Advanced Synthesis and Commercial Scalability of Arylamine-Substituted Benzodiindole Hole Transport Materials
Advanced Synthesis and Commercial Scalability of Arylamine-Substituted Benzodiindole Hole Transport Materials
The rapid evolution of perovskite solar cell technology demands hole transport materials that combine high efficiency with exceptional stability and processability. Patent CN110845503B introduces a groundbreaking class of arylamine-substituted benzodiindole organic hole transport materials, specifically designed to overcome the limitations of existing solutions in trans-planar perovskite architectures. These materials feature a rigid benzodiindole core substituted with electron-donating arylamine groups, resulting in a unique molecular architecture that offers superior energy level matching with perovskite absorbers. As a reliable display & optoelectronic materials supplier, understanding the structural nuances of these compounds is critical for optimizing device performance. The general structure, characterized by the benzodiindole backbone and variable R1 and R2 substituents, allows for fine-tuning of solubility and electronic properties to meet specific application requirements.
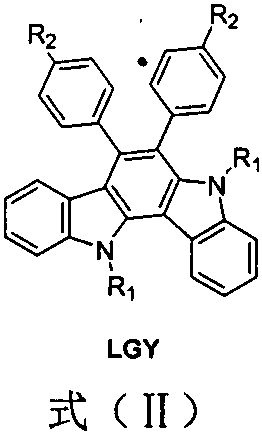
Beyond their structural elegance, these materials address critical failure modes in photovoltaic devices. Traditional polythiophene-based materials like PEEOT:PSS suffer from moisture absorption and acidity, which corrode transparent conductive electrodes and degrade device longevity. In contrast, the benzodiindole derivatives described in the patent are white powders with virtually no absorption in the visible light spectrum. This optical transparency is a game-changer, as it eliminates parasitic light absorption that typically reduces the effective photon flux reaching the active perovskite layer. Consequently, devices fabricated with these materials demonstrate significantly enhanced photoelectric conversion efficiency and operational stability, making them a prime candidate for the next generation of commercial solar modules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of hole transport layers for inverted perovskite solar cells has been hindered by the inherent drawbacks of industry-standard materials. Poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate) (PEDOT:PSS) has long been the default choice, yet its hygroscopic nature creates a persistent vulnerability to environmental moisture, leading to rapid device degradation. Furthermore, the acidic character of PSS actively corrodes the indium tin oxide (ITO) or fluorine-doped tin oxide (FTO) electrodes, creating interfacial defects that increase series resistance and lower fill factors. Additionally, many existing arylamine-based small molecules exhibit strong absorption in the visible region, effectively competing with the perovskite layer for incident photons. This spectral overlap results in a direct loss of photocurrent, capping the maximum achievable efficiency of the solar cell and limiting the commercial viability of the technology.
The Novel Approach
The innovative strategy outlined in the patent leverages a benzodiindole core to construct a new family of hole transport materials that fundamentally resolve these issues. By integrating electron-rich arylamine substituents such as triphenylamine, dimethoxytriphenylamine, or phenylcarbazole onto the benzodiindole scaffold, the resulting molecules achieve optimal highest occupied molecular orbital (HOMO) levels for efficient hole extraction. Crucially, these materials are colorless or white powders, ensuring maximum light transmission to the perovskite absorber layer. The synthesis route is remarkably streamlined, avoiding complex multi-step sequences that often plague organic semiconductor manufacturing. This simplicity not only accelerates R&D cycles but also lays the groundwork for cost reduction in electronic chemical manufacturing by minimizing unit operations and purification burdens.
Mechanistic Insights into Acid-Catalyzed Condensation and Suzuki Coupling
The synthesis of these high-performance materials relies on a sophisticated yet robust two-step sequence that ensures high purity and structural integrity. The first step involves an acid-catalyzed condensation reaction between N-alkylindole and 4,4'-dibromobenzil. In this transformation, p-toluenesulfonic acid acts as a potent proton donor in anhydrous toluene, facilitating the cyclization that forms the central benzodiindole ring system. This step is critical for establishing the rigid planar structure required for effective charge transport. The reaction proceeds under nitrogen protection at elevated temperatures, typically around 100°C to 110°C, ensuring complete conversion while minimizing side reactions. The resulting dibromo-intermediate serves as a versatile platform for further functionalization, with the bromine atoms positioned strategically for subsequent cross-coupling.
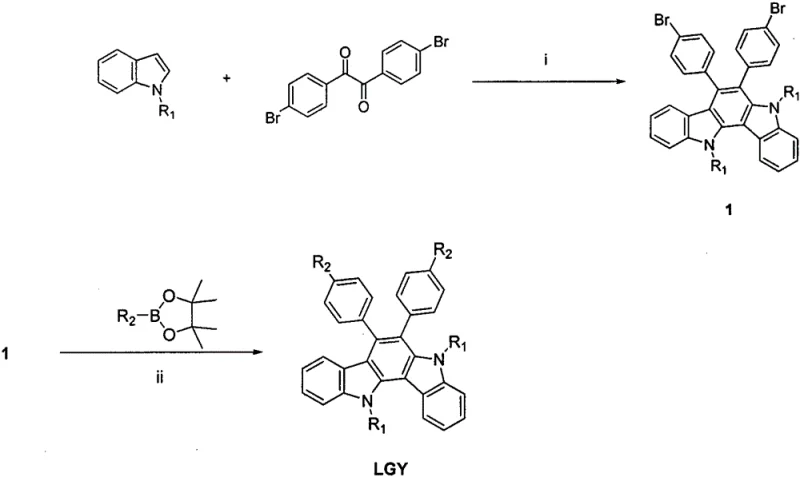
The second mechanistic phase employs a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction to install the diverse arylamine functionalities. By reacting the dibromo-intermediate with various arylamine boronate esters in the presence of a tetrakis(triphenylphosphine)palladium catalyst, the carbon-bromine bonds are selectively replaced with carbon-carbon bonds linking the electron-donating groups. This step is performed in 1,4-dioxane with an aqueous potassium carbonate base, providing a biphasic system that supports the catalytic cycle. The choice of ligands and conditions is optimized to prevent homocoupling of the boronate species, ensuring that the final product consists primarily of the desired disubstituted benzodiindole. Impurity control is managed through standard aqueous workups and recrystallization, yielding materials with the high purity specifications necessary for electronic applications.
How to Synthesize Arylamine-Substituted Benzodiindole Efficiently
Executing the synthesis of these hole transport materials requires precise control over reaction parameters to maximize yield and minimize impurities. The process begins with the rigorous drying of solvents and reagents to prevent hydrolysis of sensitive intermediates. Following the formation of the benzodiindole core, the Suzuki coupling step demands careful monitoring of temperature and catalyst loading to ensure complete consumption of the dibromo-precursor. The detailed standardized synthesis steps below outline the specific molar ratios, temperatures, and workup procedures validated in the patent examples to guide process engineers in replicating this high-value chemistry.
- Perform an acid-catalyzed condensation reaction between N-alkylindole and 4,4'-dibromobenzil in anhydrous toluene at elevated temperatures to form the dibromo-benzodiindole intermediate.
- Execute a Suzuki-Miyaura cross-coupling reaction by reacting the dibromo-intermediate with arylamine boronate esters using a palladium catalyst in 1,4-dioxane.
- Purify the final crude product through aqueous workup, organic extraction, drying over anhydrous sodium sulfate, and vacuum drying to obtain the high-purity hole transport material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this new class of hole transport materials offers substantial strategic benefits beyond mere performance metrics. The synthesis pathway is designed with manufacturability in mind, utilizing commodity chemicals that are readily available in the global market. This reliance on established supply chains mitigates the risk of raw material shortages that often plague specialty chemical sectors. Furthermore, the elimination of harsh acidic components like those found in PEDOT:PSS simplifies the downstream device assembly process, reducing the need for corrosion-resistant equipment and specialized handling protocols. These factors collectively contribute to a more resilient and predictable supply chain for photovoltaic manufacturers.
- Cost Reduction in Manufacturing: The economic viability of this technology is underpinned by the use of inexpensive starting materials such as N-alkylindoles and dibromobenzil, which are produced at scale for other industries. The synthetic route avoids the use of precious metal catalysts in the initial condensation step and utilizes standard palladium catalysts in the second step which can potentially be recovered. By eliminating the need for complex purification techniques like column chromatography on a large scale—relying instead on extraction and crystallization—the overall processing costs are significantly reduced. This streamlined approach translates directly into a lower cost of goods sold for the final hole transport material.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for flexible production scheduling. Since the key intermediate can be synthesized and stored prior to the final coupling step, manufacturers can respond rapidly to fluctuations in demand for specific derivatives (e.g., LGY-1 vs LGY-3). The use of common organic solvents like toluene and 1,4-dioxane further ensures that solvent supply is never a bottleneck. This flexibility enhances the reliability of the supply chain, ensuring that solar module producers can maintain continuous production lines without interruption due to material shortages.
- Scalability and Environmental Compliance: Scaling this chemistry from laboratory to commercial production is facilitated by the use of homogeneous reaction conditions that translate well to large reactors. The absence of highly toxic reagents or extreme pressure conditions simplifies the engineering controls required for safe operation. Moreover, the waste streams generated are typical of fine chemical synthesis and can be managed using standard treatment protocols. This alignment with environmental compliance standards reduces the regulatory burden and facilitates faster approval for new manufacturing facilities, accelerating the time-to-market for devices utilizing these advanced materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of arylamine-substituted benzodiindole hole transport materials in photovoltaic applications. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance expectations and integration strategies for engineering teams.
Q: What are the optical advantages of benzodiindole-based HTMs over traditional PEEOT:PSS?
A: Unlike PEEOT:PSS, which is hygroscopic and acidic, arylamine-substituted benzodiindoles are white powders with negligible absorption in the visible light region. This prevents competitive light absorption, allowing more photons to reach the perovskite layer, thereby enhancing photoelectric conversion efficiency and device stability.
Q: Is the synthesis route scalable for industrial production of hole transport materials?
A: Yes, the synthesis utilizes a robust two-step protocol involving standard condensation and Suzuki coupling reactions. The use of common solvents like toluene and 1,4-dioxane, along with commercially available starting materials like N-alkylindoles, facilitates straightforward commercial scale-up without requiring exotic reagents or extreme conditions.
Q: How does the thermal stability of LGY series materials compare to conventional organic semiconductors?
A: Thermogravimetric analysis indicates that these benzodiindole derivatives exhibit decomposition temperatures around 400°C. This high thermal stability ensures that the hole transport layer remains intact during device operation and encapsulation processes, significantly outperforming many less stable organic alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylamine-Substituted Benzodiindole Supplier
As the global demand for high-efficiency perovskite solar cells intensifies, securing a dependable source of advanced hole transport materials is paramount. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise synthesis requirements of complex organic semiconductors, ensuring that every batch meets stringent purity specifications. With rigorous QC labs and a commitment to process optimization, we deliver materials that consistently perform in high-stakes electronic applications, bridging the gap between academic innovation and industrial reality.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and device architecture. Contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate the commercialization of your next-generation photovoltaic technology with materials that offer the perfect balance of performance, stability, and value.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
