to-batch reproducibility essential for regulatory compliance.
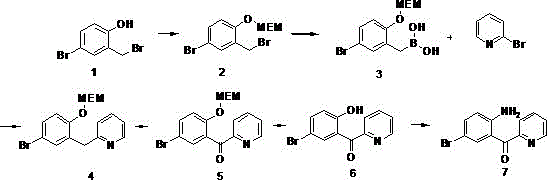
Following the coupling step, the synthesis proceeds through a selective oxidation of the benzylic methylene group to a ketone, utilizing tert-butyl hydroperoxide (TBHP) and iodine or cerium(IV) oxide as the oxidant system. This transformation is mechanistically distinct from traditional strong oxidants like chromic acid, offering a greener alternative that generates less toxic waste while maintaining high conversion rates. The final installation of the amino group is achieved through a clever rearrangement strategy involving 2-bromoisobutyramide and sodium hydroxide, which facilitates the migration of the acyl group and the introduction of the nitrogen functionality under controlled alkaline conditions. This sequence demonstrates a profound understanding of functional group interconversion, allowing for the precise assembly of the complex molecular architecture without the need for protecting group manipulations that would add unnecessary steps and cost. The result is a highly efficient pathway that maximizes atom economy and minimizes the generation of hazardous byproducts, aligning perfectly with the principles of green chemistry and sustainable manufacturing.
How to Synthesize 2-(2-Amino-5-Bromo-Benzoyl) Pyridine Efficiently
The implementation of this synthetic route requires precise control over reaction parameters to ensure optimal yields and purity, particularly during the sensitive oxidation and rearrangement phases. The process begins with the protection of 4-bromo-2-bromomethylphenol, followed by the formation of the boronic acid and the crucial Suzuki coupling step which builds the core heterocyclic framework. Subsequent oxidation converts the methylene linker to a carbonyl group, setting the stage for the final deprotection and rearrangement sequences that yield the target amino-ketone. While the general outline provides a roadmap, the specific stoichiometry, temperature profiles, and workup procedures are critical for success in a production environment. For a comprehensive, step-by-step technical guide including exact reagent quantities, reaction times, and purification protocols validated by pilot plant data, please refer to the standardized operating procedure below.
- Protect 4-bromo-2-bromomethylphenol with MEMCl in an aprotic solvent to form the MEM ether intermediate.
- Convert the bromomethyl group to a boronic acid using magnesium and trimethyl borate, followed by Suzuki coupling with 2-bromopyridine.
- Oxidize the methylene bridge to a ketone, remove the MEM protecting group, and perform a catalytic rearrangement with 2-bromoisobutyramide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from the conventional n-butyllithium route to the MEM-protected Suzuki coupling method represents a paradigm shift in risk management and cost efficiency. The elimination of cryogenic requirements (-40°C) removes the need for specialized low-temperature reactors and the associated high energy consumption, leading to a drastic simplification of the manufacturing infrastructure. Furthermore, the replacement of pyrophoric n-butyllithium with stable boronic acids and standard palladium catalysts significantly reduces the costs associated with hazardous material handling, storage, and disposal, while simultaneously lowering insurance premiums and regulatory compliance burdens. The use of commodity chemicals such as magnesium, trimethyl borate, and common organic solvents ensures that the supply chain is resilient against raw material shortages, providing a stable foundation for long-term production planning and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The new process eliminates the capital expenditure required for cryogenic cooling systems and the operational expenses linked to handling dangerous pyrophoric reagents. By utilizing standard stainless steel reactors and ambient to moderate temperature conditions, the method allows for larger batch sizes and higher throughput without proportional increases in overhead costs. The simplified purification steps, which rely on extraction and crystallization rather than resource-intensive flash chromatography, further drive down processing costs and solvent recovery expenses, resulting in substantial overall savings per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: Sourcing stable reagents like MEMCl and 2-bromopyridine is far more predictable than managing the logistics of n-butyllithium, which requires strict temperature control during transport and has a limited shelf life. The robustness of the Suzuki coupling reaction means that production schedules are less likely to be disrupted by minor fluctuations in reaction conditions or reagent quality, ensuring consistent on-time delivery to downstream API manufacturers. This reliability is crucial for maintaining continuous supply lines for critical anesthetic drugs, preventing costly stockouts and production halts in the final drug formulation stages.
- Scalability and Environmental Compliance: The synthetic route is inherently designed for scale-up, avoiding unit operations that are difficult to translate from the laboratory to the plant, such as low-temperature additions and column chromatography. The reduced generation of hazardous waste and the use of recoverable solvents align with increasingly stringent environmental regulations, facilitating easier permitting and community acceptance of manufacturing sites. This environmental stewardship not only future-proofs the production facility against regulatory changes but also enhances the corporate sustainability profile, a key metric for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-(2-amino-5-bromo-benzoyl) pyridine, derived directly from the experimental data and comparative analysis provided in patent CN110746345B. These insights are intended to clarify the operational benefits and technical feasibility of adopting this novel manufacturing route for commercial applications. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their existing supply chains.
Q: Why is the new synthesis method safer than the conventional n-butyllithium route?
A: The conventional method requires cryogenic conditions (-40°C) and pyrophoric n-butyllithium, posing significant explosion and handling risks. The new method utilizes standard Suzuki coupling conditions at mild temperatures (30-100°C) with stable reagents, drastically improving operational safety.
Q: What is the overall yield efficiency of this synthetic pathway?
A: The patented process achieves a robust total yield of approximately 53% to 55% over six steps, with individual step yields often exceeding 90%, ensuring high material throughput for commercial manufacturing.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method avoids specialized cryogenic equipment and uses common industrial solvents like THF and DMF. The mild reaction conditions and simplified purification steps make it highly adaptable for metric-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Amino-5-Bromo-Benzoyl) Pyridine Supplier
As the global demand for Remimazolam continues to surge, securing a dependable source of high-quality intermediates is critical for maintaining competitive advantage in the anesthetic market. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging the advanced synthetic methodologies described in CN110746345B to deliver 2-(2-amino-5-bromo-benzoyl) pyridine with unmatched consistency and purity. Our state-of-the-art facilities are equipped to handle complex multi-step syntheses, boasting extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the exacting standards required for pharmaceutical grade materials, providing our partners with the confidence needed to accelerate their own drug development timelines.
We invite forward-thinking pharmaceutical companies to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of this superior synthetic route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to our process can improve your bottom line. Contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us partner with you to bring safer, more effective anesthetics to patients worldwide.
