Advanced Iridium-Complex Electroluminescent Materials for High-Efficiency OLED Displays
The rapid evolution of organic light-emitting diode (OLED) technology has fundamentally transformed the display industry, driving an insatiable demand for materials that offer superior efficiency and longevity. Patent CN112759617B introduces a groundbreaking class of iridium-coordinated organic electroluminescent materials designed to address the critical bottlenecks in current phosphorescent emitter technology. Unlike conventional emitters that often struggle with stability or high driving voltages, these novel complexes utilize a specifically engineered heterocyclic ligand system to fine-tune physical properties such as emission wavelength and thermal stability. The core innovation lies in the general structural formula, where the central iridium atom is coordinated with cyclometalated ligands and a specific ancillary ligand, creating a robust molecular architecture capable of harvesting triplet excitons with exceptional quantum efficiency. 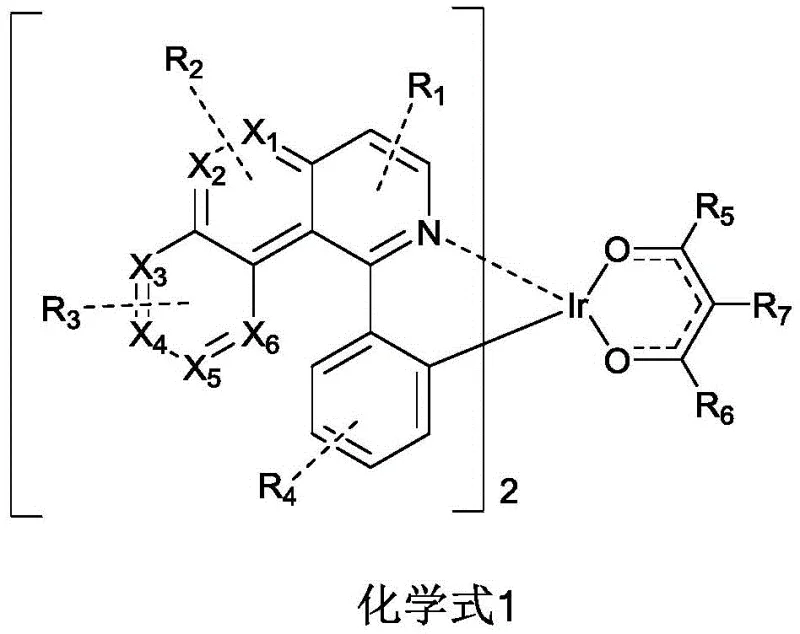
For R&D directors evaluating next-generation emissive layers, the structural versatility offered by this patent is paramount. The ability to independently modify substituents R1 through R7 allows for precise modulation of the Highest Occupied Molecular Orbital (HOMO) and Lowest Unoccupied Molecular Orbital (LUMO) energy levels. This tunability is essential for achieving pure color saturation and minimizing efficiency roll-off at high brightness levels, which are persistent challenges in red phosphorescent OLED development. By selecting specific heterocyclic rings for the ligand combination, the inventors have successfully created a material platform that not only reduces the driving voltage of the device but also significantly extends the service life compared to traditional iridium metal complexes currently dominating the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional phosphorescent OLED materials, such as the comparative compound Ir(bty)2acac often cited in prior art, frequently suffer from inherent limitations that hinder the performance of commercial displays. These legacy materials typically exhibit higher driving voltages, which directly translates to increased power consumption and heat generation in portable devices like smartphones and wearables. Furthermore, the stability of the metal-ligand bond in older generations of iridium complexes can be compromised under prolonged electrical stress, leading to faster degradation of the emissive layer and a noticeable decline in luminance over time. 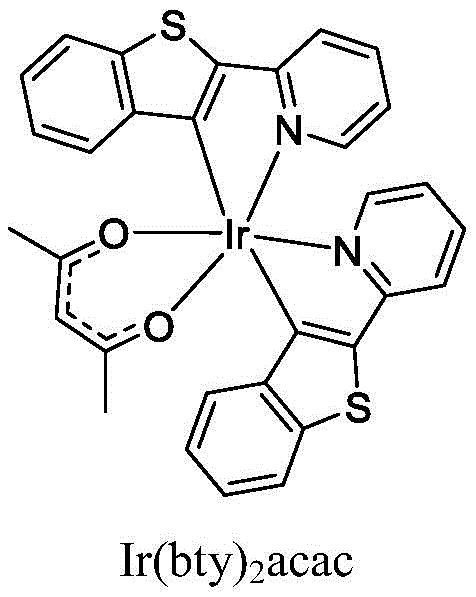
From a manufacturing perspective, the synthesis of some conventional high-performance emitters often involves complex multi-step routes with low overall yields or requires expensive, difficult-to-source precursors. This complexity creates supply chain vulnerabilities and inflates the cost of goods sold (COGS), making it challenging for panel manufacturers to reduce the final price of OLED TVs and monitors. Additionally, purification processes for these older materials sometimes fail to remove trace metal impurities effectively, which can act as quenching sites for excitons, thereby reducing the overall external quantum efficiency (EQE) of the finished device.
The Novel Approach
The approach detailed in CN112759617B represents a significant paradigm shift by focusing on ligand engineering to enhance both electronic performance and synthetic accessibility. By incorporating specific substituted aryl and heteroaryl groups into the cyclometalating ligand, the new materials achieve a more favorable balance between steric hindrance and electronic conjugation. This structural optimization prevents intermolecular aggregation, a common cause of concentration quenching, allowing for higher doping concentrations without sacrificing efficiency. The result is a material that operates at lower voltages while maintaining high brightness, directly addressing the power efficiency concerns of modern mobile electronics.
Moreover, the synthetic strategy employed for these novel complexes is inherently more robust and scalable. The use of readily available starting materials and straightforward reaction conditions means that the production process is less prone to batch-to-batch variability. This consistency is crucial for large-area display manufacturing, where uniformity of the emissive layer is non-negotiable. The patent demonstrates that these new materials outperform traditional benchmarks in key metrics such as T95 lifetime and current efficiency, providing a clear technical advantage for companies looking to differentiate their display products in a crowded marketplace.
Mechanistic Insights into Iridium-Catalyzed Cyclometalation and Coordination
The synthesis of these high-performance emitters relies on a classic yet highly optimized two-step organometallic pathway that ensures high regioselectivity and purity. The first stage involves the formation of a chloro-bridged iridium dimer, a critical intermediate that sets the stereochemistry for the final complex. In this step, the organic ligand precursor (Compound A) reacts with iridium trichloride trihydrate under reflux conditions. The presence of nitrogen atoms in the heterocyclic ring of Compound A directs the metallation to the ortho-position of the phenyl ring, forming a stable five-membered metallacycle. This cyclometalation is driven by the thermodynamic stability of the Ir-C and Ir-N bonds, which are stronger than the initial Ir-Cl bonds being displaced. 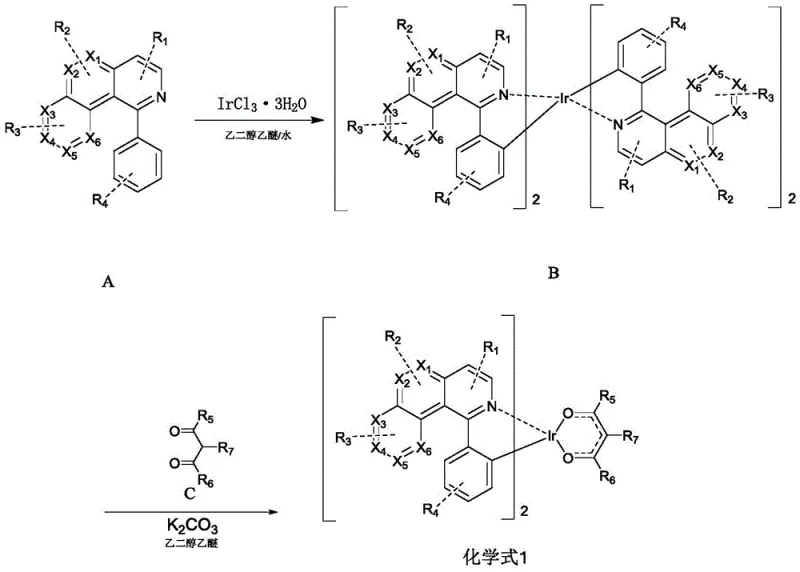
Following the formation of the bridged dimer (Intermediate B), the second step involves the cleavage of the chloride bridges and the coordination of the ancillary ligand (Compound C). This reaction is facilitated by the addition of a base, typically potassium carbonate, in a high-boiling solvent like ethylene glycol ether. The base neutralizes the HCl byproduct generated during the substitution, driving the equilibrium towards the formation of the neutral bis-cyclometalated iridium(III) complex. The choice of the ancillary ligand, often a beta-diketone derivative, plays a crucial role in tuning the solubility and sublimation properties of the final material, which is vital for vacuum deposition processes used in OLED fabrication. The rigorous control of reaction parameters, such as maintaining a temperature of 120°C for 24 hours, ensures complete conversion and minimizes the formation of homoleptic impurities.
How to Synthesize Iridium Complex OLED Dopants Efficiently
The preparation method outlined in the patent provides a clear roadmap for producing these advanced materials with high reproducibility. The process begins with the precise weighing of the ligand precursor and the iridium salt, followed by dissolution in a mixed solvent system of ethylene glycol ether and water. This specific solvent ratio is critical for solubilizing both the organic ligand and the inorganic metal salt, facilitating efficient contact between reactants. After the initial reflux to form the dimer, the intermediate is isolated via filtration and washing, removing unreacted starting materials and inorganic salts before proceeding to the final coupling step. For a detailed breakdown of the specific molar ratios, solvent volumes, and purification techniques required to achieve >99% purity, please refer to the standardized synthesis guide below.
- Step 1: Reflux compound A with IrCl3·3H2O in ethylene glycol ether/water (3:1 v/v) for 24 hours to form the chloro-bridged dimer intermediate B.
- Step 2: React intermediate B with compound C (beta-diketone derivative) and potassium carbonate in ethylene glycol ether at 120°C for 24 hours to yield the final iridium complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this new material class offers tangible strategic benefits beyond mere performance metrics. The synthetic route described eliminates the need for exotic catalysts or hazardous reagents that often complicate logistics and increase safety compliance costs. By utilizing common industrial solvents and bases, the manufacturing process integrates seamlessly into existing fine chemical production facilities, reducing the need for capital expenditure on new equipment. This compatibility significantly de-risks the supply chain, ensuring a steady flow of materials even during periods of global chemical shortages.
- Cost Reduction in Manufacturing: The streamlined two-step synthesis significantly lowers the operational complexity compared to multi-step alternatives found in older patents. By avoiding expensive transition metal catalysts for C-H activation and relying on direct cyclometalation with iridium chloride, the raw material costs are substantially optimized. Furthermore, the high yields reported in the examples, ranging from approximately 40% to over 50% for the final step, indicate a material-efficient process that minimizes waste disposal costs. The ability to purify the product through simple precipitation and standard chromatography rather than complex recrystallization or sublimation cycles further drives down the cost per gram, making high-end red phosphorescent dopants more economically viable for mass-market applications.
- Enhanced Supply Chain Reliability: The precursors required for this synthesis, such as substituted pyridines, phenylboronic acids, and beta-diketones, are commodity chemicals available from multiple global suppliers. This diversification of the raw material base prevents single-source bottlenecks that can plague the production of specialized OLED materials. The robustness of the reaction conditions, which tolerate standard industrial heating and stirring equipment, ensures that production can be scaled from kilogram to tonne quantities without encountering unforeseen engineering hurdles. This scalability guarantees long-term supply continuity for display manufacturers planning multi-year product roadmaps.
- Scalability and Environmental Compliance: The process operates at atmospheric pressure and moderate temperatures, reducing the energy footprint associated with high-pressure hydrogenation or cryogenic reactions. The use of ethylene glycol ether, while requiring proper handling, is a well-understood solvent in the industry with established recovery and recycling protocols, aligning with increasingly strict environmental regulations. The solid nature of the intermediates and final products simplifies storage and transportation, eliminating the risks associated with shipping unstable liquid precursors. This environmental and logistical efficiency positions the material as a sustainable choice for green manufacturing initiatives within the electronics sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these iridium complexes in OLED production lines. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy for technical decision-makers.
Q: What are the key performance advantages of the new iridium complexes over traditional materials?
A: The novel iridium complexes described in patent CN112759617B exhibit significantly lower driving voltages and improved luminous efficiency compared to traditional materials like Ir(bty)2acac, alongside extended operational lifetimes due to optimized ligand structures.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the preparation method utilizes standard reflux conditions and readily available raw materials such as IrCl3·3H2O and common beta-diketones, making it highly suitable for commercial scale-up without requiring exotic catalysts or extreme pressures.
Q: What purity levels can be achieved with this manufacturing route?
A: The patent examples demonstrate that through precipitation, filtration, and silica gel column chromatography, the final products consistently achieve HPLC purity greater than 99%, meeting the stringent requirements for high-end display applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iridium Complex OLED Dopant Supplier
As the demand for high-resolution, energy-efficient displays continues to surge, securing a dependable source of advanced emissive materials is critical for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this technological wave, leveraging deep expertise in organometallic chemistry to deliver premium OLED dopants. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of major panel manufacturers without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instruments to verify every batch against the highest industry standards.
We invite you to collaborate with us to optimize your display stack architecture using these next-generation iridium complexes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and device requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate the commercialization of your high-performance OLED products with confidence and precision.
