Advanced Catalytic Oxidation for 3-Methyl-1-Phenyl-1H-1,2,4-Triazole-5(4H)-One Manufacturing
Introduction to Patent CN112225705B and Strategic Value
The global agrochemical sector is constantly seeking more sustainable and cost-effective pathways for producing critical herbicide intermediates, and patent CN112225705B represents a significant technological leap in this domain. This intellectual property discloses a novel preparation method for 3-methyl-1-phenyl-1H-1,2,4-triazole-5(4H)-one, a pivotal building block for the synthesis of sulfentrazone, a high-value protoporphyrinogen oxidase (PPO) inhibitor herbicide. The core innovation lies in replacing traditional, hazardous stoichiometric oxidants with a catalytic system utilizing vanadium pentoxide and molecular oxygen from air. For R&D directors and procurement strategists, this shift is not merely a chemical curiosity but a fundamental restructuring of the supply chain economics, offering a reliable agrochemical intermediate supplier pathway that mitigates regulatory risks associated with chlorinated waste streams.
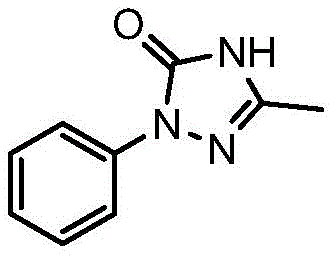
Understanding the molecular architecture of 3-methyl-1-phenyl-1H-1,2,4-triazole-5(4H)-one is essential for appreciating the synthetic challenges involved. As depicted in the structural diagram, the triazolinone ring system requires precise construction to ensure biological efficacy in the final herbicide formulation. The patent highlights that while sulfentrazone sales have grown substantially, reaching billions in annual value, the bottleneck has often been the efficient production of this specific ketone intermediate. By adopting the methodology outlined in CN112225705B, manufacturers can align their production capabilities with the increasing market demand for broad-spectrum, low-toxicity herbicides, ensuring a steady flow of high-purity materials for downstream formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 3-methyl-1-phenyl-1H-1,2,4-triazole-5(4H)-one has relied heavily on oxidation using sodium hypochlorite (NaClO). While this conventional route offers a degree of operational familiarity, it is fraught with significant logistical and environmental drawbacks that modern supply chains can ill afford. The reliance on sodium hypochlorite necessitates stringent quality control measures, as the stability and concentration of the oxidant can vary, leading to inconsistent reaction outcomes and potential safety hazards. Furthermore, the involvement of chlorine-based chemistry inherently generates substantial amounts of saline wastewater, imposing heavy burdens on effluent treatment facilities and escalating the overall cost of compliance with environmental regulations.
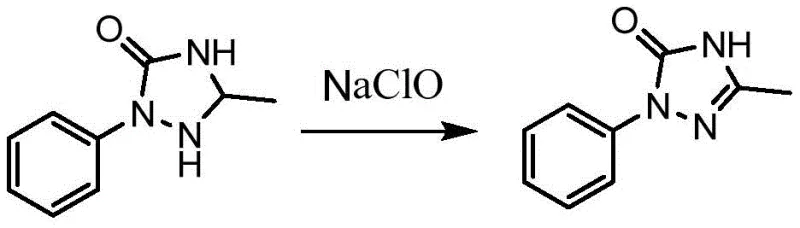
Beyond the environmental impact, the conventional sodium hypochlorite route suffers from low equipment utilization rates due to the complex handling requirements of hazardous substances. The need for specialized corrosion-resistant equipment and rigorous safety protocols to manage chlorine exposure slows down production cycles and increases capital expenditure. For procurement managers focused on cost reduction in agrochemical manufacturing, these hidden costs associated with waste disposal, safety monitoring, and equipment maintenance erode profit margins. The process is essentially a stoichiometric oxidation that consumes large quantities of reagents to produce relatively small amounts of product, making it economically inefficient in an era where green chemistry principles are becoming mandatory rather than optional.
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent utilizes a catalytic oxidation strategy driven by vanadium pentoxide (V2O5) and air. This paradigm shift transforms the oxidation step from a hazardous, waste-generating process into a streamlined, atom-economical reaction. By employing air as the terminal oxidant, the process eliminates the need for purchasing, storing, and handling dangerous chlorine sources, thereby drastically simplifying the operational workflow. The reaction proceeds smoothly at mild temperatures ranging from 40 to 50°C in tert-butyl alcohol, conditions that are far less energy-intensive and safer for plant personnel compared to the exothermic risks often associated with hypochlorite additions.
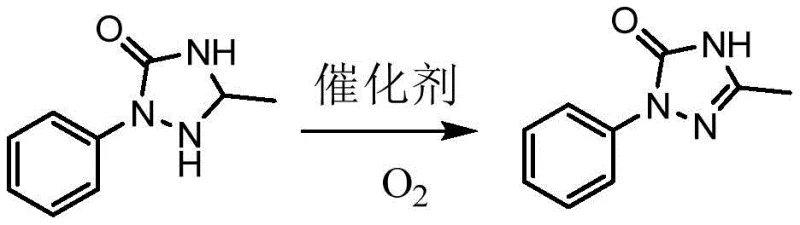
The implementation of this novel route offers immediate advantages in terms of equipment throughput and safety profiles. Since the oxidant is essentially free and unlimited (atmospheric air), the supply chain vulnerability associated with specialized chemical reagents is removed. The use of a heterogeneous or soluble vanadium catalyst allows for high turnover numbers, meaning a small amount of catalyst can process large batches of substrate, directly contributing to raw material cost savings. Moreover, the simplicity of the workup—concentration followed by water washing—suggests a robust process that minimizes solvent loss and maximizes recovery, addressing the critical need for high equipment utilization rates in large-scale commercial production.
Mechanistic Insights into V2O5-Catalyzed Aerobic Oxidation
From a mechanistic perspective, the utilization of vanadium pentoxide as a catalyst for the aerobic oxidation of triazolinone derivatives represents a sophisticated application of transition metal chemistry. Vanadium, existing in multiple oxidation states, acts as an electron transfer mediator that facilitates the activation of molecular oxygen. In this catalytic cycle, the vanadium species likely interacts with the substrate to form a reactive intermediate, which is then oxidized by oxygen from the air, regenerating the active catalyst species. This mechanism avoids the formation of chlorinated byproducts that are typical of hypochlorite oxidations, thereby simplifying the impurity profile of the crude product. For R&D teams, understanding this redox cycle is crucial for optimizing reaction parameters such as oxygen flow rate and catalyst loading to maximize efficiency.
Impurity control is another critical aspect where this catalytic mechanism excels. Traditional chlorination routes often lead to over-chlorination or the formation of N-chloro intermediates, which are unstable and difficult to remove, potentially affecting the stability of the final herbicide. The V2O5-catalyzed pathway, being non-halogenated, inherently prevents the introduction of chlorine atoms into the molecular scaffold. This results in a cleaner reaction mixture where the primary impurities are likely unreacted starting materials or minor oxidation byproducts that are easily separated during the aqueous wash step. The ability to achieve a product content of 93-95% directly from the crude isolation step underscores the selectivity of this catalytic system, reducing the need for extensive and costly recrystallization or chromatographic purification steps.
How to Synthesize 3-Methyl-1-Phenyl-1H-1,2,4-Triazole-5(4H)-One Efficiently
Implementing this synthesis requires precise control over reaction parameters to ensure reproducibility and safety on a commercial scale. The process begins with the dissolution of the substrate, 5-methyl-2-phenyl-1,2,4-triazole-3-one, in tert-butyl alcohol, which serves as both a solvent and a stabilizer for the reaction medium. The addition of the vanadium pentoxide catalyst must be done under continuous stirring to ensure homogeneous distribution before the introduction of the oxidant. The detailed standardized synthesis steps below outline the specific molar ratios, temperature controls, and gas flow rates required to achieve the reported yields of 85-90%, providing a clear roadmap for process engineers to follow during technology transfer.
- Charge 5-methyl-2-phenyl-1,2,4-triazole-3-one and tert-butyl alcohol solvent into a reactor equipped with stirring and temperature control.
- Add vanadium pentoxide catalyst (0.5%-1.5% mass ratio) under continuous stirring and begin heating the system.
- Introduce air at a controlled flow rate (20-30 mL/min) and maintain the reaction temperature between 40-50°C for 4-10 hours.
- Upon completion, concentrate the reaction solution and wash the resulting solid with water to isolate the product with 85-90% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the V2O5-catalyzed process translates into tangible strategic benefits that extend beyond simple chemical yield. The elimination of sodium hypochlorite removes a volatile cost component from the bill of materials, as the price of industrial bleach can fluctuate based on chlorine market dynamics. More importantly, the shift to air oxidation decouples the production process from the supply constraints of hazardous oxidants, ensuring a more resilient and continuous manufacturing schedule. This reliability is paramount for maintaining the supply continuity of high-purity agrochemical intermediates, especially during peak seasons when demand for herbicides surges.
- Cost Reduction in Manufacturing: The economic argument for this new process is compelling, primarily driven by the drastic reduction in waste treatment costs. By avoiding the generation of massive volumes of saline wastewater, manufacturers can significantly lower their environmental compliance expenditures and avoid the capital investment required for advanced effluent treatment plants. Additionally, the use of air as a free oxidant and the high catalyst efficiency mean that the variable cost per kilogram of product is substantially reduced, allowing for more competitive pricing in the global market without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the use of commodity chemicals. Tert-butyl alcohol and vanadium pentoxide are widely available industrial chemicals with stable supply lines, unlike specialized oxidants that may face logistical bottlenecks. The mild reaction conditions (40-50°C) also reduce the energy load on the facility, lowering utility costs and minimizing the risk of thermal runaway incidents that could halt production. This stability ensures that delivery timelines to downstream formulators are met consistently, strengthening the manufacturer's reputation as a dependable partner.
- Scalability and Environmental Compliance: Scalability is inherent in the design of this catalytic process, which relies on gas-liquid mass transfer rather than complex batch additions of hazardous solids or liquids. The simplicity of the operation allows for easy scale-up from pilot plants to multi-ton reactors with minimal re-engineering. Furthermore, the "green" nature of the process, characterized by zero chlorine usage and minimal waste, aligns perfectly with the increasingly stringent environmental regulations faced by the fine chemical industry, future-proofing the manufacturing asset against regulatory tightening.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and economic impact for potential adopters. Understanding these nuances is vital for stakeholders evaluating the transition from legacy chlorination methods to this advanced aerobic oxidation platform.
Q: What are the primary advantages of the V2O5 catalytic method over traditional sodium hypochlorite oxidation?
A: The V2O5 catalytic method eliminates the use of hazardous chlorine sources and sodium hypochlorite, significantly reducing wastewater generation and safety risks associated with handling strong oxidants. It utilizes air as a benign oxidant, leading to a greener process with lower environmental compliance costs.
Q: What yield and purity levels can be expected from this catalytic oxidation process?
A: According to patent data, the process consistently achieves a yield of 85-90% and a product content (purity) of 93-95%. These metrics demonstrate high efficiency suitable for industrial scale-up without compromising quality.
Q: Is the vanadium pentoxide catalyst difficult to remove from the final product?
A: The process involves a simple workup where the reaction solution is concentrated and the solid product is washed with water. This straightforward isolation step suggests that catalyst removal and product purification are manageable within standard pharmaceutical manufacturing protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methyl-1-Phenyl-1H-1,2,4-Triazole-5(4H)-One Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the long-term sustainability of the agrochemical industry. Our team of expert chemists and process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the V2O5-catalyzed oxidation of triazolinones can be seamlessly integrated into your supply chain. We are committed to delivering materials that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-methyl-1-phenyl-1H-1,2,4-triazole-5(4H)-one supports the high efficacy standards required for modern herbicide formulations.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific production needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic route for your operations. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and efficiency for your organization.
