Advanced Polycyclic Aromatic Organic Compounds for High-Efficiency Blue OLED Displays
The rapid evolution of the organic light-emitting diode (OLED) industry demands continuous innovation in emissive materials, particularly for the challenging blue spectrum. Patent CN112961175B introduces a groundbreaking class of polycyclic aromatic organic compounds designed specifically to address the longevity and efficiency deficits often observed in blue OLED dopants. These novel molecules, characterized by their complex fused ring systems incorporating boron and nitrogen heteroatoms, represent a significant leap forward in optoelectronic material science. By stabilizing the excited states through rigid molecular architectures, these compounds facilitate high-efficiency radiative transitions while minimizing non-radiative decay pathways. 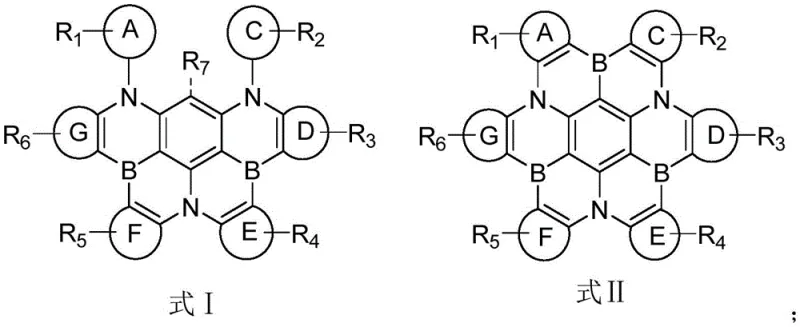 As depicted in the general structural formulas, the versatility of substituents R1 through R7 allows for fine-tuning of electronic properties, making them adaptable for various device configurations. For procurement specialists and R&D directors seeking a reliable OLED material supplier, understanding the synthesis and application of these advanced structures is critical for securing a competitive edge in the display market.
As depicted in the general structural formulas, the versatility of substituents R1 through R7 allows for fine-tuning of electronic properties, making them adaptable for various device configurations. For procurement specialists and R&D directors seeking a reliable OLED material supplier, understanding the synthesis and application of these advanced structures is critical for securing a competitive edge in the display market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing blue-emitting organic materials often struggle with balancing color purity against operational stability. Many conventional dopants suffer from rapid degradation under electrical stress, leading to shortened device lifespans that fail to meet the rigorous standards of modern consumer electronics. Furthermore, older synthetic routes frequently rely on harsh reaction conditions or unstable intermediates that complicate scale-up and introduce difficult-to-remove impurities. These impurities can act as quenching sites within the emissive layer, drastically reducing the overall quantum efficiency of the organic electroluminescent device. Additionally, the lack of rigid structural motifs in many legacy materials allows for excessive molecular vibration, which dissipates energy as heat rather than light. This inherent instability necessitates frequent replacement of display panels, driving up costs for end-users and creating sustainability challenges for manufacturers aiming for cost reduction in electronic chemical manufacturing.
The Novel Approach
The methodology outlined in patent CN112961175B overcomes these historical barriers through the strategic design of boron-nitrogen fused polycyclic frameworks. This novel approach leverages the electron-deficient nature of boron and the electron-rich character of nitrogen to create a push-pull electronic system that stabilizes the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) levels. The resulting compounds exhibit exceptional thermal stability and morphological stability, which are prerequisites for the vacuum deposition processes used in OLED fabrication. By incorporating specific heteroaryl rings into the core structure, the invention ensures that the materials emit pure blue light with high color saturation. Moreover, the synthetic pathway is designed to be robust and scalable, utilizing well-established catalytic systems that minimize side reactions. This strategic molecular engineering not only enhances the intrinsic photophysical properties of the dopant but also simplifies the purification process, ensuring that the final product meets the stringent purity specifications required for high-performance display applications.
Mechanistic Insights into Palladium-Catalyzed Amination and Boron Fusion
The synthesis of these high-performance polycyclic aromatic compounds relies on a sophisticated sequence of transition metal-catalyzed transformations, primarily centered around palladium-catalyzed C-N bond formation. The process initiates with the coupling of poly-brominated benzene derivatives with various amine substrates, facilitated by a catalytic system comprising tris(dibenzylideneacetone)dipalladium [Pd2dba3] and bulky phosphine ligands such as tri-tert-butylphosphine. This ligand choice is critical as it promotes the oxidative addition of the aryl halide and stabilizes the active palladium species, allowing the reaction to proceed efficiently at moderate temperatures ranging from 90°C to 120°C. The mechanism involves the formation of a palladium-amine complex which subsequently undergoes reductive elimination to forge the carbon-nitrogen bonds that link the peripheral aromatic rings to the central core. 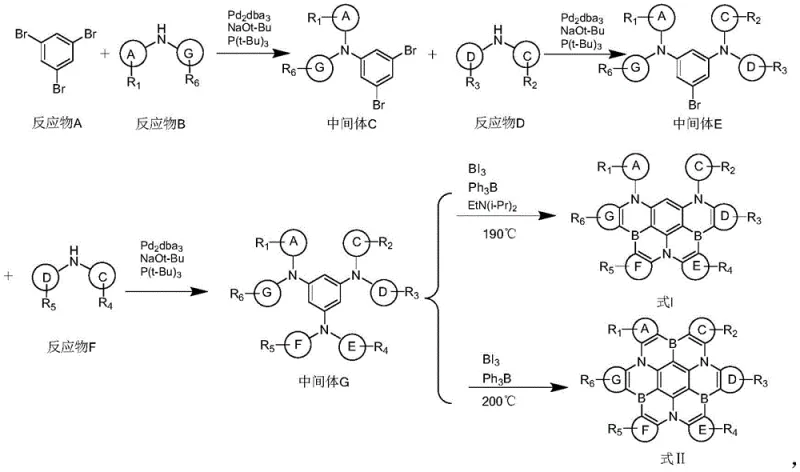 Following the assembly of the poly-amine precursor, the crucial cyclization step employs boron triiodide (BI3) and triphenylborane (Ph3B) in high-boiling solvents like 1,2,4-trichlorobenzene. This step effectively fuses the aromatic rings via boron bridges, locking the molecular conformation into a rigid planar structure that is essential for efficient charge transport and blue emission.
Following the assembly of the poly-amine precursor, the crucial cyclization step employs boron triiodide (BI3) and triphenylborane (Ph3B) in high-boiling solvents like 1,2,4-trichlorobenzene. This step effectively fuses the aromatic rings via boron bridges, locking the molecular conformation into a rigid planar structure that is essential for efficient charge transport and blue emission.
Controlling the impurity profile during this multi-step synthesis is paramount for achieving the high purity levels demanded by the semiconductor and display industries. The patent details specific purification protocols, including repeated recrystallization from solvent pairs such as toluene and ethanol or acetone and petroleum ether. These steps are designed to remove residual palladium catalysts, unreacted starting materials, and oligomeric by-products that could otherwise degrade device performance. The use of column chromatography on silica gel or filtration through diatomaceous earth further ensures the removal of particulate matter and metal residues. By rigorously monitoring the reaction progress via thin-layer chromatography (TLC) and optimizing the stoichiometry of reagents—typically maintaining molar ratios between 1:1 and 1:1.2—the process minimizes the formation of undesired side products. This meticulous attention to detail in the synthetic workflow guarantees that the final polycyclic aromatic organic compounds possess the electronic homogeneity necessary for uniform light emission across large-area display panels.
How to Synthesize Polycyclic Aromatic Organic Compounds Efficiently
The production of these advanced OLED materials requires precise control over reaction parameters and a deep understanding of organometallic chemistry. The patented process provides a clear roadmap for transforming simple aromatic precursors into complex, high-value emissive dopants. Operators must maintain strict inert atmosphere conditions, typically using nitrogen protection, to prevent the oxidation of sensitive palladium catalysts and boron reagents. The sequential addition of reagents, careful temperature ramping, and extended reaction times (often exceeding 16 hours for coupling steps) are essential to drive the reactions to completion. Detailed standardized synthesis steps see the guide below.
- Perform sequential palladium-catalyzed amination reactions between brominated benzene derivatives and amine substrates using Pd2dba3 and P(t-Bu)3 ligands to form poly-amine intermediates.
- Execute the final cyclization step by reacting the poly-amine intermediate with boron triiodide (BI3) and triphenylborane (Ph3B) in high-boiling solvents like 1,2,4-trichlorobenzene at temperatures between 180°C and 240°C.
- Purify the crude product through recrystallization using solvent systems such as toluene/ethanol or acetone/petroleum ether to achieve high-purity electronic grade materials.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN112961175B offers substantial strategic benefits beyond mere performance metrics. The synthetic route is constructed using widely available commodity chemicals, such as bromobenzenes and common aniline derivatives, which mitigates the risk of raw material shortages. Unlike processes that rely on bespoke or highly specialized reagents, this method utilizes standard industrial catalysts and solvents, facilitating easier sourcing and inventory management. The robustness of the palladium-catalyzed coupling reactions means that the process is less susceptible to minor fluctuations in reaction conditions, leading to more consistent batch-to-batch quality. This consistency is vital for maintaining yield stability in large-scale production environments, directly contributing to cost reduction in electronic chemical manufacturing by minimizing waste and rework.
- Cost Reduction in Manufacturing: The elimination of exotic reagents and the use of recyclable solvents significantly lower the direct material costs associated with production. Furthermore, the high selectivity of the catalytic system reduces the burden on downstream purification units, lowering energy consumption and solvent usage. By streamlining the synthesis into fewer distinct operational stages compared to alternative complex molecule constructions, the overall processing time is optimized, enhancing throughput without compromising quality.
- Enhanced Supply Chain Reliability: Since the starting materials are foundational building blocks in the fine chemical industry, supply continuity is inherently more secure. The process does not depend on single-source suppliers for critical intermediates, allowing for flexible procurement strategies. This diversification of the supply base ensures that production schedules remain uninterrupted even in the face of global logistical challenges, providing a stable flow of high-purity OLED materials to downstream device manufacturers.
- Scalability and Environmental Compliance: The reaction conditions, while requiring elevated temperatures, are well within the operational limits of standard stainless steel reactors used in fine chemical plants. The absence of highly toxic or environmentally hazardous by-products simplifies waste treatment protocols, ensuring compliance with increasingly stringent environmental regulations. This ease of scale-up from laboratory gram quantities to multi-ton annual production capacities makes the technology commercially viable for meeting the growing global demand for OLED displays.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these polycyclic aromatic compounds in OLED manufacturing. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance expectations and handling requirements. Understanding these details is essential for integrating these materials into existing production lines effectively.
Q: What are the key structural features of the polycyclic aromatic compounds in patent CN112961175B?
A: The compounds feature a rigid core structure defined by Formula I or II, containing multiple fused rings (A-G) with at least one heteroaryl ring. Crucially, they incorporate boron and nitrogen atoms within the fused system, which enhances electron transport and stability for blue emission.
Q: How does this synthesis method improve supply chain reliability for OLED manufacturers?
A: The synthesis utilizes commercially available starting materials such as bromobenzenes and common amines. The process relies on robust palladium catalysis and standard purification techniques like recrystallization, avoiding exotic reagents that could cause supply bottlenecks.
Q: What performance improvements do these materials offer in organic electroluminescent devices?
A: When used as blue dopants in an ADN host matrix, these compounds demonstrate significantly improved current efficiency and extended operational lifetime (T95) compared to conventional comparative compounds, alongside superior color purity and reduced efficiency roll-off.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polycyclic Aromatic Organic Compounds Supplier
As the demand for high-resolution, energy-efficient displays continues to surge, the need for superior blue emissive materials has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this technological shift, leveraging the innovations detailed in patent CN112961175B to deliver next-generation OLED dopants. Our facility is equipped with state-of-the-art synthesis and purification capabilities, ensuring that every batch meets the rigorous standards required for commercial display applications. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that we can support your volume requirements regardless of market fluctuations. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize advanced analytical techniques to verify the structural integrity and optical properties of every compound we ship.
We invite you to collaborate with us to optimize your OLED device architecture using these advanced polycyclic aromatic materials. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our streamlined supply chain can reduce your overall material costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to accelerate the development of longer-lasting, more efficient blue OLED displays that define the future of visual technology.
